Abstract
Introduction. Optical genome mapping (OGM, Bionano Genomics) is a novel whole genome approach based on the imaging of long DNA molecules that allows detecting structural and copy number variants (CNV) at a high resolution. Recent publications in several hematologic malignancies have demonstrated the good performance of OGM to identify clinically relevant genetic aberrations, otherwise found by karyotyping, FISH and/or genomic microarrays. Despite its high potential as a diagnostic tool, consensus guidelines for OGM implementation in the clinical setting are still lacking. The aims of the study were: (1) To describe the inter-laboratory reproducibility in the identification and reporting of clinically relevant cytogenomic variants by OGM; (2) To outline the main causes of discrepancies among centers, to be addressed in future OGM recommendations.
Patients and Methods. Six Spanish centers participated in this pilot inter-laboratory comparison. A total of 12 patients were included, analyzing three centers each. The analyzed samples comprised tumoral specimens from two chronic lymphocytic leukemia (CLL), one follicular lymphoma (FL), two multiple myeloma (MM), two primary myelofibrosis (PMF), one acute myeloid leukemia (AML), two adult T-cell acute lymphoblastic leukemia (T-ALL) and two pediatric B-cell acute lymphoblastic leukemia (B-ALL). High molecular weight DNA from peripheral blood (n=6) or bone marrow (n=6) samples was labelled, processed and imaged in a Saphyr System (Bionano Genomics). Molecule files from the rare variant analysis (obtained from the Bionano Access software) were shared among hospitals and each center analyzed the samples following the manufacturer's filtering/analysis recommendations. The concordance rate was calculated taking into account those variants included in the final curated variant list and those that would be included in the clinical report.
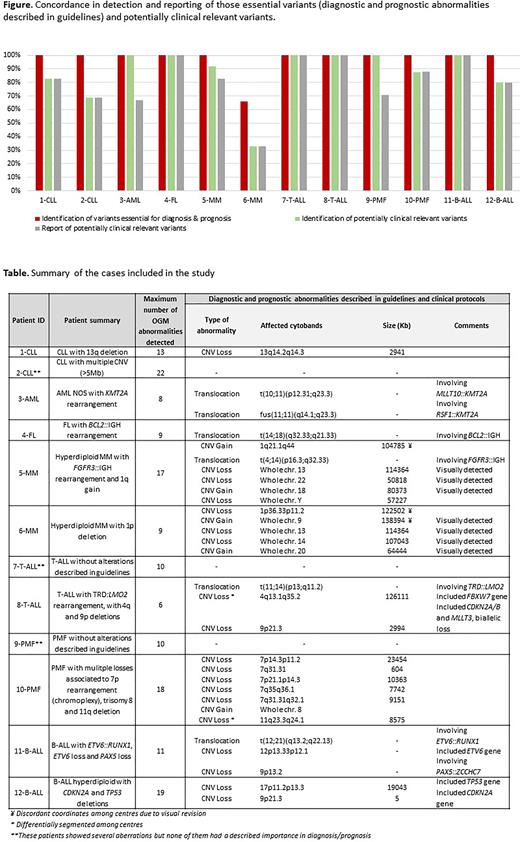
Results. Globally, 152 variants were recorded by at least one center (median: 11/sample, range: 6-22), being 100 CNV, 44 translocations and 8 small insertions or inversions. Among them, 98 (68%) were considered clinically relevant as they were classified as pathogenic (disease-related or CNV >5Mb, n=62) or likely pathogenic (CNV<5Mb associated with translocations or translocations associated with pathogenic CNV, n=36). The remaining 54 were classified as variants of unknown significance (VUS). Regarding the detection of pathogenic and likely-pathogenic variants, the global concordance among centers was 83%, being 100% in 8/12 cases, around 90% in 3/12 cases and only 33% in one case. The latter had a low tumor burden and one laboratory did not perform a visual revision of the whole genome data (Figure). Remarkably, except for this case, 100% of concordance was obtained in the detection of the diagnostic and prognostic abnormalities described in guidelines (Table). Similar concordance rates were obtained for rearrangements (79%) and CNV gains and losses (86% and 85%, respectively). Concerning the inclusion in the clinical reports of those variants detected by all the centers, 94% (75/80) were concordantly reported. Discordances in the reporting included 3 pathogenic variants (one complex rearrangement and 2 losses with a low infiltration) and 2 likely pathogenic variants (complex rearrangements associated with CNV). In contrast, only 6/54 VUS (11%) were concordantly identified (3 CNV <500Kb and 3 translocations) and none of them was reported by all the centers.
Conclusions. 1. The inter-laboratory concordance for the identification of the clinically relevant (diagnostic and/or prognostic) abnormalities was optimal. 2. Nevertheless, certain potential pathogenic aberrations were discordantly classified and/or reported; discrepancies were explained by a variable previous knowledge on the Bionano Access software, different filtering/analysis criteria and diverse professional expertise; 3. Future OGM international guidelines are needed to harmonize the introduction of such cytogenomic results in the clinical reports for hematological malignancies.
Acknowledgements: 2017SGR437 and 2017SGR288 and CERCA Programme, Generalitat de Catalunya; Fundació Internacional Josep Carreras; PI19/01828 and PI21/00022 from ISCIII; Premios FIHGUV 20/425; SEHH (C. G-G personal grant) and ACIF/2021/169, from Generalitat Valenciana (R.G-S predoctoral contract).
Disclosures
García-Serra:AbbVie: Other: received educational activity; Novartis: Other: received educational activity.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal