Introduction: Enhancer of zeste homolog 2 (EZH2) and EZH1 are alternative subunits of polycomb repressive complex 2 that catalyze the tri-methylation of lysine 27 residue of histone H3. This histone modification epigenetically regulates gene expression and may play an important role in tumor progression. Valemetostat (DS-3201) is a potent and highly specific orally bioavailable dual inhibitor of EZH2 and EZH1 demonstrating anti-tumor activity against various hematological malignancies in preclinical studies (Honma D, et al. Cancer Sci 2017; Fujita S, et al. Leukemia 2018). Valemetostat demonstrated clinical activity as a novel oral therapeutic option for both B-cell and T-cell NHLs in the interim analysis of a phase I study (Maruyama D, et al. ASH 2017). Here we report the updated results of this phase I study focusing on ATL patients. ATL is a peripheral T-cell malignancy caused by human T-cell leukemia virus type I (HTLV-1), and is divided into aggressive and indolent subtypes. Aggressive ATL has an extremely poor prognosis, with a median survival time of only 8 to 10 months (Katsuya H, et al. Blood 2015). Although conventional chemotherapeutic agents, anti-CCR4 antibody mogamulizmab, and oral immunomodulator lenalidomide have been used for treatment of aggressive ATL patients, most patients eventually become resistant to treatment. In addition, allogenic hematopoietic stem cell transplantation is not available for elderly patients. Therefore, new therapeutic options are urgently needed.
Methods: This ongoing open-label, single-arm phase I study consists of the dose escalation part (NHLs including ATL and peripheral T-cell lymphoma [PTCL]) and the expansion part (ATL and PTCL). The drug was administered orally once daily (QD) continuously over 28-days (1 cycle) until disease progression or intolerance.
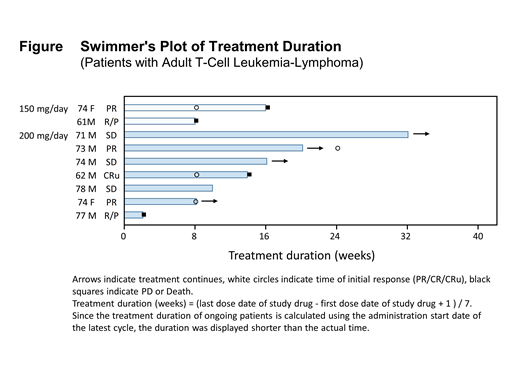
Results: Thirty-eight patients (15 females) with a median age of 69 (44-88) were enrolled in this study as of the data cut-off of 24 January 2019. Of 38 patients, 25 patients were enrolled in the dose-escalation part, and additional 13 patients (7 ATLs and 6 PTCLs) were treated in the expansion part with 200 mg of valemetostat. In all patients, adverse events (≥30%) on treatment with all grades included; platelet count decreased (73.7%), dysgeusia (52.6%), anemia (42.1%), lymphocyte count decreased (39.5%), neutrophil count decreased (39.5%), and white blood cell count decreased (39.5%). Preliminary efficacy was based on investigator's assessment with an objective response rate (ORR) of 47.2%. Fifteen patients were able to stay on valemetostat for more than 24 weeks with tumor shrinkage. Of the 9 ATL patients in the study (2 in dose escalation and 7 in dose expansion), baseline characteristics are as follows: age, median age 74 (range 61-78 yrs); sex, 7 males and 2 females; ATL subtype, 6 acute- and 3 lymphoma-subtypes; median number of prior therapies, 2 (range 1-8). Adverse events (≥30%) on treatment with all grades in the 9 ATL patients included; platelet count decreased (77.8%), dysgeusia (66.7%), neutrophil count decreased (44.4%), white blood cell count decreased (44.4%), anemia (33.3%), alopecia (33.3%), and dry skin (33.3%). No grade 4 and 5 adverse events were detected. Grade 3 adverse events included; white blood cell count decreased (33.3%), platelet count decreased (22.2%), neutrophil count decreased (22.2%), lymphocyte count decreased (11.1%), and anemia (11.1%), which are consistent with those in all population. Responses for the 9 ATL patients included; 1 unconfirmed complete remission (CRu), 3 partial remission (PR), and 3 stable disease (SD) (ORR = 44.4%; 4/9). Of the 6 prior mogamulizumab treated patients, 3 patients demonstrated a response (ORR = 50%; 3/6). Five of 9 ATL patients continued on valemetostat treatment for more than 12 weeks with tumor shrinkage (Figure). Four ATL patients continue on treatment. The latest study results will be presented.
Conclusion: The updated results of this ongoing phase I study showed that the oral EZH1/2 dual inhibitor valemetostat has demonstrated acceptable safety and promising preliminary efficacy in NHL.
In addition, the results of ATL patients, including mogamulizumab pretreated patients, showed promising clinical activity. A subsequent pivotal phase II study for ATL will soon be initiated.
Clinical trial information: NCT02732275
Ishitsuka:Novartis: Honoraria, Research Funding; sanofi: Honoraria; Celgene: Honoraria; Astellas Pharma: Honoraria, Research Funding; Pfizer: Honoraria; Takeda Pharmaceutical: Honoraria, Research Funding; Janssen Pharmaceutical: Honoraria; Janssen Pharmaceutical: Honoraria; Shire: Honoraria; Eisai: Honoraria, Research Funding; Otsuka Pharmaceutical: Honoraria; Mochida: Honoraria, Research Funding; Shire: Honoraria; Teijin Pharma: Research Funding; Sumitomo Dainippon Pharma: Honoraria, Research Funding; Astellas Pharma: Honoraria, Research Funding; Sumitomo Dainippon Pharma: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Genzyme: Honoraria; Genzyme: Honoraria; Eisai: Honoraria, Research Funding; Ono Pharmaceutical: Honoraria, Research Funding; Teijin Pharma: Research Funding; MSD: Research Funding; Yakult: Research Funding; Asahi kasei: Research Funding; MSD: Research Funding; Asahi kasei: Research Funding; Eli Lilly: Research Funding; Eli Lilly: Research Funding; Daiichi Sankyo: Consultancy, Honoraria; Daiichi Sankyo: Consultancy, Honoraria; Kyowa Hakko Kirin: Honoraria, Research Funding; mundiharma: Honoraria; Taiho Pharmaceutical: Honoraria, Research Funding; Alexion: Honoraria; Mochida: Honoraria, Research Funding; Takeda Pharmaceutical: Honoraria, Research Funding; mundiharma: Honoraria; Taiho Pharmaceutical: Honoraria, Research Funding; Otsuka Pharmaceutical: Honoraria; Ono Pharmaceutical: Honoraria, Research Funding; Yakult: Research Funding; Bristol-Myers Squibb: Honoraria; Chugai Pharmaceutical: Honoraria, Research Funding; Pfizer: Honoraria; Alexion: Honoraria; sanofi: Honoraria. Izutsu:Eisai, Chugai, Zenyaku: Honoraria; Chugai, Celgene, Daiichi Sankyo, Astra Zeneca, Eisai, Symbio, Ono, Bayer, Solasia, Zenyaku, Incyte, Novartis, Sanofi, HUYA Bioscience, MSD, Astellas Amgen, Abbvie, ARIAD, Takeda, Pfizer: Research Funding; Celgene: Consultancy; Eisai, Symbio, Chugai, Zenyaku: Research Funding; Kyowa Kirin, Eisai, Takeda, MSD, Chugai, Nihon Medi-physics, Janssen, Ono, Abbvie, Dainihon Sumitomo, Bayer, Astra Zeneca, HUYA Japan, Novartis, Bristol-Byers Squibb, Mundi, Otsuka, Daiichi Sankyo, Astellas, Asahi Kasei: Honoraria. Kusumoto:Chugai Pharmaceutical Co., Ltd.: Consultancy, Honoraria, Research Funding; Kyowa Kirin Co., Ltd.: Honoraria, Research Funding. Araki:Daiichi Sankyo: Employment. Adachi:Daiichi Sankyo: Employment. Yamashita:Daiichi Sankyo: Employment. Atsumi:Daiichi Sankyo: Employment. Tsukasaki:Eisai: Research Funding; Mundi Pharma: Honoraria; Chugai Pharmaceutical: Honoraria, Research Funding; Huya: Consultancy, Honoraria, Research Funding; Byer: Research Funding; Kyowa Kirin: Honoraria; Ono Pharmaceutical: Consultancy; Celgene: Honoraria, Research Funding; Daiichi Sankyo: Consultancy. Tobinai:Eisai: Honoraria, Research Funding; Kyowa Kirin: Honoraria, Research Funding; Daiichi Sankyo: Consultancy, Honoraria; Takeda Pharmaceutical: Consultancy, Honoraria, Research Funding; Chugai Pharmaceutical: Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Zenyaku Kogyo: Consultancy, Honoraria; Meiji Seika: Honoraria; Verastem: Honoraria; Solasia: Honoraria; Janssen Pharmaceutical: Honoraria, Research Funding; Yakult: Honoraria; AbbVie: Research Funding; Ono Pharmaceutical: Consultancy, Honoraria, Research Funding; Mundi Pharma: Consultancy, Honoraria, Research Funding; HUYA Bioscience: Consultancy, Honoraria; Bristol-Myers Squibb: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal