Abstract
Introduction Diffuse large B-cell lymphoma (DLBCL) is the most common B-cell non-Hodgkin lymphoma. The risk stratification tools continue to evolve and currently, there are various known prognostic factors including revised international prognostic index (R-IPI), MYC/BCL2 overexpression by IHC, MYC/BCL2, and or BCL6 rearrangement (double hit/triple hit lymphomas [DHL/THL]), CD5 expression, and metabolic tumor volume. Outside of case reports and small case series (Muringampurath-John, D et al BJH 2012), the relevance of circulating lymphoma (CL) in patients with DLBCL at diagnosis is largely unknown. Hence, we sought to evaluate the impact of CL at diagnosis on outcomes in patients with DLBCL.
Methods This is a single-center, retrospective cohort study of DLBCL patients treated at Ohio State University and included patients who were ≥18 years old, diagnosed with DLBCL from 2010-2021, and had information on flow cytometry at diagnosis. Patients were grouped according to positive or negative flow cytometry results as CL+ and CL-, respectively. The primary endpoint was progression-free survival (PFS), and the secondary endpoint was overall survival (OS) in CL+ and CL- groups. PFS was defined as the time from the start of first-line therapy until lymphoma relapse/progression or death from any cause, censoring at the last clinical assessment. OS was defined as the time from the start of first-line therapy until death or last follow-up. The first-line therapy was categorized into two groups, R-CHOP, and intensive induction therapy (R-EPOCH, R-hyperCVAD, and R-CODOX-M/R-IVAC).
Results Among the 181 eligible patients with newly diagnosed DLBCL, 30 (17%) had detectable CL at diagnosis, while 151 (83%) did not. Patients in the CL+ group had a higher median age (66 vs 64 years), more advanced stage disease (97% vs 83%), and R-IPI score of 3-5 (89% vs 62%) compared to the CL- group. The two groups were balanced in regard to gender, race, ECOG performance status, bulky disease, presence of B symptoms, cell of origin, synchronous central nervous system (CNS) involvement, DHL/THL, and first-line therapy.
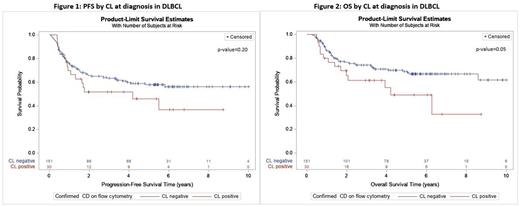
The median PFS was 4.2 years (95%CI=0.9-not reached [NR]) in the CL+ group compared to 10.9 years (95%CI=4.8-NR) in the CL- group (p=0.20, Figure 1). While the median PFS was not significantly different between CL+ and CL- groups among patients treated with R-CHOP chemotherapy (median PFS NR vs 10.9 years, respectively, p=0.99). However, compared to the CL+ group, the median PFS was significantly longer in the CL- group receiving intensive induction therapy (median PFS 1.8 years vs NR, respectively, p=0.04).
After adjusting for factors associated with PFS in multivariable cox regression (gender, R-IPI, first-line therapy, synchronous CNS involvement, and DHL/THL), there was no difference in PFS between CL+ and CL- groups (HR=1.48, 95%CI=0.77-2.85, p=0.24).
The median OS was 4.2 years (95%CI=1.7-NR) in the CL+ group while it was NR in the CL- group (p=0.05, Figure 2). After adjusting for factors associated with PFS in multivariable cox regression (gender, R-IPI, first-line therapy, synchronous CNS involvement, and DHL/THL), there was no difference in OS between CL+ and CL- groups (HR=1.85, 95%CI=0.90-3.79, p=0.09).
Conclusions This is the first study to compare the outcomes of patients with CL+ and CL- groups in newly diagnosed DLBCL. In this largest study to date evaluating the prognostic relevance of CL in patients with DLBCL, we found that CL at diagnosis was not associated with a shorter PFS but trended towards inferior OS. The CL+ group represents a potentially high-risk patient population and needs to be prioritized for experimental and cellular therapies at first relapse. Future prospective studies should include testing for CL and validate the findings found in our study.
Disclosures
Sawalha:TG Therapeutics: Research Funding; Celgene/BMS: Research Funding; BeiGene: Research Funding; Epizyme: Consultancy. Voorhees:Incyte: Research Funding; AstraZeneca: Research Funding. Maddocks:AstraZeneca: Consultancy; Genentech: Consultancy; Gilead: Consultancy; Genmab: Consultancy; Lilly: Consultancy; Kite: Consultancy; Incyte: Consultancy; Acerta: Consultancy; ADC Therapeutics: Consultancy; Morphosys: Consultancy; Janssen: Consultancy; Abbvie: Consultancy; Celgene: Consultancy; Beigene: Consultancy; BMS: Consultancy, Research Funding; Pharmacyclics: Consultancy, Research Funding; Pfizer: Research Funding. Christian:Seattle Genetics: Research Funding; Triphase: Research Funding; Acerta: Research Funding; Celgene/Bristol-Myers Squibb: Research Funding; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees, Research Funding; Morphosys: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genmab: Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Millennium: Research Funding. Epperla:Incyte: Speakers Bureau; Novartis: Honoraria; TG Therapeutics: Other: Ad Board; BeiGene: Other: Ad Board; Seattle Genetics: Other: Ad Board; Pharmacyclics: Other: Ad Board.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal