In this issue of Blood, Heeney et al1 report negative findings from HESTIA3, the phase 3 placebo-controlled ticagrelor trial for the reduction of the annual incidence of vaso-occlusive crises in children with sickle cell disease (SCD).
Publishing this study’s negative results is commendable. The work highlights what sometimes seem like competing SCD concerns. Therapeutic advances and mechanistic studies are needed to reduce end-organ damage, extend life expectancy, and improve quality of life for people with SCD. At the same time, established preventive and disease-modifying interventions do not yet reach all people with SCD. Multinational SCD treatment trials like HESTIA3 are tethered to these realities.2
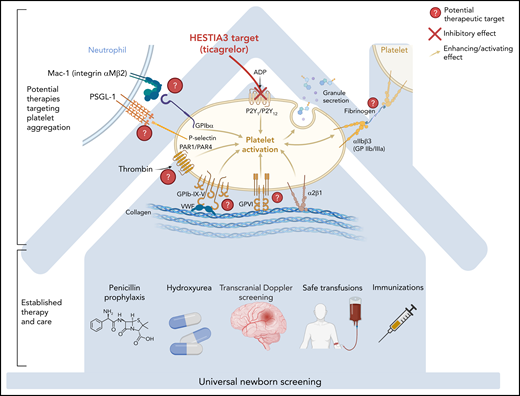
Two negative studies now show that P2Y12 inhibitors are insufficient to prevent vaso-occlusive crisis. However, they do not altogether extinguish the possibility that inhibiting platelet function could help treat SCD. HESTIA3 relied on preclinical data that showed P2Y12 inhibition decreased platelet activation and aggregation.3 Unfortunately, this promising evidence did not deliver measurable clinical benefit. Redundancies in human physiology may enable vaso-occlusive pathways to circumvent inhibition of a single cellular target. Possibly, other mechanisms of cellular aggregation will be successfully targeted alone or in combination and some have robust preclinical models.4 For example, neutrophil-platelet interactions play a prominent role in vaso-occlusion and depend partly on cellular activation by P-selectin and platelet glycoprotein 1b-alpha (GPIbα) (see figure). In preclinical models, inhibiting P-selectin and GPIbα more completely attenuated platelet-neutrophil aggregation than inhibiting either protein alone. This result provides a rationale for a study of antiplatelet therapy and crizanlizumab. As many recognize, future advances in SCD treatment may well involve rigorous study of multiagent therapy.5
The identification of novel therapeutic targets, like platelet activation shown here, builds upon established interventions to diagnose sickle cell disease and prevent and treat complications. These approaches may complement one another, with standard of care serving as a foundation for exploratory approaches. P2Y12 inhibition, at the top of the figure, is the mechanism affected by ticagrelor. While inhibition of this transmembrane molecule blocks platelet activation (and aggregation via increase GP IIb/IIIa expression), there are other important mechanisms that are candidates that may be additive or alternative to P2Y12 inhibition. The red question marks indicate targetable mechanisms that have been investigated with preclinical animal and in vitro models. There is particularly robust preclinical data to support targeting plateletneutrophil interactions. Meanwhile, the standard of care, including universal newborn screening, immunizations, and antibiotic prophylaxis in childhood, needs to be expanded to include people with sickle cell disease living in low and middle income countries, regardless of participation in clinical trials.
The identification of novel therapeutic targets, like platelet activation shown here, builds upon established interventions to diagnose sickle cell disease and prevent and treat complications. These approaches may complement one another, with standard of care serving as a foundation for exploratory approaches. P2Y12 inhibition, at the top of the figure, is the mechanism affected by ticagrelor. While inhibition of this transmembrane molecule blocks platelet activation (and aggregation via increase GP IIb/IIIa expression), there are other important mechanisms that are candidates that may be additive or alternative to P2Y12 inhibition. The red question marks indicate targetable mechanisms that have been investigated with preclinical animal and in vitro models. There is particularly robust preclinical data to support targeting plateletneutrophil interactions. Meanwhile, the standard of care, including universal newborn screening, immunizations, and antibiotic prophylaxis in childhood, needs to be expanded to include people with sickle cell disease living in low and middle income countries, regardless of participation in clinical trials.
For now, ticagrelor monotherapy joins a growing list of agents with no apparent benefit for preventing vaso-occlusive crisis defined as painful events or acute chest syndrome. Vaso-occlusive crisis prevention is a challenging endpoint for SCD drug trials as structural, personal, and biological attributes contribute to experiences of pain and opportunities to receive treatment or pursue care for pain.6 Geographic location and cultural norms affect patient-reported pain perception, pain intensity, and analgesic use and may affect whether patients present to clinics or hospitals for care.7 This complicates the use of vaso-occlusive crisis as an endpoint in multinational studies and may help explain the HESTIA3 investigators’ inclusion of painful events managed at home. Laboratory measures are not intrinsically preferable to patient-reported outcomes but formed the basis for the Food and Drug Administration’s approval of voxelotor, a drug that raises hemoglobin, on average, 1 g/dL. As adults with SCD accumulate morbid and mortal end-organ damage we would ideally have evidence that new treatments ameliorate this relentless injury, even if they do not reduce painful events. Unfortunately, changes to markers of end-organ functionare unlikely endpoints for time-constrained clinical trials. Ultimately, trials that rely solely on laboratory endpoints risk overlooking the patient's experience as a valid study endpoint while those that focus on SCD’s hallmark symptom, pain, also have challenges.
HESTIA3 reminds us of additional SCD challenges: enduring disparities in SCD outcomes. The study enrolled a multinational population of children mostly from low- and middle-income countries (85%). This multinational approach achieves two goals: it includes children from countries with the highest burden of SCD in clinical trials and may accelerate trial recruitment by including regions with high disease prevalence. The multinational approach also highlights sobering SCD realities. In this study, four children died, all from Africa or Asia. The randomized controlled trial of hydroxyurea for children in Sub-Saharan Africa had similar mortality rates.8 The deaths of children with SCD in a clinical trial, who likely had better access to hematology care than local peers, are a grim reminder of the early mortality caused by SCD in Africa and Asia. Aggressive care and infrastructure expansions are needed to provide life-saving preventive, supportive and disease-modifying care to people with SCD globally. It is unrealistic to imagine that industry-sponsored trials alone will deliver the changes needed to ensure that in Africa and Asia children also achieve near-universal survival into adulthood.
HESTIA3 included a genetically homogeneous populationof children with hemoglobin SS and hemoglobin Sβ0-thalassemia Accounting for heterogeneous care contexts is more complicated; subjects were required to receive standard SCD care and vaccinations in accordance with local guidelines. Nevertheless, deaths from suspected or confirmed meningitis and septicemia occurred; both death and meningitis are rare in children with SCD in the high-income countries, in part due to widespread implementation of highly effective vaccines. An encouraging 64% of children in this study took stable doses of hydroxyurea pointing toclinically realities and care centers' care standards, howeverhydroxyurea use by region was not reported. Regional use data will be especially important when positive multinational, randomized controlled trials provide opportunities to expand the therapeutic armamentarium for SCD. When this happens, the external validity of randomized controlled trial data will be a clinically relevant concern.9
The publication of HESTIA3 can invigorate the work ahead, informing future mechanistic approaches to treating SCD. We also hope that reporting of region-specific care standards and trial data in multinational studies will provide information to advance construction of a strong care floor for people with SCD around the world, many of whom would benefit from access to established interventions alongside access to trials developing novel SCD therapeutics.
Conflict-of-interest disclosure: L.H.P. is a consultant for Global Blood Therapeutics. The remaining author declare no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal