Abstract
Modulation of neutrophil recruitment and function is crucial for targeting inflammatory cells to sites of infection to combat invading pathogens while, at the same time, limiting host tissue injury or autoimmunity. The underlying mechanisms regulating recruitment of neutrophils, 1 of the most abundant inflammatory cells, have gained increasing interest over the years. The previously described classical recruitment cascade of leukocytes has been extended to include capturing, rolling, adhesion, crawling, and transmigration, as well as a reverse-transmigration step that is crucial for balancing immune defense and control of remote organ endothelial leakage. Current developments in the field emphasize the importance of cellular interplay, tissue environmental cues, circadian rhythmicity, detection of neutrophil phenotypes, differential chemokine sensing, and contribution of distinct signaling components to receptor activation and integrin conformations. The use of therapeutics modulating neutrophil activation responses, as well as mutations causing dysfunctional neutrophil receptors and impaired signaling cascades, have been defined in translational animal models. Human correlates of such mutations result in increased susceptibility to infections or organ damage. This review focuses on current advances in the understanding of the regulation of neutrophil recruitment and functionality and translational implications of current discoveries in the field with a focus on acute inflammation and sepsis.
Introduction
A functioning immune system is crucial to combat invading pathogens. Nonetheless, uncontrolled activation and infiltration of immune cells can lead to organ damage, placing the host in danger. Clinical manifestations of or disease associated with such uncontrolled immune reactions include glomerulonephritis, vaso-occlusion–related pain crises in sickle cell disease, ischemia reperfusion damage, acute kidney injury, hepatic steatosis, and septic shock. Sepsis is associated with a high mortality and involves uncontrolled immune cell infiltration and activation, edema formation, tissue damage, and organ dysfunction.1,2 Consequently, understanding the individual regulatory steps, as well as specific recruitment and activation cues of immune cells in varying tissues and inflammatory settings, is a prerequisite to developing potential therapeutic approaches.
Leukocytes consist of different cell populations, including granulocytes (neutrophils, eosinophils, and basophils), monocytes, and T and B lymphocytes, each fulfilling distinct immunologic functions. Neutrophils, the most abundant leukocyte, reach the inflammatory site in a cascade-like manner, leading to activation of specific effector functions, such as release of reactive oxygen species, degranulation, formation of neutrophil extracellular traps (NETs), and phagocytosis.3-6 Imbalance in the number, recruitment, or functionality of neutrophils during this process results in clinical maladies presenting with uncontrolled infections, immunopathology, or autoimmunity.
Traditional steps of the leukocyte recruitment cascade: a basic understanding
Effector-based functionality requires precise targeting of neutrophils to the site of infection. This process relies on differentially regulated steps, discovered by various pioneers in the field,7-14 which can be described in a simplified cascade-like manner, resulting in successful organ infiltration.
Capturing and rolling
Neutrophils are free-flowing cells within the blood stream. Localized targeting toward the vessel wall and transient interactions manifest via rheologic- and deformability-dependent margination, followed by capturing.15,16 Capturing (or tethering) itself can be separated into 2 independent, but closely related, steps: the primary capture, implying direct neutrophil-endothelial interaction, and secondary L-selectin–mediated capturing, in which neutrophils interact with already rolling leukocytes or leukocytic fragments.17 Subsequently, the neutrophil engages in a constant slower interaction termed “rolling” (Figure 1). Although fast rolling is crucially reliant on endothelial and/or platelet P-selectin, slow rolling relies on endothelial-expressed E-selectin.18-21 Rolling involves sialyl-LewisX and the neutrophil-expressed glycosylation-dependent22,23 receptor P-selectin glycoprotein ligand 1 (PSGL-1), which, following binding to E- or P-selectin, can induce an integrin conformational change from low to intermediate or even high affinity.24,25 PSGL-1 is linked to the actin cytoskeleton, which facilitates actin polymerization changes needed for slow rolling. Indeed, defects in the polymerization of the actin cytoskeleton have been described that result in inadequate, “intermittent rolling.”26,27 When observing rolling leukocytes at very high shear rates, small membrane formations, termed “tethers” (at the rear) and “slings” (at the front), that increase the membrane adhesive area could be observed. Membrane slings feature patches of high PSGL-1 expression, together with leukocyte function antigen 1 (LFA-1) that serves as an adhesive substrate, allowing rolling even at high shear rates.28 Notably, although tethers appear to engage in more stable interactions, at very high shear rates, such as during septic conditions, they rupture from neutrophils, forming so-called S100A8/A9-rich “elongated neutrophil-derived structures.”29 These structures release S100A8/A9 into the environment, depending on calcium levels, which may actively promote the inflammatory milieu at the endothelial surface.
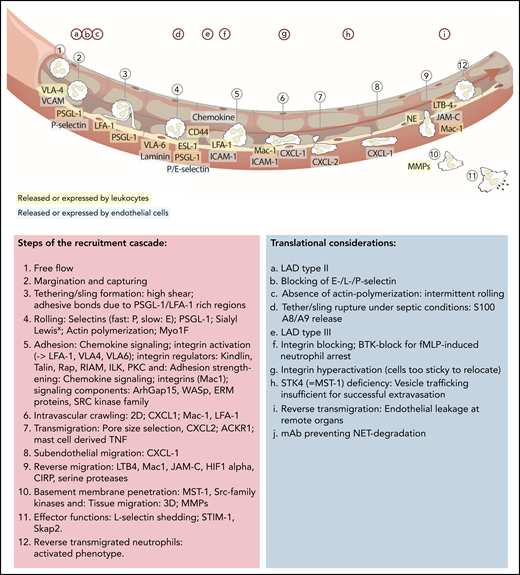
Updated leukocyte recruitment cascade and translational implications. Free-flowing neutrophils are captured by transient PSGL-1–selectin and VLA-4–VCAM/VLA-6–laminin interactions. Rolling (fast: P-selectin; slow: E-selectin) is impacted by PSGL-1 and ESL-1 binding to their selectin partners, as well as CD44, which, in a lipid raft and Bruton tyrosine kinase (BTK)-dependent manner, affects E-selectin–mediated slow rolling. Chemokine-mediated full activation of LFA-1 leads to ICAM-1–dependent firm neutrophil adhesion. This adhesive bond is further strengthened by Mac-1 interactions. Signaling elements and recruitment cues involved in integrin activation include talin-1 and kindlin-3. The subsequent slower crawling toward endothelial access points is primarily controlled by CXCL-1, whereas the transmigration toward the abluminal side is CXCL 2 dependent, followed by another step of abluminal CXCL-1–dependent crawling. Neutrophil transmigration depends on pore size selection and is additionally facilitated by mast cell–derived tumor necrosis factor (TNF). Neutrophils exert their functions within the tissue but are capable of reentering the blood circulation (reverse transmigration) in a Mac-1, neutrophil elastase, LTB-4, JAM-C–dependent pathway, taking on an activated phenotype. Different translational studies and observations have been performed to highlight implications of distinct mechanistic cues in neutrophil recruitment and functionality (upper letters). mAb, monoclonal antibody; MMPs, matrix metalloproteinase; NE, neutrophil elastase; PEU, perivascular extravasation unit; 3D, three dimensional.
Updated leukocyte recruitment cascade and translational implications. Free-flowing neutrophils are captured by transient PSGL-1–selectin and VLA-4–VCAM/VLA-6–laminin interactions. Rolling (fast: P-selectin; slow: E-selectin) is impacted by PSGL-1 and ESL-1 binding to their selectin partners, as well as CD44, which, in a lipid raft and Bruton tyrosine kinase (BTK)-dependent manner, affects E-selectin–mediated slow rolling. Chemokine-mediated full activation of LFA-1 leads to ICAM-1–dependent firm neutrophil adhesion. This adhesive bond is further strengthened by Mac-1 interactions. Signaling elements and recruitment cues involved in integrin activation include talin-1 and kindlin-3. The subsequent slower crawling toward endothelial access points is primarily controlled by CXCL-1, whereas the transmigration toward the abluminal side is CXCL 2 dependent, followed by another step of abluminal CXCL-1–dependent crawling. Neutrophil transmigration depends on pore size selection and is additionally facilitated by mast cell–derived tumor necrosis factor (TNF). Neutrophils exert their functions within the tissue but are capable of reentering the blood circulation (reverse transmigration) in a Mac-1, neutrophil elastase, LTB-4, JAM-C–dependent pathway, taking on an activated phenotype. Different translational studies and observations have been performed to highlight implications of distinct mechanistic cues in neutrophil recruitment and functionality (upper letters). mAb, monoclonal antibody; MMPs, matrix metalloproteinase; NE, neutrophil elastase; PEU, perivascular extravasation unit; 3D, three dimensional.
Integrin activation and adhesion
Rolling and deceleration of the cell lead to successful interaction of chemokine receptors, such as CXCR-2 and formyl peptide receptors, with their ligands presented on the endothelium.30 This evokes a multistep signaling response that results in the conformational change of integrins and allowing for shear-resisting cellular adhesion.14,31 Integrins are heterodimers consisting of an α subunit and a β subunit. The most prominent class of neutrophil-expressed integrins is the β2-integrins, of which they express 4 (LFA-1, Mac-1, CR4, and αDβ2) to varying degrees.32-34 To our knowledge, integrins can exist in 4 conformations, ranging from inactive (bent-closed) to fully active (open). All of the G-protein–coupled receptor (inside-out signaling)–mediated integrin activation cascades commonly result in the recruitment of integrin regulators, including talin-1 and kindlin-3, to the cytoplasmic β tail of the LFA-1 integrin, with differential involvement of other integrin modulators, such as Rap1, RIAM, ILK, and PKC (Figure 2A).35-38 This successful chemokine-mediated full activation results in the high-affinity conformation of the integrin. The high-affinity conformation of LFA-1 interacts with its endothelial ligand ICAM-1, which stops slow rolling and arrests the neutrophil on the endothelial surface.39 In addition to the β2-integrin LFA-1, the β1-integrins very late antigen 6 (VLA-6; binding to laminins) and VLA-4 (binding to VCAM-1) have been implicated in neutrophil adhesion.40-42 This adhesion is strengthened by interaction of the neutrophilic integrin Mac-1 with ICAM-1, a step that is impacted by signaling components Wiskott-Aldrich-syndrome protein (WASp) and ArhGAP15.43,44
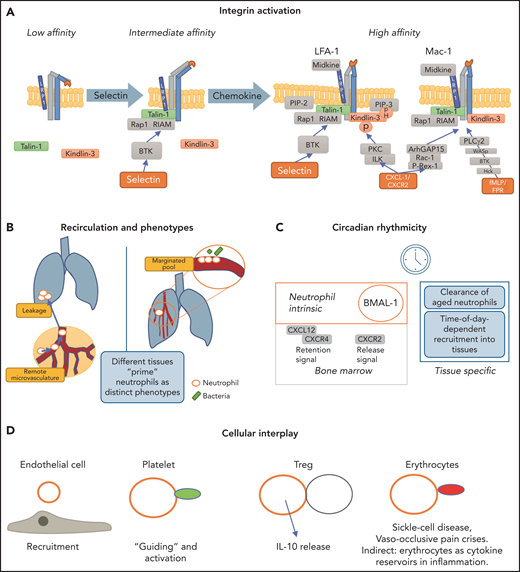
Regulators of neutrophil trafficking. (A) Integrin activation is distinctly regulated. Selectin engagement results in the intermediate conformation of the integrin, which requires talin-1 binding to the integrin cytoplasmic tail. The high-affinity conformation requires the presence of talin-1 and kindlin-3. Talin-1 binding relies on formation of a complex with Rap1 and RIAM, whereas kindlin-3 is recruited to the integrin prior to induction of the high-affinity conformation and transmits its activation signal toward LFA-1 in an ILK, PKC, and pH-domain–dependent manner. Mechanistic differences between LFA-1 and Mac-1 exist in that Mac-1 activation depends on ArhGAP15 and Bruton tyrosine kinase (BTK). LRP1 can bind via the I domain to Mac-1 and to a lesser extent to LFA-1. (B) Reverse-transmigrated neutrophils can recirculate and impact leakage even at remote organs, such as the lung. The lung-resident marginated pool of neutrophils undergoes fast Abl kinase–dependent phenotypic changes upon LPS challenge to prime the cells for bacterial “hunting.”85 Overall, neutrophils exert effects within different tissues but are equally impacted and modulated by their surroundings. (C) Circadian rhythms dictate leukocyte release, function and clearance, impacting and modulating many subsequent activation and recruitment mechanisms. (D) Neutrophils interact with endothelial cells, which mediates recruitment and activation. Interaction with platelets helps in activation, priming, and recruitment to inflamed or injured tissue but also occurs during thrombus formation. Interaction of neutrophils with regulatory T cells (Treg) evokes interleukin-10 (IL-10) release by neutrophils. Neutrophil-erythrocyte interaction is prominent in sickle cell disease. Additionally, erythrocytes contain a variety of cytokines that can impact neutrophil functionality.
Regulators of neutrophil trafficking. (A) Integrin activation is distinctly regulated. Selectin engagement results in the intermediate conformation of the integrin, which requires talin-1 binding to the integrin cytoplasmic tail. The high-affinity conformation requires the presence of talin-1 and kindlin-3. Talin-1 binding relies on formation of a complex with Rap1 and RIAM, whereas kindlin-3 is recruited to the integrin prior to induction of the high-affinity conformation and transmits its activation signal toward LFA-1 in an ILK, PKC, and pH-domain–dependent manner. Mechanistic differences between LFA-1 and Mac-1 exist in that Mac-1 activation depends on ArhGAP15 and Bruton tyrosine kinase (BTK). LRP1 can bind via the I domain to Mac-1 and to a lesser extent to LFA-1. (B) Reverse-transmigrated neutrophils can recirculate and impact leakage even at remote organs, such as the lung. The lung-resident marginated pool of neutrophils undergoes fast Abl kinase–dependent phenotypic changes upon LPS challenge to prime the cells for bacterial “hunting.”85 Overall, neutrophils exert effects within different tissues but are equally impacted and modulated by their surroundings. (C) Circadian rhythms dictate leukocyte release, function and clearance, impacting and modulating many subsequent activation and recruitment mechanisms. (D) Neutrophils interact with endothelial cells, which mediates recruitment and activation. Interaction with platelets helps in activation, priming, and recruitment to inflamed or injured tissue but also occurs during thrombus formation. Interaction of neutrophils with regulatory T cells (Treg) evokes interleukin-10 (IL-10) release by neutrophils. Neutrophil-erythrocyte interaction is prominent in sickle cell disease. Additionally, erythrocytes contain a variety of cytokines that can impact neutrophil functionality.
Crawling, transmigration, and abluminal crawling
Neutrophil adhesion is followed by slower movement along the endothelium termed “crawling,” which can be in the direction of or against the blood flow. Crawling relies on delicate activation-deactivation steps of integrins, integrin-activation pathway modulators,43 and actin flow dynamics.45 Mac-1 is the major regulator of intravascular crawling on ICAM-1 in the microvasculature.46 However, neutrophil crawling on primary mouse brain microvascular endothelial cells is regulated by LFA-1 and Mac-1 recognizing the ligands ICAM-1 and ICAM-2, hinting toward organ-specific differences in recruitment patterns.6,47 Following the active search of neutrophils for extravasation sites, diapedesis can occur in a transjunctional (paracellular) or a transcellular fashion. Paracellular diapedesis is the favored route for exit of leukocytes from the vasculature. During this process, neutrophils exert mechanical forces on vascular endothelial cells, resulting in opening of junctional gaps, followed by invasive thrusting of the neutrophil into the abluminal space.48 This process is enhanced by inflammatory signals, facilitating endothelial cell contractility, which allows leukocytes to pass.48 Interestingly, mast cell–derived tissue factor as an inflammatory signaling component also contributes to the efficient extravasation of neutrophils in different models, such as hypersensitivity-induced skin inflammation or lipopolysaccharide (LPS)-triggered inflammation.49 Overall, neutrophils seem to favor the route of least resistance in their extravasation, because they probe the surrounding surface for large pores to engage in repositioning.50 In addition to these biomechanics cues, crawling and transmigration are differentially affected by a chemokine-milieu of CXCL-1 and CXCL-2, in that CXCL-1 is the major regulator of intravascular and subluminal crawling, whereas CXCL-2 and ACKR1 mediate transmigration.51 Transmigration is followed by an abluminal crawling step, which can be regarded as another cascade-like series of events ultimately resulting in full tissue invasion, once the basement membrane and pericyte layer are breached.52
Reverse migration
Notably, the previously described 1-way route of extravasation is also accessible in an abluminal-to-luminal direction, because neutrophils are capable of re-entering the bloodstream in a process termed “reverse transendothelial migration” (rTEM).53 This process is driven by LTB4, neutrophil elastase, and Mac-1, which together lead to the proteolytic cleavage of JAM-C, allowing neutrophils to re-enter the bloodstream.54,55 Further regulatory factors involve activation of the hypoxic factor 1α subunit as a negative regulator and cold-inducible RNA-binding protein as a positive regulator of rTEM of neutrophils.56,57 The reverse migratory step is crucially dependent on chemokine gradients, because deficiency of the chemokine receptor CXCR2 reduces neutrophil rTEM.58 Overall, rTEM results in neutrophil phenotype switching toward an activated morphology.59 Reverse transmigrated neutrophils may lodge in remote organs, such as the lung, leading to vascular leaks at sites distant from the original inflammatory injury (Figure 2B).60 Notably, in zebrafish, reverse transmigration also occurs during wound healing, which is at least partially dependent on the presence of macrophages that mediate the resolution of neutrophil infiltration.61,62 Neutrophil reverse migration has been regarded as a cornerstone of resolution of inflammation in wound healing.63 Therefore, these findings could imply that lodging of preactivated neutrophils in remote organs is the unwanted consequence of ongoing/uncontrolled resolution processes, including reverse migration, at sites of inflammation.62 Because neutrophils need to access the circulatory system to return to the circulation, tissue-disrupting factors must be considered. Indeed, in a model of sterile hepatic wounding in the mouse, although neutrophil primary extravasation was not impacted by the absence of cathepsin C, neutrophil reverse transmigration crucially depended on serine protease activity, affecting organ revascularization.64 Notably, reverse transmigration also occurs in other leukocyte subsets, such as monocytes, and has been linked to phenotypic differences in mature dendritic cells (reverse migrated) compared with macrophages (remaining and not engaging in rTEM).65
Tissue migration
Once the neutrophil has breached the basement membrane barrier to enter the interstitium, it engages in a different “amoeboid” form of migration that requires cellular deformation, actin polymerization, and structural tissue reorganization.66 The mechanisms regulating amoeboid interstitial migration are distinct from those controlling intravascular adhesion/migration. For example, deficiency in the unconventional class I myosin 1f does not affect migration along 2-dimensional surfaces, such as endothelial crawling, but it does block successful migration of neutrophils in 3-dimensional collagen networks.67 During interstitial migration, neutrophils release proteases to degrade matrix elements while also repositioning their nucleus, propelling themselves forward through distinct collagen network pores.68 In contrast to 2-dimensional intraluminal crawling in which serine proteinases control cell surface adhesion molecules (eg, CD44), matrix metalloproteinases play the dominant role in tissue remodeling during interstitial migration.66 Nonetheless, the migration of neutrophils in interstitial tissue can also occur in a protease-independent fashion via persistent rearward polarization of F-actin.69
Effector functions
Once neutrophils are recruited to the site of infection, combatting of invading pathogens is required. For this purpose, the neutrophil is equipped with a repertoire of effector functions, such as phagocytosis, NETosis, reactive oxygen species production, and granule secretion. These effector functions can be induced by transmigration of neutrophils, in which they take on a preactivated phenotype.70,71 Regulators of integrin signaling (eg, Src kinase-associated phosphoprotein 2) that modulate inside-out integrin activation equally affect outside-in signaling responses (eg, phagocytosis of bacterial particles), emphasizing the dual contribution of integrins and integrin regulators to recruitment and effector functions.72 As another signaling element, calcium shuttling plays a major role in efficient phagocytosis, because absence of the calcium sensor STIM1 significantly decreases the phagocytosis of Staphylococcus aureus particles.73 A rather surprising observation is the effect of L-selectin shedding, which occurs regularly during neutrophil activation, on outside-in–mediated effector functions. Herein, ADAMTS17 based L-selectin shedding enhances phagocytosis, bacterial clearance, and tissue navigation of neutrophils in infected tissue.74
In addition to this direct “eating” response toward bacterial particles, another effector component, NETs, is needed to combat and trap invading pathogens, as demonstrated by the use of a novel monoclonal antibody that protects NETs from degradation and which enhances outcome in preclinical sepsis models.75 Also, sterile inflammatory diseases, such as rheumatoid arthritis, liver injury, and kidney damage, are impacted by NETs, because excessive NETosis can result in organ damage.76-78
Overarching factors influencing neutrophil functionality and inflammation
Organ-specific properties and neutrophil pools
Organ-specific recruitment patterns of neutrophils have been uncovered over the years.79-82 Different organs exhibit distinct rheologic, cellular, and receptor properties. For example, the lung and liver feature the recently identified receptor dipeptidase-1, which impacts leukocyte sequestration and the outcome of endotoxemia-related pulmonary inflammation.83 Interestingly, the kidney also features distinct recruitment patterns, because the recruitment of neutrophils to the glomerulus does not involve prior rolling of neutrophils but immediate arrest due to platelet–P-selectin.84 It is tempting to speculate about possible therapeutics that are capable of blocking the organ-specific receptors responsible for neutrophil infiltration, which would allow for directed targeting of these inflammatory cells to a site of infection, away from, for example, ischemia-reperfusion–injured organs. Major sites of neutrophil interaction, such as the lung, are additionally known to feature neutrophil “pools” and capillary-based recruitment patterns (Figure 2B).85 Thus, although neutrophil infiltration is known to affect organ function, the cell itself is also modulated by its surrounding tissue. Corroborating this notion, Ballesteros et al elegantly demonstrated very recently that neutrophil phenotypes, including longevity, differed according to their target tissues.86
Neutrophil heterogeneity and circadian rhythmicity
Originally described as a homogenous population, it is now well accepted that different neutrophil subtypes exist, underscoring the importance of neutrophil heterogeneity for disease-related outcomes.87 Although the discussion about the origin of these distinct phenotypic features is ongoing,88 it seems that intrinsic and extrinsic factors contribute to neutrophil diversity. One prominent internal factor is the clock gene Bmal1 (Figure 2C). This factor acts as an internal clock, affecting CXCL2/CXCR2- and contrasting CXCR4-dependent processes associated with neutrophil aging and clearance from the circulation.89 Additionally, external circadian features and tissue-dependent properties affect neutrophils, because clearance from the circulation is impacted by oscillations of interacting molecules, such as P-/E-selectin and VCAM-1, in the bone marrow microvasculature.90,91 Although circadian rhythms, in general, have long been known,92,93 and clinical observations highlight the time-of-day variability in inflammatory-disease presentations,94-96 therapeutic and clinical approaches (eg, scheduling of surgeries/interventions) incorporating this knowledge remain scarce.97
Cellular interplay affecting neutrophils
The interaction of neutrophils with endothelial cells is crucial for activation and recruitment, but other cells also influence neutrophil responses. Neutrophils are capable of directly interacting with platelets, megakaryocytes, other leukocytes, tumor cells, and erythrocytes to various degrees, depending on the underlying (disease) model (Figure 2D).98-101 Although direct erythrocyte-neutrophil interplay occurs primarily in sickle cell disease, platelets are ascribed a more prominent role in the interaction with neutrophils, “guiding” them to extravasation sites.102 Depletion of platelets protects mice from lung injury in a number of models.103,104 Nonetheless, thrombocytopenia itself is linked to an increased risk for acute respiratory distress syndrome.105 Also, in the liver, platelets assist in the recruitment of neutrophils to sterile injury sites, a process that is crucial for organ repair.106 In addition to platelets, neutrophils interact directly with T cells, specifically regulatory T cells, which, once stimulated with LPS, provoke the production of interleukin-10 in neutrophils.107
Pericytes in the regulation of neutrophil invasion and inflammation
Neutrophil emigration is impacted by endothelial cells, and pericytes are also key contributors to efficient neutrophil tissue invasion.108 It has been known for more than a decade that low-expression regions/hotspots within the basement membrane are preferred sites for neutrophil emigration.109 These low-expression regions are aligned with pericyte gaps and both can enlarge to a similar extent, controlling neutrophil invasion.109,110 Neutrophils interact with pericytes in an ICAM-1–dependent manner by the formation of extensions that lead to interactions with pericyte-based extensions,111 but conflicting reports exist that question the necessity of neutrophil-interaction for gap enlargement.110,112 The contractility of pericytes, which has been attributed to the regulation of neutrophil invasion permissiveness, is impacted by endothelial cell–secreted macrophage inhibitory factor, showing the mechanistic interplay of the blood-tissue barrier during neutrophil emigration.113 Distinct subsets of pericytes, such as NG2+ pericytes, which can be found in proximity to arterioles and capillaries, show unique expression characteristics, offering inflammatory sensors, such as tumor necrosis factor receptor, Toll-like receptors, and formyl peptide receptor 2. These pericytes lead to increased migratory velocity and altered invasive characteristics (including regulation of matrix metalloproteinases) of neutrophils by macrophage inhibitory factor and CXCL8.114 Although pericytes are well recognized for their contribution to the regulation of neutrophil invasion, therapeutic endeavors remain scarce and demand further mechanistic research.
Translational considerations
For clinicians, understanding the underlying mechanisms of neutrophil recruitment is highly important (Figures 1, 3, and 4). As described above, inflammation impacts neutrophil activation responses,115 and the course of inflammatory conditions relies on neutrophil functionality and neutrophil activation.116-118 Therapeutic and diagnostic approaches are underway to specifically target neutrophils in inflammatory diseases.
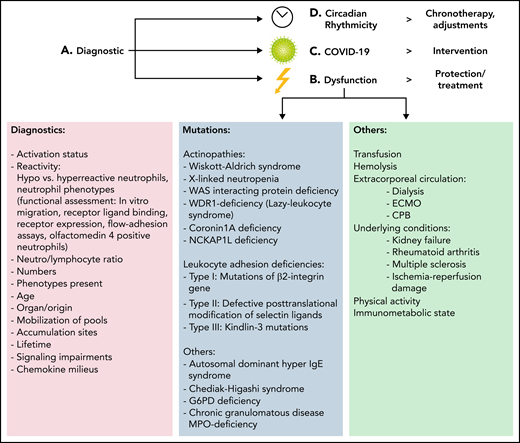
Translational considerations. (A) An adequate assessment of neutrophils present, the degree and necessity of inflammatory reactions, and evaluation of possible therapeutics must be performed. (B) Leukocyte dysfunction is observable in genetic diseases. Leukocyte adhesion deficiencies result in an increased risk for infections due to different underlying causes. Also, other causes affecting neutrophils are known, including transfusion, extracorporeal circulation, and inflammatory diseases. Patients require isolation and immunoprotective measures or adequate control of neutrophil hyperactivation and organ-protection strategies. (C) COVID-19–based considerations must take neutrophil phenotypes into account. Anti-inflammatory strategies are being discussed, but more precise interventions are needed. Distinguishing between the acute and long phases of COVID are important. Targeted approaches seem to be promising for future endeavors in the therapy of COVID, sepsis, or acute respiratory distress syndrome. (D) Consideration of circadian rhythmicity is crucial for future therapeutic endeavors. Rescheduling of surgeries or interventions is needed, and chronotherapy, light therapy, and sleep interventions are in place. CPB, cardiopulmonary bypass; ECMO, extracorporeal membrane oxygenation; IgE, immunoglobulin E; WAS, Wiskott-Aldrich syndrome.
Translational considerations. (A) An adequate assessment of neutrophils present, the degree and necessity of inflammatory reactions, and evaluation of possible therapeutics must be performed. (B) Leukocyte dysfunction is observable in genetic diseases. Leukocyte adhesion deficiencies result in an increased risk for infections due to different underlying causes. Also, other causes affecting neutrophils are known, including transfusion, extracorporeal circulation, and inflammatory diseases. Patients require isolation and immunoprotective measures or adequate control of neutrophil hyperactivation and organ-protection strategies. (C) COVID-19–based considerations must take neutrophil phenotypes into account. Anti-inflammatory strategies are being discussed, but more precise interventions are needed. Distinguishing between the acute and long phases of COVID are important. Targeted approaches seem to be promising for future endeavors in the therapy of COVID, sepsis, or acute respiratory distress syndrome. (D) Consideration of circadian rhythmicity is crucial for future therapeutic endeavors. Rescheduling of surgeries or interventions is needed, and chronotherapy, light therapy, and sleep interventions are in place. CPB, cardiopulmonary bypass; ECMO, extracorporeal membrane oxygenation; IgE, immunoglobulin E; WAS, Wiskott-Aldrich syndrome.
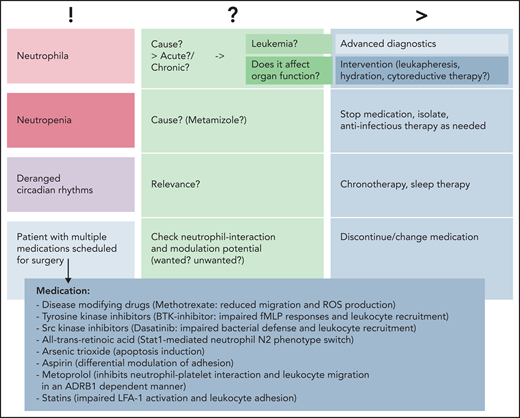
Common clinical findings. Common findings can include changes in neutrophil numbers (neutrophilia vs neutropenia), deranged circadian rhythms, or patients with various medications scheduled for surgery or other medical interventions. Causality is to be assessed, and advanced diagnostics or interventions should be planned in a timely manner prior to/in parallel with subsequent treatments. BTK, Bruton tyrosine kinase; ROS, reactive oxygen species; !, current situation; ?, possible cause/s; >, consequences.
Common clinical findings. Common findings can include changes in neutrophil numbers (neutrophilia vs neutropenia), deranged circadian rhythms, or patients with various medications scheduled for surgery or other medical interventions. Causality is to be assessed, and advanced diagnostics or interventions should be planned in a timely manner prior to/in parallel with subsequent treatments. BTK, Bruton tyrosine kinase; ROS, reactive oxygen species; !, current situation; ?, possible cause/s; >, consequences.
Targeting the classical recruitment cascade
Selectins
Selectins are prominently involved in the primary steps of neutrophil recruitment. From a translational point of view, although preclinical (swine)119 and clinical data from a pilot study suggested favorable application of an E-selectin antibody in patients with septic shock,120 primate data using E- and L-selectin antibodies did not reveal any benefit in lung injury or mortality in a sepsis model.121 Because involvement of P-selectin in neutrophil-dependent lung injury has long been known,122 applicability of P-selectin blockage in patients with COVID-19 is being discussed.123 Nonetheless, the necessity of selectins and selectin ligands for efficient immune defense is seen in patients presenting with leukocyte adhesion deficiency type 2; they show impaired leukocyte rolling due to a fucosylation defect of selectin ligands.124
Integrin activation and neutrophil adhesion
Studies using genetically engineered mice resulting in integrin hyper- or hypoactivation, as well as utilization of blocking antibodies in disease models, demonstrate the involvement of integrin functionality in acute inflammatory disease models, such as pneumonia, sepsis, and acute kidney injury.125-129 Overall, blocking or genetic targeting of integrins seems to be promising in animal studies, but it fails to show outcome-relevant effects in humans or results in major adverse events. Studies targeting the Mac-1 and LFA-1 ligand ICAM-1 using an antibody or antisense oligonucleotide yielded negative results in ischemic stroke and steroid-refractory Crohn’s disease.130,131 Also, when directly targeting CD18-containing integrins, the results for myocardial infarction were negative,132 whereas preclinical data for polymicrobial sepsis showed reduced lung damage following antibody blocking of Mac-1 and LFA-1.126 Nonetheless, causality of therapeutics is difficult to determine because of the involvement of various cell populations and the inability to distinguish between the different recruitment steps in which integrins are involved.133 Notably, various adverse events have been reported in leukocyte-related studies (eg, the ICAM-1–blocking antibody enlimomab resulted in an increased rate of infections) (Table 1).130
Exemplary clinical trials in inflammatory, autoimmune, and inflammation-related diseases
| Study focus . | Disease . | Substance/intervention . | Molecular/cellular target . | Cell target . | Success/failure . | Study type . | Reference (year of publication) . |
|---|---|---|---|---|---|---|---|
| BTK inhibition in patients with severe COVID-19 | Severe COVID-19 | Acalabrutinib | BTK inhibition | B cells, platelets, monocytes, macrophages, neutrophils, dendritic cells | Positive (improvement of oxygenation); international confirmatory study ongoing | Single-center probatory trial | 157 (2020) |
| BTK | Rheumatoid arthritis | Spebrutinib (CC-292) | BTK inhibition | B cells, platelets, monocytes, macrophages, neutrophils, dendritic cells | Mixed (did not meet primary end points; but subgroup analysis might hold potential; downward trend in symptoms; furthermore, modulation of B-cell populations) | Multicenter phase 2a trial | 158 (2020) |
| JAK inhibition in skin inflammation | Atopic dermatitis | Tofacitinib | JAK inhibition | Ubiquitous | Positive (greater efficacy and early onset of effect) | Phase 2a trial | 159 (2016) |
| JAK inhibition in RA | RA | Filgotinib | JAK1 inhibition | Ubiquitous | Positive (regulation of biomarkers and correlation with disease activity) | Phase 2b trial | 160 (2020) |
| MS | MS | Evobrutinib | BTK inhibition | B cells, platelets, monocytes, macrophages, neutrophils, dendritic cells | Mixed positive (fewer enhancing lesions during weeks 12 to 24 compared with placebo but no difference in relapse rate or disability progression) | Multicenter phase 2 trial | 161 (2019) |
| MS | Highly active relapsing-remitting MS | Natalizumab | VLA-4 | HSPCs, T cells, B cells, monocytes, NK cells, eosinophils, neutrophils | Mixed: first failure (progressive multifocal leukoencephalopathy) and then reallowance | 162, 163 | |
| MS | Relapsing remitting multiple sclerosis | Firategrast | VLA-4 | HSPCs, T cells, B cells, monocytes, NK cells, eosinophils, neutrophils | Positive for phase 2 trial (imaging end points improved); awaiting phase 3 | Phase 2 | 164 (2012) |
| Enlimomab Acute Stroke Trial | Ischemic stroke | Enlimomab | ICAM-1 antibody (murine into human) | Endothelial cells, epithelial cells, vascular smooth muscle cells, certain leukocyte subsets | Negative (worse Modified Rankin Scale score in treated patients; more adverse events including infections) | 130 (2001) | |
| Crohn’s disease | Steroid-refractory Crohn’s disease | ICAM-1 antisense oligonucleotide (ISIS-2302) | Adhesion/outside-in signaling | Endothelial cells, epithelial cells, vascular smooth muscle cells, certain leukocyte subsets | Negative for primary end point: steroid-free remission (but treatment led to increased rate of glucocorticoid dose 0 mg prednisone equivalent) | 131 (2001) | |
| HALT-MI study | Myocardial infarction with direct primary angioplasty | Rovelizumab (= LeukArrest) | CD11/CD18 (humanized Ab) | Leukocytes | Negative (no infarct size reduction) | Multicenter placebo-controlled double blinded RCT | 132 (2002) |
| Meta-analysis for moderate to severe psoriasis | Psoriasis | Alefacept, efalizumab, etanercept, infliximab | LFA-1 (efalizumab), TNF-α (infliximab, etanercept), | Leukocytes (efalizumab); systemic (TNF) | Positive (infliximab > etanercept > efalizumab > alefacept). But increased SAEs in efalizumab trials. | Systematic review | 165 (2005) |
| Dry eye disease | Dry eye disease | Lifitegrast (LFA-1 antibody) | Adhesion | Leukocytes | Positive, but increased adverse events. | Multicenter RCT | 166 (2016) |
| Pneumonia | Pneumonia | Simvastatin | NETosis, chemotaxis | Positive (improved neutrophil function and improved SOFA scores compared with placebo) | RCT | 167 (2019) | |
| BMS-936559 study | Sepsis | Antiprogrammed cell death-ligand 1 antibody (hinders binding of PD-L1 to PD-1 and CD80) | Safe increased monocyte human leukocyte antigen-DR expression, but future randomized trials are needed | Placebo-controlled dose-escalation RCT | 168 (2019) | ||
| Sepsis | Severe sepsis | Polymyxin B hemoperfusion | Endotoxin elimination | Systemic | Negative for sepsis outcomes, but improved mHLA-DR expression in severe sepsis | RCT | 169, 170 (2018) |
| Crohn’s disease | Ulcerative colitis, Crohn’s disease | Madcam-1 receptor (α4β7 integrin) antibody | α4β7 inhibitor (Madcam receptor) | NK cells, eosinophils, T cells, B cells, and, to some extent, endothelial cells | Approved (vedolizumab); discontinued (abrilumab) | 154 | |
| RA | RA | TNF-α blocker | Systemic | Positive, but almost similar effectiveness to methotrexate | Meta-analysis | 171 (2012) | |
| Septic shock | Septic shock | TNF blocker (mAb) | Systemic | Negative. (No overall difference in mortality. Significant difference at day 3 reduction in mortality but no significant difference at day 28) | Multicenter RCT | 172 (1995) | |
| Sepsis | Sepsis | TNF blocker | Systemic | Positive (decreased risk of death) but emphasizes only modest effects with past results possibly negative because of underpowering | Meta-analysis | 173 (2014) |
| Study focus . | Disease . | Substance/intervention . | Molecular/cellular target . | Cell target . | Success/failure . | Study type . | Reference (year of publication) . |
|---|---|---|---|---|---|---|---|
| BTK inhibition in patients with severe COVID-19 | Severe COVID-19 | Acalabrutinib | BTK inhibition | B cells, platelets, monocytes, macrophages, neutrophils, dendritic cells | Positive (improvement of oxygenation); international confirmatory study ongoing | Single-center probatory trial | 157 (2020) |
| BTK | Rheumatoid arthritis | Spebrutinib (CC-292) | BTK inhibition | B cells, platelets, monocytes, macrophages, neutrophils, dendritic cells | Mixed (did not meet primary end points; but subgroup analysis might hold potential; downward trend in symptoms; furthermore, modulation of B-cell populations) | Multicenter phase 2a trial | 158 (2020) |
| JAK inhibition in skin inflammation | Atopic dermatitis | Tofacitinib | JAK inhibition | Ubiquitous | Positive (greater efficacy and early onset of effect) | Phase 2a trial | 159 (2016) |
| JAK inhibition in RA | RA | Filgotinib | JAK1 inhibition | Ubiquitous | Positive (regulation of biomarkers and correlation with disease activity) | Phase 2b trial | 160 (2020) |
| MS | MS | Evobrutinib | BTK inhibition | B cells, platelets, monocytes, macrophages, neutrophils, dendritic cells | Mixed positive (fewer enhancing lesions during weeks 12 to 24 compared with placebo but no difference in relapse rate or disability progression) | Multicenter phase 2 trial | 161 (2019) |
| MS | Highly active relapsing-remitting MS | Natalizumab | VLA-4 | HSPCs, T cells, B cells, monocytes, NK cells, eosinophils, neutrophils | Mixed: first failure (progressive multifocal leukoencephalopathy) and then reallowance | 162, 163 | |
| MS | Relapsing remitting multiple sclerosis | Firategrast | VLA-4 | HSPCs, T cells, B cells, monocytes, NK cells, eosinophils, neutrophils | Positive for phase 2 trial (imaging end points improved); awaiting phase 3 | Phase 2 | 164 (2012) |
| Enlimomab Acute Stroke Trial | Ischemic stroke | Enlimomab | ICAM-1 antibody (murine into human) | Endothelial cells, epithelial cells, vascular smooth muscle cells, certain leukocyte subsets | Negative (worse Modified Rankin Scale score in treated patients; more adverse events including infections) | 130 (2001) | |
| Crohn’s disease | Steroid-refractory Crohn’s disease | ICAM-1 antisense oligonucleotide (ISIS-2302) | Adhesion/outside-in signaling | Endothelial cells, epithelial cells, vascular smooth muscle cells, certain leukocyte subsets | Negative for primary end point: steroid-free remission (but treatment led to increased rate of glucocorticoid dose 0 mg prednisone equivalent) | 131 (2001) | |
| HALT-MI study | Myocardial infarction with direct primary angioplasty | Rovelizumab (= LeukArrest) | CD11/CD18 (humanized Ab) | Leukocytes | Negative (no infarct size reduction) | Multicenter placebo-controlled double blinded RCT | 132 (2002) |
| Meta-analysis for moderate to severe psoriasis | Psoriasis | Alefacept, efalizumab, etanercept, infliximab | LFA-1 (efalizumab), TNF-α (infliximab, etanercept), | Leukocytes (efalizumab); systemic (TNF) | Positive (infliximab > etanercept > efalizumab > alefacept). But increased SAEs in efalizumab trials. | Systematic review | 165 (2005) |
| Dry eye disease | Dry eye disease | Lifitegrast (LFA-1 antibody) | Adhesion | Leukocytes | Positive, but increased adverse events. | Multicenter RCT | 166 (2016) |
| Pneumonia | Pneumonia | Simvastatin | NETosis, chemotaxis | Positive (improved neutrophil function and improved SOFA scores compared with placebo) | RCT | 167 (2019) | |
| BMS-936559 study | Sepsis | Antiprogrammed cell death-ligand 1 antibody (hinders binding of PD-L1 to PD-1 and CD80) | Safe increased monocyte human leukocyte antigen-DR expression, but future randomized trials are needed | Placebo-controlled dose-escalation RCT | 168 (2019) | ||
| Sepsis | Severe sepsis | Polymyxin B hemoperfusion | Endotoxin elimination | Systemic | Negative for sepsis outcomes, but improved mHLA-DR expression in severe sepsis | RCT | 169, 170 (2018) |
| Crohn’s disease | Ulcerative colitis, Crohn’s disease | Madcam-1 receptor (α4β7 integrin) antibody | α4β7 inhibitor (Madcam receptor) | NK cells, eosinophils, T cells, B cells, and, to some extent, endothelial cells | Approved (vedolizumab); discontinued (abrilumab) | 154 | |
| RA | RA | TNF-α blocker | Systemic | Positive, but almost similar effectiveness to methotrexate | Meta-analysis | 171 (2012) | |
| Septic shock | Septic shock | TNF blocker (mAb) | Systemic | Negative. (No overall difference in mortality. Significant difference at day 3 reduction in mortality but no significant difference at day 28) | Multicenter RCT | 172 (1995) | |
| Sepsis | Sepsis | TNF blocker | Systemic | Positive (decreased risk of death) but emphasizes only modest effects with past results possibly negative because of underpowering | Meta-analysis | 173 (2014) |
Studies include data in which the target cell is unclear; inhibitors or blocking agents do not only target neutrophils.
Ab, antibody; BTK, Bruton tyrosine kinase; HSPCs, hematopoietic stem and progenitor cells; mAb, monoclonal antibody; mHLA-DR, monocyte HLA-DR; MS, multiple sclerosis; NK, natural killer; RA, rheumatoid arthritis; RCT, randomized controlled trial; SAE, serious adverse event; SOFA, Sequential Organ Failure Assessment; TNF, tumor necrosis factor.
Crawling and migration
Blocking of leukocyte integrins LFA-1/Mac-1 or ICAM-1 reduces transendothelial migration similar to the blocking of CXCL-1 or CXCL-2 that results in diminished neutrophil influx into the abdominal cavity at early time points in an LPS model.134,135 Also, human data using a CXCR2 antagonist seems to be promising because it reduces neutrophil influx into airways following LPS challenge in healthy volunteers and is under debate for treatment of COVID-19.136,137 With regard to neutrophils undergoing rTEM, these cells impact vascular leakage at sites distant from the original inflammatory injury, but clinical targeting of the specific cells is not yet possible.60
Effector functions
Priming of neutrophils for anti-infectious purposes, while at the same time limiting host tissue damage, holds great potential for a future in which increasing microbial antibiotic resistance must be considered. Because the integrin Mac-1 is needed for anti-infectious purposes, as well as the prevention of immunopathology, a novel Mac-1 antibody that specifically targets the interaction of CD40L with Mac-1 was developed. Animals treated with this antibody show reduced neutrophil recruitment, thus limiting organ damage but preserving antimicrobial functions.138
Inborn neutrophil defects
Consequences of neutrophil dysfunction can be observed in patients with specific mutations. Genetic mutations affecting neutrophils were summarized recently.139 Prominent examples are leukocyte adhesion deficiencies and actinopathies, such as Wiskott-Aldrich syndrome or coronin-1A deficiency, which result in impressive phenotypes when it comes to neutrophil morphodynamics.140 Leukocyte adhesion deficiencies are rare mutations affecting β2-integrin functionality via mutations within the integrin gene itself (type 1), defects in selectin ligand posttranslational modification (type 2), or mutations within the kindlin-3 gene (type 3).141 These mutations lead to recurrent infections due to leukocyte recruitment defects.142-144 Another mutation affecting neutrophil functions is gain-of-function mutations of WASp, which lead to X-linked neutropenia. This disease is associated with neutropenia, and neutrophils show a hyperactivated phenotype with WASp activity uncoupled from its regulation by phosphatidylinositol 3-kinase.145
Consideration of circadian rhythmicity
As stated above, circadian rhythms are fundamental regulators of neutrophil receptor expression, numbers, and tissue homing. Although these findings are of great interest for the scientific community, neutrophil-related translational endeavors are scarce. Rescheduling of surgeries or therapeutic interventions based on considerations of wanted/unwanted inflammatory consequences cannot be observed in regular hospital care, although awareness for increased cardiovascular disease risk is being raised in shift workers, for example.146 Current endeavors in circadian rhythmicity–related therapies focus on chronotherapy, light therapy, and sleep interventions.97 Of note, neutrophil inflammatory responses are affected by circadian rhythms, as well as an inflammatory trigger itself that leads to reorganization of circadian rhythms in preclinical animal models.147
COVID-19
In the past year, clinicians worldwide have been confronted with patients infected by SARS-CoV2. Notably, COVID-19 is associated with massive immunothrombotic dysfunctions. Nicolai et al showed that phenotypic changes in neutrophils correlate with disease severity.148 Additionally, immature band neutrophils can be found in the blood of critically ill patients with COVID-19.149 Although the observed neutrophil characteristics were rather specific for severe COVID-19 cases, it is possible that multipanel assessment of neutrophils might aid in diagnosing and selecting patients for appropriate treatments in other disease settings as well.150 In this direction, screening for deformability of neutrophils in COVID-19 and long-COVID-19 patients revealed disturbed biomechanics properties with diagnostic implications.151 Different anti-inflammatory strategies have been proposed by researchers and clinicians, such as studying the effects of tumor necrosis factor blockade152 or blocking neutrophil NET formation in the lung.153 Not all anti-inflammatory approaches have worked, possibly also as a result of unspecificity (Table 1).
Outlook
Current research endeavors focus on the discovery of novel components in signaling, phenotypic switching, and recruitment of neutrophils with the use of multi-omics and next-generation sequencing. Furthermore, available data on clinical trials highlight that a vast number of studies concentrate on better phenotyping of patients, neutrophil subsets, and disease subclasses (eg, sepsis). For the keyword “neutrophil,” clinicaltrials.gov lists 2618 completed studies and 800 recruiting studies (last updated on 20 June 2021). The underlying topics are diverse, ranging from NET formation in systemic sclerosis (NCT03374618) to neutrophil phenotypic profiling in various patients, such as those with diabetes (NCT02080741), patients postsurgery (NCT03058328), and patients experiencing cardiovascular events (NCT04581057) or sepsis (NCT04103268, NCT02803346). Inhibitor studies include research on neutrophil elastase inhibition in bronchiolitis obliterans syndrome (NCT02669251), as well as a granulocyte-macrophage colony-stimulating factor inhibitor (otilimab) in patients with arthritis (NCT04134728). Additionally, routinely available interventions that include diet or physical activity are being assessed for their implications in inflammation and neutrophil recruitment. Novel applications for in-use medications (eg, methotrexate) are being studied, and new drugs are being put to the test. In this sense, development of more specific integrin modulators that avoid exerting unintended agonistic signaling are on the way.154 A tremendous boost in translational inflammatory studies has occurred as a result of the COVID-19 pandemic. These findings are a big step toward the next level of neutrophil research in which phenotyping will lead to a personalized targeted therapy, also taking into account circadian (dys)regulations and allowing the precise directing of each inflammatory cell to the infected site away from uninvolved tissues.155,156 Additionally, targeted on-off switching of effector functions will help in preventing immunopathology to improve organ function.
Summary and conclusions
In summary, the recruitment of neutrophils relies on a distinct multifactorial sequence of events that requires selectins, integrins, accessible ligands, appropriate chemokine milieus, cell-cell interactions, and different signaling cascades in the cells. Current evidence shows that the previously reported 1-way street of leukocyte recruitment is, in fact, a 2-way road by which neutrophils can reenter the circulation and cause organ damage, even at a remote location in the body. The regulation of each step for the successful infiltration of neutrophils is crucially impacted by integrin interactors and chemokine gradients. Recruitment, numbers, and phenotypes of neutrophils are affected by circulating chemokines and cytokines, and consumption, reproduction, and mobilization of neutrophil pools also must be considered when interpreting the full magnitude of circulating neutrophil populations. Thus, it appears that we are still on the brink of understanding the complex interplay of inflammation, stressors, resolution, and the variety of functions that neutrophils exert. Thus, future research is needed to optimize diagnostic options for neutrophil subpopulations, and large randomized controlled trials are crucially needed to address immunomodulatory therapeutics that are individualized for specific patient subsets.
Acknowledgment
This work was supported by the Deutsche Forschungsgemeinschaft (KFO342, ZA428/18-1, ZA428/14-1, ZA 428/12-1, INST 211/984-1, INST 211/604-3 [to A.Z.]; MA 9604/1-1 [to A.M.]) and the Interdisziplinäres Zentrum für Klinische Forschung Münster (SEED 12/18 [to A.M.] and Za2/001/18 [to A.Z.]).
Authorship
Contribution: A.M. wrote and designed the outline of the manuscript; C.A.L. wrote the manuscript; and A.Z. supervised the study and wrote and designed the outline of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Alexander Zarbock, Department of Anesthesiology, Intensive Care Medicine and Pain Therapy, University of Münster, Albert-Schweitzer Campus 1, Building A1, Münster 48149, Germany; e-mail: zarbock@uni-muenster.de.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal