In this issue of Blood, Verdier et al pioneer a new method to predict platelet reactivity from complete blood count data, and they apply it to identify associated genetic variants and explore associations with thrombotic diseases.1
Platelets play a critical role in hemostasis and thrombosis, and antiplatelet drugs are widely used in cardiovascular disease prevention and treatment. There is interindividual variability in platelet reactivity, as measured by platelet aggregation responses to various agonists.2 Identifying the genetic determinants of platelet reactivity could lead to a greater understanding of these differences and to the identification of additional targets for new antiplatelet drugs. Traditional methods for measuring platelet reactivity are difficult to implement at large scales, which has limited the platelet reactivity data that are available in epidemiological studies. Hypothesis-free genome-wide association studies have identified associated genetic variants in several genetic regions, including those near genes known to be relevant to platelets such as GP6 and PEAR1.2,3
In this issue, Verdier et al devised a new approach to predict platelet reactivity to adenosine diphosphate using complete blood count scattergrams from widely implemented Sysmex analyzers.1 The model was trained on 533 participants of the Cambridge Platelet Function Cohort. The predictive ability of this model was modest, with r2 = 0.26. The authors then used the approach to predict platelet reactivity in 29,806 participants of the INTERVAL study. Strikingly, the large sample size of the INTERVAL study ensures that the effective sample size after accounting for the modest predictive ability of the model is still considerably larger than of the largest published genome-wide association study of platelet reactivity.
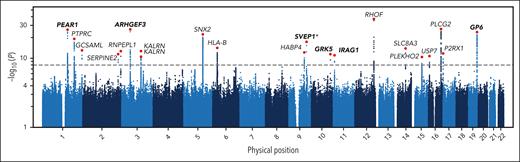
The authors identify 20 genetic regions containing variants associated with their predicted platelet reactivity phenotype (see figure), including 6 regions that have previously been identified by genome-wide association studies and 14 that have not. Many of the newly associated regions harbor candidate causal genes with plausible biological roles in platelet function. For example, SERPINE2 encodes a potent thrombin inhibitor, thereby reducing platelet activation by thrombin. Other candidate genes, such as PTPRC, GCSAML, and KALRN, also have evidence linking them to platelet activation pathways. Interestingly, some of the candidate genes likely mediate platelet reactivity to agonists other than adenosine diphosphate, suggesting that the predicted platelet reactivity phenotype may serve as a broader readout of platelet reactivity beyond adenosine diphosphate. Previously published evidence that these candidate genes may regulate platelet activation in cellular and animal models does not diminish the impact of these findings, since the present study represents a powerful in vivo demonstration of their importance in humans.
A Manhattan plot showing the results of the genome-wide association study of predicted platelet reactivity. Each dot corresponds to an included genetic variant: the position on the x-axis indicates the chromosome number and genomic position of the variant, and the position on the y-axis indicates −log10 of the P value of the variant. The horizontal dashed line corresponds to the genome-wide significance threshold. The gene names indicate the protein-coding gene that is nearest to each of these associations. Regions associated with a platelet reactivity phenotype by a previous genome-wide association study are shown in bold. See Figure 2 in the article by Verdier et al that begins on page 1895 for further details.1
A Manhattan plot showing the results of the genome-wide association study of predicted platelet reactivity. Each dot corresponds to an included genetic variant: the position on the x-axis indicates the chromosome number and genomic position of the variant, and the position on the y-axis indicates −log10 of the P value of the variant. The horizontal dashed line corresponds to the genome-wide significance threshold. The gene names indicate the protein-coding gene that is nearest to each of these associations. Regions associated with a platelet reactivity phenotype by a previous genome-wide association study are shown in bold. See Figure 2 in the article by Verdier et al that begins on page 1895 for further details.1
Verdier et al then went on to use the results from their genetic study to identify significant associations between genetically determined platelet reactivity and risk of thrombotic diseases, including coronary artery disease, ischemic stroke, and venous thromboembolism. This causal inference approach, called Mendelian randomization, aims to provide causal effect estimates free from confounding and reverse causation by using genetic variants associated with an exposure (such as platelet reactivity) as “instruments” to assess the association of the exposure with disease. Mendelian randomization leverages the fact that genetic variants are fixed at conception and do not change as a result of confounders or disease. Importantly, these analyses are based on first occurrence of thrombotic diseases, emphasizing platelet reactivity as a target for prevention as well as treatment. These analyses highlight the importance platelet function in these 3 thrombotic diseases. The association with coronary artery disease is notable because the vast majority of identified genetic associations appear to be driven by atherosclerotic plaque development and not by the thrombotic response to plaque rupture.4
Although the insights gleaned from the first application of this approach are impressive in their own right, the true promise of this approach lies in future applications on even larger data sets, which would expand our understanding of the genetic architecture of platelet reactivity and potentially lead to the discovery of novel drug targets.
Conflict-of-interest disclosure: P.S.d.V. declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal