Presence of deletion17p (del17p) in frontline (1L) and relapsed/refractory (R/R) CLL has been associated with shorter time to treatment initiation, early relapse after therapy and inferior overall survival (OS) (Mato et.al., Haemotologica 2022). However, there exists notable clinical heterogeneity in terms of outcome within this group (Tam et.al., Blood 2009, Stephens et.al; AJH 2015). Previous reports in 1L and R/R CLL have identified percentage of del17p nuclei by fluorescence in situ hybridization (FISH), having TP53 mutations on the second allele, copy number alterations and complex karyotype (CK) as additional risk factors within the del17p population (Yu et al., CCR 2017; Baliakas et al., Blood 2019).
Here we profile a cohort of wait and watch (W&W, n=14), 1L treated (n=36) and R/R CLL (n=3) patients by whole genome sequencing (WGS) and RNA sequencing (RNAseq) to gain insights into genomic factors and gene expression pathways contributing to the high-risk nature of del17p across the spectrum of CLL patients.
Using the Mayo Clinic CLL Database, we identified 53 patients with del17p (identified by FISH testing in routine clinical practice at diagnosis) who had stored peripheral blood mononuclear cells (PBMCs) available. RNA and DNA from matched peripheral blood (tumor) and DNA from CD19- (germline) samples at diagnosis (n=50) and relapse (n=3) was extracted using Qiagen RNeasy (RNA) and Purene (DNA) kits and subjected to RNAseq and WGS respectively. Additional FISH assays (del11q, trisomy 12, del13q) and CK by chromosomal banding analysis (CBA) were performed. Baseline clinical characteristics, time to first treatment, types of treatment, and survival were abstracted from the CLL Database. The Mayo Clinic IRB approved this study.
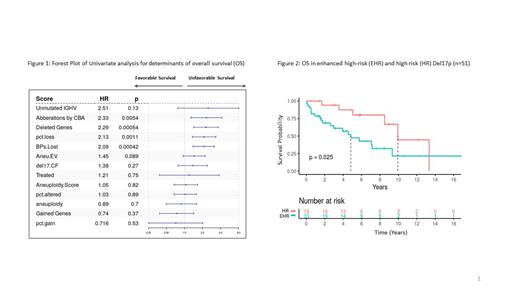
Copy number analysis identified deletions in 3p, 4p, 8p, 9p, 9q and gains in 2p, 8q, 15q and 17q chromosomal lesions occurring in patients. We explored the association of clinical and genetic factors, and genomic instability signatures with survival to identify high-risk features in del17p patients. Both total number of deletions by WGS and number of chromosomal aberrations by CBA as a continuous variable were significantly associated with unfavorable OS in the entire cohort (n=53) in a univariate analysis (Hazard ratio, HR= 2.29, p= 0.00054 and HR= 2.33, p=0.0054 respectively). However, there was no significant association of number of gained genes by WGS or aneuploidy scores with survival (HR=0.74, p=0.37). In patients with CK data (n=41), deletion burden, but not gains, were highly correlated to chromosomal aberrations by CBA. Using cutpointr package for identifying deletion threshold corresponding to CK≥ 3, we defined patients with deletion burden ≥ 514 as enhanced-high risk (EHR; median OS = 4.83 years) compared to the group with high-risk (HR; median OS=9.94 years) (p=0.025). There was no significant difference in number of deleted genes between WW and treated patients. The EHR group is enriched in patients with high clonal cell fraction of del17p by FISH, unmutated IGHV and CK≥ 3. The EHR group remains significant in a multi-variable analysis with IGHV-mutation status, ZAP70, and age (HR: 5.16, 95% CI: 1.04 - 25.6, p=0.04).
Mutational analysis identified 35 known CLL driver mutations with TP53 (86%), ATM (15%), NOTCH1(13%), MGA (13%), CHD2 and MED12(11%) as the top mutated genes. Total number of driver genes mutated was not significantly different between EHR and HR subgroups, but the EHR subset was significantly enriched with mutations in NOTCH1 (21%), and MGA (21%) as compared to no mutations in the HR group in these genes. Deleted genes (≥ 514) was prognostic in bi-allelic TP53 aberration patients as well (p=0.021)
Differential expression gene-set enrichment analysis indicated a significant enrichment of DNA repair, cellular response to DNA damage and downregulation of inflammatory responses in the EHR group.
CK is a known prognostic marker which includes both chromosomal losses and gains. In this small cohort of del17p we were able to show the prognostic value of deletions only, but not gains, as captured by WGS. Using genome wide sequencing, we identify increasing genomic deletions as a feature of enhanced-high risk del17p. While deletion burden cut-offs identified here are specific to our research method and require further validation in additional independent cohorts, EHR subgroup remained significant after adjusting for other known prognostic variables .
Disclosures
Trisal:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Stong:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Parikh:Bristol Myers Squibb-Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Agios Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Accerta Pharmaceuticals: Research Funding; AbbVie Inc: Membership on an entity's Board of Directors or advisory committees, Research Funding; Dava Oncology: Membership on an entity's Board of Directors or advisory committees; Boehringer Ingelheim Pharmaceuticals Incc: Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Research Funding; Dren Bio: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees; CSL Behring: Membership on an entity's Board of Directors or advisory committees; Beigene: Membership on an entity's Board of Directors or advisory committees; Sunesis: Research Funding; Vincerx: Research Funding. Huang:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Gandhi:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Kay:Vincerx: Research Funding; Beigene: Membership on an entity's Board of Directors or advisory committees; Bristol Meyer Squib / Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Acerta Pharma: Research Funding; Genentech: Research Funding; Sunesis: Research Funding; Pharmcyclics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees; Dren Bio: Membership on an entity's Board of Directors or advisory committees; Behring: Membership on an entity's Board of Directors or advisory committees; Astra Zeneca: Membership on an entity's Board of Directors or advisory committees; boehringer ingelheim: Membership on an entity's Board of Directors or advisory committees; Agios Pharm: Membership on an entity's Board of Directors or advisory committees; Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees; Dava Oncology: Membership on an entity's Board of Directors or advisory committees; Abbvie: Membership on an entity's Board of Directors or advisory committees, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal