In this issue of Blood, Chiodin et al1 show that diffuse large B-cell lymphomas (DLBCLs) of a particular subgroup frequently acquire N-glycosylation sites in their B-cell receptor (BCR) genes and carry oligomannose-type glycans that promote BCR signaling, most likely by binding to lectins in the lymphoma microenvironment.
Most human B-cell lymphomas express a BCR.2 This expression is not trivial, as many lymphomas have chromosomal translocations that destroy immunoglobulin loci. Moreover, most lymphomas derive from germinal center B (GCB) cells that have undergone somatic hypermutation, which can result in destructive mutations in IGV region genes. Thus, immunoglobulin locus–involving translocations are tolerated only when the nonexpressed immunoglobulin alleles are targeted, and destructive somatic mutations are stringently counterselected in B-cell lymphomas and their precursor cells.2 Therefore, even malignant B cells are still dependent on BCR expression.2 For several types of B-cell lymphomas, not only the presence of a BCR and the tonic BCR signaling deriving from it are essential for the lymphoma cells, but the BCRs of these lymphomas recognize autoantigens that provide sustained triggering of the BCR.3 In some instances, foreign antigens may also provide a trigger for the lymphoma (and/or its precursor cell) BCR.3
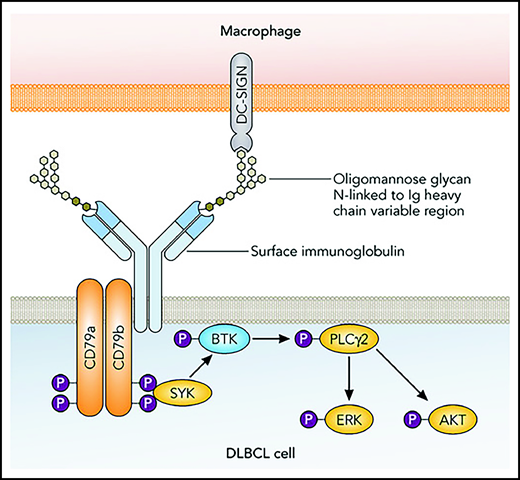
Model of lectin-mediated BCR signaling in DLBCL. Somatic mutations in the IGV region genes of DLBCL (mostly in the complementarity-determining regions) generate motifs for the addition of N-linked glycans. The BCRs carry unusual oligomannose glycans. These can be bound by DC-SIGN and perhaps additional lectins that are expressed by macrophages in the lymphoma microenvironment. Binding of DC-SIGN to the modified BCRs causes signaling that involves phosphorylation of SYK and factors farther downstream, finally leading to activation of ERK and AKT, which have prosurvival effects on the lymphoma cells. Modified from Strout et al.10 Professional illustration by Patrick Lane, ScEYEnce Studios.
Model of lectin-mediated BCR signaling in DLBCL. Somatic mutations in the IGV region genes of DLBCL (mostly in the complementarity-determining regions) generate motifs for the addition of N-linked glycans. The BCRs carry unusual oligomannose glycans. These can be bound by DC-SIGN and perhaps additional lectins that are expressed by macrophages in the lymphoma microenvironment. Binding of DC-SIGN to the modified BCRs causes signaling that involves phosphorylation of SYK and factors farther downstream, finally leading to activation of ERK and AKT, which have prosurvival effects on the lymphoma cells. Modified from Strout et al.10 Professional illustration by Patrick Lane, ScEYEnce Studios.
Nearly 20 years ago, Stevenson and colleagues recognized a unique form of BCR triggering in lymphomas: most follicular lymphomas have acquired somatic mutations in their rearranged IGV genes that create N-glycosylation sites (ie, the amino acid motif asparagine-X-serine/threonine, where X is any amino acid except proline).4 Follicular lymphomas indeed have unusual oligomannose-type glycans on these sites.5 Such glycans can bind to lectins (sugar-binding molecules). There is now evidence that the lectin DC-SIGN on macrophages and dendritic cells in the microenvironment of follicular lymphomas causes BCR signaling in the lymphoma cells, thereby contributing to lymphoma cell survival and, perhaps, proliferation.5 This phenomenon is lymphoma specific, as normal B cells very rarely have such modifications.4
It was recognized in the first description of acquired N-glycosylation sites (AGSs) in lymphomas that a fraction of DLBCLs also had IGV gene mutations creating AGSs.4 However, it was not clear whether N-glycosylation of the IGV domains was linked to particular subsets of DLBCL, and whether they had functional consequences. DLBCL is a heterogenous type of lymphoma. In an early subdivision based on gene expression patterns, DLBCLs were subdivided into activated B-cell like-DLBCL (ABC-DLBCL), GCB-DLBCL, and a third group of unclassified cases.6 More recently, DLBCLs have been grouped based on their pattern of frequently mutated genes.7-9
Chiodin et al studied the occurrence of AGSs in more than 300 cases of DLBCL. AGSs were much more frequent in GCB-DLBCL than in ABC-DLBCL (60% and 13%, respectively), and the sites were mainly located in the antigen-binding areas of the V domains, which are the complementarity-determining regions. In the genetically defined subgroups of DLBCL, cases with AGSs in the antigen-binding sites were highly enriched in the EZB-type of DLBCL, with 65% of such DLBCLs carrying the mutations. The EZB-type of DLBCL is characterized by BCL2 translocations and mutations in genes encoding epigenetic regulators.8 Importantly, further analyses showed that BCRs with AGSs in the antigen-binding sites were consistently mannosylated, and that DC-SIGN bound specifically to these surface immunoglobulins. Furthermore, exposure of primary DLBCL samples or cell lines carrying AGSs caused a moderate BCR signaling, as evidenced by the phosphorylation of SYK, extracellular signal-regulated kinase (ERK), and AKT. Thus, these modified BCRs are competent to signal after DC-SIGN binding (see figure). The sugar modification of the BCR may be of clinical relevance, because when focusing on DLBCL without a MYC translocation (which itself has a strong prognostic impact), DLBCL with AGSs in the complementarity determining regions of their BCRs showed a significantly faster clinical progression, although this finding needs validation in an independent cohort .
The findings presented by Chiodin and colleagues have important implications. The observation that the AGSs are highly enriched among EZB-type DLBCLs demonstrates a further similarity of this type of DLBCL to follicular lymphomas. These 2 types of lymphomas share many transcriptomic features and a similar pattern of mutated proto-oncogenes and tumor suppressor genes, indicating that they are closely related.9 EZB-type DLBCL may sometimes present as a transformation from an (unrecognized) follicular lymphoma. The findings presented herein that both carry AGSs further supports a close relationship and similar pathogenesis of these entities. Earlier, there was a focus on BCR signaling in ABC-DLBCL, because these lymphomas have strong constitutive BCR signaling causing NF-κB activity, termed chronic active signaling.6 Besides mutations in BCR pathway components, frequent autoreactivity of the BCR of ABC-DLBCLs most likely contributes to chronic active BCR signaling in ABC-DLBCL.6 Importantly, ABC-DLBCL depend on this activity, because inhibition of this pathway is toxic for these lymphoma cells.6 In the earlier studies on BCR dependency of DLBCL, GCB-DLBCL was felt to depend much less, if at all, on BCR signaling.6 The present study by Chiodin and colleagues modifies this picture. BCR signaling is of relevance for GCB-DLBCL (in particular, EZB-DLBCL), at least during early stages of lymphoma development, when the lymphoma cells or their precursors obtain the AGSs through somatic hypermutation. The signaling is distinct in ABC-DLBCL vs GCB-DLBCL and causes moderate AKT pathway activation in GCB-DLBCL, which is a hallmark of tonic BCR signaling. Considering the known moderate toxic effect of BCR pathway inhibition in GCB-DLBCL, it remains unclear how effective inhibition of, for example, DC-SIGN binding to the AGS of DLBCL would be. Finally, Chiodin et al have broadened the picture of human B-cell lymphomas in which the BCR is involved in lymphoma pathogenesis. Besides several lymphoma types with BCRs specific for autoantigens or foreign antigens,3 follicular lymphomas, and GCB-DLBCL represent a situation in which the BCRs are modified so that they can receive chronic stimulation by switching from stimulation through the original cognate antigen to a lectin-mediated stimulation of sugar-modified BCR (see figure).
Conflict-of-interest disclosure: The author declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal