Introduction: Use of alloHCT is increasing in older patients, but the best means of determining alloHCT candidacy is uncertain. Geriatric assessment (GA), including validated geriatric metrics such as Instrumental Activities of Daily Living (IADL), offers promise to improve prognostication and decision making. We identified a multicenter retrospective cohort of older patients (pts) with pre-alloHCT GA data and examined the impact of GA impairments on post-alloHCT outcomes as reported to the Center for International Blood and Marrow Transplant (CIBMTR) registry.
Methods: Pts across 6 participating centers were included in the cohort who met the following inclusion criteria: first alloHCT performed in the U.S. from 2011-2017; age ≥50 years; consented to CIBMTR registry study; and GA incorporating an IADL score collected within 3 months prior to transplant. Additional pre-alloHCT geriatric metrics were collected where available and included: Medical Outcomes Study Physical Health score (MOS-PH), Timed Up and Go (TUG), and Blessed Orientation Memory Concentration (BOMC). Univariate regression analyses examined the effect of demographic, clinical, and geriatric covariates on overall survival (OS), progression-free survival (PFS) and non-relapse mortality (NRM). Variables significant at p<0.1 were included in multivariate models.
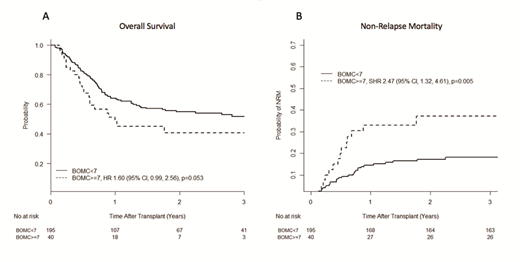
Results: Among 330 pts meeting inclusion criteria, median age was 63 (range 50-77), 60% were male, and 89% were Caucasian. AML and MDS accounted for 79% of transplant indications (46% and 33% respectively). Conditioning was myeloablative in 32%. Karnofsky performance status (KPS) was ≤ 80 in 27%, and 51% of patients had Hematopoietic Cell Transplant Comorbidity Index (HCTCI) ≥3. Median follow up was 24 months (range 3-61). GA metrics available for analysis included IADL in 100% of pts, MOS-PH in 70%, TUG in 83%, and BOMC in 71%. GA impairments, using published cutoffs, were frequent: of evaluable pts, 36% had IADL impairment (score <14), 57% had MOS-PH ≤ the cohort median of 80, 14% had TUG ≥ 13.5 seconds and 17% had cognitive impairment (BOMC score ≥7). In multivariate analysis (MVA), only BOMC ≥7 was significantly associated with inferior 1-year OS (hazard ratio [HR] 1.94, 95% confidence interval [CI] 1.14-3.31, p=0.01). IADL impairment was of marginal significance (HR 1.41, 95% CI 0.96-2.08, p=0.08). Similar findings were seen for 2-year OS (BOMC ≥7 HR 1.60, 95% CI 0.99-2.56, p=0.05; and IADL HR 1.33, 95% CI 0.96-1.86, p=0.09). With respect to PFS, high/very high Disease Risk Index score (HR 1.62, 95% CI 1.17-2.24, p=0.004) and BOMC ≥7 (HR 1.79, 95% CI 1.14-2.81, p=0.01) both were associated with inferior 2-year PFS. With respect to NRM, HCTCI ≥3 and BOMC ≥7 were the only significant predictors of increased NRM at both 1 and 2 years (subdistribution HR [SHR] 1.82, 95% CI 1.07-3.09, p=0.03 and SHR 2.41, 95% CI 1.29-4.53, p=0.006 respectively for 2-year NRM). Figure 1A and 1B show unadjusted OS and NRM according to the presence of cognitive impairment by BOMC ≥7. No GA metrics were predictive of relapse risk, and age was not associated with any post-alloHCT outcomes. In a pre-planned subgroup analysis of GA metrics in 224 patients aged ≥60 years, impaired cognition by BOMC ≥7 was the sole geriatric metric predictive of 2-year NRM (SHR 2.72, 95% CI 1.32-5.63, p=0.007).
Conclusion: In this multicenter study of patients ≥50 years undergoing alloHCT, pre-alloHCT cognitive impairment was significantly associated with inferior OS due to increased NRM. In contrast to prior single center studies, IADL impairment was not significantly associated with outcomes. GA validation studies, integrating more refined cognitive testing, should be pursued both to determine candidacy for alloHCT and to identify those likely to benefit from strategies to reduce NRM.
Olin:Daiichi Sankyo: Research Funding; Astellas: Research Funding; Genentech: Consultancy, Research Funding; Pfizer: Research Funding; Jazz Pharmaceuticals: Consultancy, Honoraria; Revolution Medicine: Consultancy; AstraZeneca: Research Funding; Clovis: Research Funding; Ignyta: Research Funding; MedImmune: Research Funding; Mirati Therapeutics: Research Funding; Novartis: Research Funding; Spectrum: Research Funding. Pasquini:Kite Pharmaceuticals: Research Funding; Novartis: Research Funding; BMS: Research Funding; Medigene: Consultancy; Amgen: Consultancy; Pfizer: Consultancy. Bhatt:Agios: Consultancy; Abbvie: Consultancy; Partner therapeutics: Consultancy; Incyte: Consultancy, Research Funding; Tolero Pharmaceuticals: Research Funding; National Marrow Donor Program: Consultancy; CSL Behring: Consultancy; Pfizer: Consultancy. Giralt:Celgene: Consultancy, Research Funding; Amgen: Consultancy, Research Funding; Takeda: Consultancy, Research Funding; Novartis: Consultancy; Actinium: Consultancy, Research Funding; Miltenyi: Research Funding; Johnson & Johnson: Consultancy, Research Funding; Jazz Pharmaceuticals: Consultancy; Kite: Consultancy; Spectrum Pharmaceuticals: Consultancy. Popat:Jazz: Consultancy; Bayer: Research Funding; Incyte: Research Funding. Weisdorf:Fate Therapeutics: Consultancy; Pharmacyclics: Consultancy; Incyte: Research Funding. Artz:Miltenyi: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal