Introduction
Follicular lymphoma (FL) is typically indolent, but relapses and transformation to higher grade disease are common. FL is often radiosensitive and can show complete response (CR) even to very low dose radiotherapy (VLDRT, 4Gy). There are no known genetic markers of FL radiosensitivity. We sought to identify molecular signatures of FL radiosensitivity to aid in patient selection for VLDRT.
Methods
We analyzed our institutional database and identified 110 FL patients with 113 tumors treated with radiotherapy (RT) and preexisting MSK-IMPACT, a targeted exon sequencing panel. For each patient, we collected key clinicodemographic information including pre- and post-RT treatment history, prior large cell lymphoma, and Ann Arbor stage at RT. For treated tumors, we obtained tumor site, grade, diameter, maximum SUV, BCL2 translocation (trans) status through fluorescence in situ hybridization, RT dose and fractionation, and the treatment strategy ( i.e. treating all sites of disease vs a subsite of disease). Tumor site was dichotomized by pelvic tumors vs all others, as this was the most frequently altered site. The presence of mutant (mut) genes compared to wild type (WT) were associated with RT response, measured by PET scan within 6 months of RT, using logistic regression. Log-rank testing and Cox proportional hazards models were used to analyze local progression free survival (LPFS), censored at start of unplanned therapy, with 2-year (2y) survival reported. Multivariate modeling was used to adjust gene associations with outcomes, controlling for the clinicodemographic and tumor characteristics mentioned above.
Results
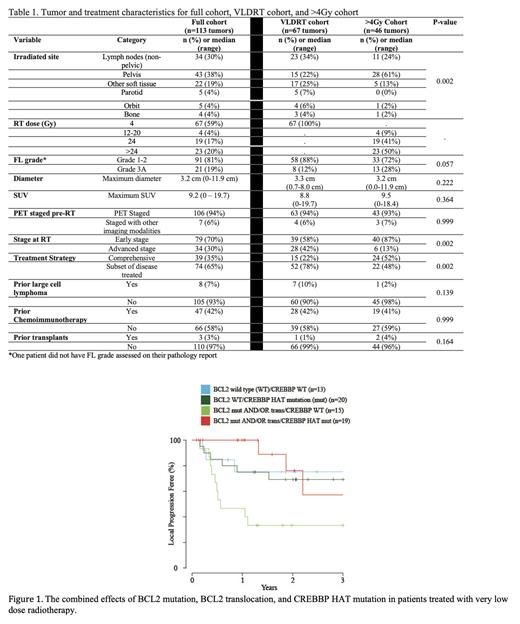
The patient and tumor characteristics are summarized in table 1. We found CREBBP to be the most frequently altered gene (66% of tumors). We identified 5 signatures of altered genes, most of which showed altered CREBBP and relatively long LPFS (e.g. one signature showed CREBBP mut, TNFRSF14 mut and IRF8/STAT6 mut with a 2y LPFS 78%); however, one signature with concurrent KMT2D mut and altered BCL2 including trans and mut had significantly shorter LPFS than all other signatures (2y LPFS 47%, p < 0.01) and lower odds of CR relative to the other signatures (p= 0.03). CREBBP mut was the only alteration associated with increased odds of CR (Odds ratio: 2.39 (95% CI: 1.06-5.37, p = 0.04), and this effect remained significant after adjusting for pelvic disease site (p=0.04). Mutations of BCL2, IRF8, and KMT2D were associated with LPFS using log-rank testing (p<0.01, p=0.02, and p=0.04, respectively). BCL2 mut or concurrent BCL2 mut and BCL2 trans (mut/trans) had shorter LPFS for the overall cohort (p<0.01 for both), while for VLDRT, only BCL2 mut/trans was associated with shorter LPFS (p=0.02). In the VLDRT cohort, we found that CREBBP histone acetyltransferase (HAT) mut was associated with improved LPFS compared to WT (n=39, 2y LPFS 74% vs 52%, HR:0.41 (95% CI: 0.18-0.93, p=0.03)), but this was not observed for tumors receiving >4Gy (n=44, HR: 1.27 (95% CI: 0.32-5.08), p=0.74)). We observed that either BCL2 mut, BCL2 trans, or BCL2 mut/trans and CREBBP HAT WT had a 2y LPFS of 13% whereas BCL2 mut, BCL2 trans or BCL2 mut/trans and CREBBP HAT mut had a 2y LPFS of 83% (p<0.01), and this relationship persisted on multivariate analysis (Figure 1).
Conclusions
Incorporating genetic signatures associated with radiosensitivity, and specifically alterations involving BCL2 and CREBBP, may independently improve patient selection for RT. CREBBP HAT domain mutations are potentially targetable and may have important implications for augmenting radiosensitivity to VLDRT.
Disclosures
Lebow:Oncia Technologies, Inc: Current equity holder in publicly-traded company. Joffe:Beigene: Honoraria; Abbvie: Honoraria. Dogan:Seattle Genetics: Consultancy; Physicians' Education Resource: Consultancy, Honoraria; EUSA Pharma: Consultancy; Loxo: Consultancy; Peer View: Honoraria; Incyte: Consultancy; Takeda: Other: Research Funding; Roche: Other: Research Funding. Zelenetz:BeiGene: Consultancy, Honoraria, Research Funding; BMS: Consultancy, Honoraria; None other than mutual funds (401K): Current equity holder in publicly-traded company; F. Hoffmann-La Roche Ltd: Consultancy, Honoraria, Research Funding; SAB: Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Consultancy, Honoraria; MEI Pharma Inc: Consultancy, Honoraria, Research Funding; Lymphoma Research Foundation: Membership on an entity's Board of Directors or advisory committees; Janssen Pharmaceuticals: Consultancy, Honoraria; Pharmacyclics: Consultancy, Honoraria; Abbvie: Research Funding; Gilead: Consultancy, Honoraria. Salles:Janssen: Consultancy, Research Funding; Nurix: Consultancy; Ipsen: Consultancy, Research Funding; Orna: Consultancy; Nordic Nanovector: Consultancy; BeiGene: Consultancy; BMS/Celgene: Consultancy; Debiopharm: Consultancy; ATB Therapeutics: Consultancy; Kite/Gilead: Consultancy; Merck: Consultancy, Honoraria; Molecular Partners: Consultancy; Incyte: Consultancy; Novartis: Consultancy; Loxo/Lilly: Consultancy; Genmab: Consultancy; Genentech, Inc./F. Hoffmann-La Roche Ltd: Consultancy, Research Funding; Owkin: Current holder of stock options in a privately-held company; EPIZYME: Consultancy; AbbVie: Consultancy, Honoraria. Imber:GT Medical Technologies: Honoraria. Yahalom:Convergent R.N.R Ltd.: Other: Provision of Services (uncompensated).

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal