The NK receptor ligands, PVR and B7-H6, are required for AML cell recognition and immunological synapse formation with DOT cells.
PVR expression predicts primary AML sample targeting by cytotoxic DOT cells.
Visual Abstract
Relapsed or refractory acute myeloid leukemia (AML) remains a major therapeutic challenge. We have recently developed a Vδ1+ γδ T cell–based product for adoptive immunotherapy, named Delta One T (DOT) cells, and demonstrated their cytolytic capacity to eliminate AML cell lines and primary blasts in vitro and in vivo. However, the molecular mechanisms responsible for the broad DOT-cell recognition of AML cells remain poorly understood. Here, we dissected the role of natural killer (NK) cell receptor ligands in AML cell recognition by DOT cells. Screening of multiple AML cell lines highlighted a strong upregulation of the DNAM-1 ligands, CD155/pulmonary vascular resistance (PVR), CD112/nectin-2, as well as the NKp30 ligand, B7-H6, in contrast with NKG2D ligands. CRISPR-mediated ablation revealed key nonredundant and synergistic contributions of PVR and B7-H6 but not nectin-2 to DOT-cell targeting of AML cells. We further demonstrate that PVR and B7-H6 are critical for the formation of robust immunological synapses between AML and DOT cells. Importantly, PVR but not B7-H6 expression in primary AML samples predicted their elimination by DOT cells. These data provide new mechanistic insight into tumor targeting by DOT cells and suggest that assessing PVR expression levels may be highly relevant to DOT cell–based clinical trials.
Introduction
Acute myeloid leukemia (AML) remains an unmet medical need, given its relatively high incidence and morbidity and mortality rates because of chemotherapy resistance and relapse in the absence of breakthrough therapies.1 In contrast to B-cell malignancies, in which adoptive autologous chimeric antigen receptor (CAR) T-cell therapies have delivered impressive rates of complete responses and changed clinical practice,2 AML remains a major challenge for current immunotherapies, especially because of on-target off-tumor effects on vital healthy myeloid progenitor cells that share expression of targetable antigens like CD33 or CD123, thus promoting a life-threatening myeloablation.3 By contrast, nonengineered lymphocytes, although likely less potent, may preserve their ability to distinguish AML cells from healthy progenitors, thus avoiding severe myeloablation. Such is the case of Delta One T (DOT) cells, a cell therapy product based on Vδ1+ γδ T cells,4-6 which display robust cytotoxic potency against multiple AML cell lines and samples from patients with primary AML, including chemotherapy-resistant AML cells, while sparing normal CD33+ or CD123+ myeloid cells and progenitor cells.7 Moreover, we recently showed that, although requiring a higher effector-to-target ratio to achieve equivalent AML elimination compared with CD123 CAR-transduced DOT cells, nonengineered DOT cells were able to control AML growth in patient-derived xenografts.8 This is highly relevant because DOT cells have been evolved into an allogeneic cellular product that is being tested in patients with AML (ClinicalTrials.gov identifier: NCT05886491).
On the path to a DOT cell–based treatment for AML, a fundamental question urges: how do DOT cells recognize AML cells, that is, which molecules on the surface of AML cells are essential for efficient cytotoxic targeting by DOT cells? We previously reported a striking T-cell receptor (TCR) polyclonality of DOT-cell products, and a very limited impact of anti-TCRVδ1 antibody blockade on AML targeting by DOT cells.7 In contrast, we have clearly demonstrated that DOT cells upregulate multiple natural killer (NK) cell receptors (NKR), especially NKp30, NKG2D, and DNAM-1, when compared with ex vivo Vδ1+ γδ T cells.4,5 Building on these foundations, here we hypothesized that the broad reactivity of DOT cells against AML cells is NKR mediated and thus dependent on the expression of key counter NKR ligands on AML cells, which we set out to identify.
Study design
Ethics
Primary AML cells were obtained from diagnostic bone marrow or peripheral blood samples, after informed consent and institutional review board approval, in accordance with the Declaration of Helsinki.
DOT cells, AML samples, and cell lines
DOT cells were analyzed for their NKR phenotype, as well as for the expression of TIGIT and CD96, by flow cytometry (supplemental Figure 1, available on the Blood website). In this study we used DOT cells with <10% of TIGIT+ cells. Primary AML samples were used either fresh or cryopreserved, upon a 2-hour incubation after thawing in RPMI 1640 supplemented with 20% fetal bovine serum, 1% penicillin/streptomycin, 100 μg/mL of stem cell factor and FMS-like tyrosine kinase 3, and 20 μg/mL of thrombopoetin. The source and culture media of AML cell lines are summarized in supplemental Table 1. Genetic deletion of pulmonary vascular resistance (PVR) gene, nectin-2, and B7-H6 was performed by cloning the guide RNAs (supplemental Table 2) into the PX459 plasmid (Addgene), as previously described.9 Cells were electroporated using Neon Transfection System (1300 V, 20 ms, 2 pulses). Knockout (KO) cells were sorted via fluorescence-activated cell sorting based on protein expression. Analysis of ligand expression was performed using the antibodies provided in supplemental Table 3, upon incubation with Fc receptor inhibitor. A given AML sample was considered positive for a specific ligand when the difference in percentage of positive cells between the test and isotype control samples was >5%. Messenger RNA from AML cells was isolated, reverse transcribed, and quantified, as previously described.10 Primer sequences are summarized in supplemental Table 2.
AML cell targeting
For killing assays, AML cell lines or primary samples were stained with a dye and washed before coincubation with DOT cells at an effector-to-target ratio of 5:1, for 3 or 24 hours, in the presence of 100 ng/mL of interleukin-15 (Peprotech). For blocking experiments, cells were preincubated for 15 minutes at 4°C with blocking antibodies (supplemental Table 3). Cells were stained with annexin V and analyzed in an LSR Fortessa (BD Biosciences) and data analyzed with FlowJo software (Tree Star). For imaging experiments, cells were stained with different dyes for 10 minutes at room temperature before coincubation; then fixed with 1.5% paraformaldehyde and stained for phalloidin during the permeabilization step (phosphate-buffered saline + 0.1% Triton X-100 + 2% fetal bovine serum). Before acquisition in Amnis ImageStreamX (Luminex), 4′,6-diamidino-2-phenylindole was added. Results were analyzed using IDEAS software. Details of concentrations are provided in supplemental Table 3.
Statistical analysis
Individual values, mean, and standard error of the mean are plotted. The statistical tests used, and significant P values are indicated in each figure.
Results and discussion
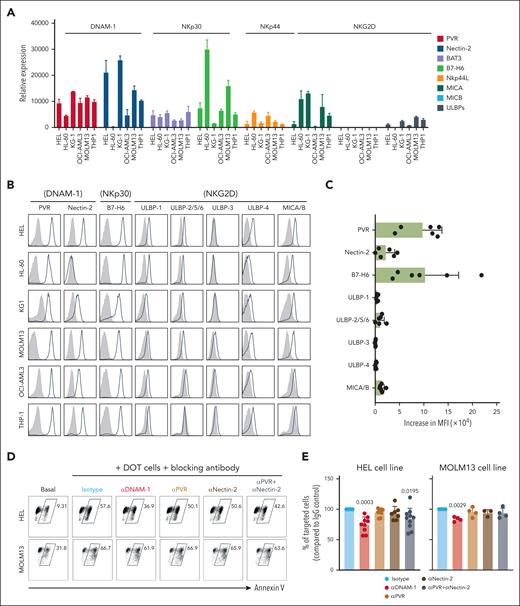
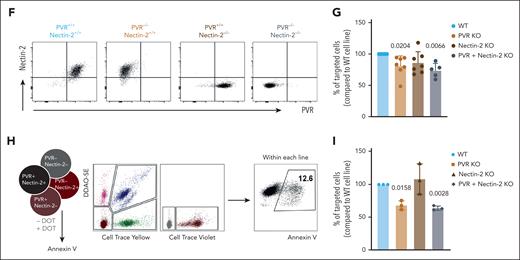
To study NKR-ligand expression on a panel of AML cell lines (supplemental Table 1), we quantified both messenger RNA (Figure 1A) and surface protein (Figure 1B) levels and found a marked upregulation of PVR and B7-H6, which contrasted with milder expression of nectin-2 and very low NKG2D ligands (Figure 1A-C). We first focused on DNAM-1 and its ligands, PVR and nectin-2, emerging players in tumor immunology,11 previously shown to be important for Vδ2+ γδ T-cell responses to liver or brain cancers.12-14 We used blocking antibodies in 3-hour killing assays against HEL or MOLM13 AML cells, and found a 20% to 30% decrease in DOT-cell cytotoxicity upon DNAM-1 neutralization (Figure 1D-E). However, because ligand blockade failed to reproduce this effect, we used a more stringent methodology, CRISPR–CRISPR-associated protein 9 gene editing, to knock out each or both ligands (Figure 1F). Upon coincubation with DOT cells, we found lower targeting (compared with mock transduced) of PVR KO but not nectin-2 KO MOLM13 cells, whereas the double KO behaved similarly to PVR KO cells (Figure 1G). We then designed competitive killing assays in the presence of all the edited HEL lines labeled with different fluorescent dyes (Figure 1H). Consistently, PVR KO (and double KO) but not nectin-2 KO AML cells were ∼40% less targeted than control HEL cells (Figure 1I).
The DNAM-1 ligand, CD155/ PVR is required for DOT-cell targeting of AML cell lines. (A) Gene expression of ligands for DNAM-1, NKp30, NKp44, and NKG2D receptors in a panel of AML cell lines; results are normalized to the housekeeping genes GUSB and PSM6. (B) Protein expression of ligands for DNAM-1, NKp30, NKp44, and NKG2D receptors in a panel of AML cell lines; gray histograms represent isotype; black lines represent sample. (C) Summary of flow cytometry data depicted in panel B. Data represented as mean fluorescence intensity (MFI) increase relative to isotype control. (D) Representative flow cytometry plots of killing assay performed in the absence (basal) or presence of DOT cells that were previously incubated with blocking antibodies for the proteins indicated. (E) Summary of killing assays performed against HEL (left) and MOLM13 (right) cell lines. Results are normalized to the percentage of tumor cells targeted in the condition in which DOT cells are incubated with isotype control antibodies. Each dot represents a different DOT-cell donor. Data were generated in ≥3 independent experiments. (F) Flow cytometry plots depicting the phenotype of MOLM13 cell lines modified via CRISPR–CRISPR-associated protein 9 editing. (G) Summary of killing assays performed against the indicated KO cell lines, compared with the mock-transfected cell line. Each dot represents a different DOT-cell donor. Data were generated in >3 independent experiments. (H) Schematic representation of the experimental design of competitive killing assays. (I) Summary of killing assays performed against the indicated KO HEL cell lines in a competitive setting, compared with the mock-transfected cell line. Each dot represents a different DOT-cell donor. Data were generated in 3 independent experiments. Statistical analysis was performed using 1-sample t test (hypothetical value: 100). WT, wild type.
The DNAM-1 ligand, CD155/ PVR is required for DOT-cell targeting of AML cell lines. (A) Gene expression of ligands for DNAM-1, NKp30, NKp44, and NKG2D receptors in a panel of AML cell lines; results are normalized to the housekeeping genes GUSB and PSM6. (B) Protein expression of ligands for DNAM-1, NKp30, NKp44, and NKG2D receptors in a panel of AML cell lines; gray histograms represent isotype; black lines represent sample. (C) Summary of flow cytometry data depicted in panel B. Data represented as mean fluorescence intensity (MFI) increase relative to isotype control. (D) Representative flow cytometry plots of killing assay performed in the absence (basal) or presence of DOT cells that were previously incubated with blocking antibodies for the proteins indicated. (E) Summary of killing assays performed against HEL (left) and MOLM13 (right) cell lines. Results are normalized to the percentage of tumor cells targeted in the condition in which DOT cells are incubated with isotype control antibodies. Each dot represents a different DOT-cell donor. Data were generated in ≥3 independent experiments. (F) Flow cytometry plots depicting the phenotype of MOLM13 cell lines modified via CRISPR–CRISPR-associated protein 9 editing. (G) Summary of killing assays performed against the indicated KO cell lines, compared with the mock-transfected cell line. Each dot represents a different DOT-cell donor. Data were generated in >3 independent experiments. (H) Schematic representation of the experimental design of competitive killing assays. (I) Summary of killing assays performed against the indicated KO HEL cell lines in a competitive setting, compared with the mock-transfected cell line. Each dot represents a different DOT-cell donor. Data were generated in 3 independent experiments. Statistical analysis was performed using 1-sample t test (hypothetical value: 100). WT, wild type.
Next, we inquired the role of the NKp30 ligand, B7-H6.15 We observed a reduction (compared with mock-transduced HEL cells) in B7-H6 KO killing by DOT cells, plus an additive effect (to 60% loss in reactivity) in PVR/ B7-H6 double-KO cells (Figure 2A). To complement these loss-of-function experiments, we ectopically expressed PVR, nectin-2, or B7-H6 in murine E0771 tumor cells, given their natural lack of expression of these human ligands (supplemental Figure 2). Upon coincubation with DOT cells selected for high expression of DNAM-1 or NKp30, we found that ectopic PVR and B7-H6 but not nectin-2 conferred susceptibility to DOT-cell targeting (Figure 2B). To gain further mechanistic insight, we analyzed cytotoxic molecule expression and formation of immune synapses between DOT cells and AML cells. We observed perforin induction on DOT cells upon coincubation with HEL cells, which was fully dependent on the presence of both PVR and B7-H6 (Figure 2C-D). Importantly, imaging synapse formation (via F-actin staining) also showed its synergistic dependence on PVR and B7-H6 (Figure 2F-G), thus strengthening the importance of these 2 ligands in AML cell line recognition by DOT cells. To validate these findings in vivo, we transplanted the KO cell lines into NOD scid gamma (NSG)-HuIL-15 transgenic mice and observed that the absence of PVR and B7-H6 impaired tumor clearance by DOT cells (Figure 2E).
PVR and B7-H6 are essential for formation of robust immunological synapses between DOT cells and AML cells. (A) Summary of killing assays performed against the indicated KO HEL cell lines, compared with the mock-transfected cell line. Each dot represents a different DOT-cell donor. Data were generated in 3 independent experiments. (B) Killing assay performed against the mouse breast cancer cell line E0771 previously transfected to express human PVR, nectin-2, or B7-H6 compared with the mock-transfected cell line. DOT cells were selected for high expression of the respective counterreceptors (DNAM-1 or NKp30). Each dot represents a technical replicate. Data were generated in 2 independent experiments. (C) Representative flow cytometry plots of granzyme B (GzmB) and perforin (Prf) expression on DOT cells, when in contact with different cell lines or in the presence of interleukin-15 (IL-15) only. (D) Summary of the flow cytometry data depicted in panel C. Each dot represents a DOT-cell donor. Data were generated in 2 independent experiments. (E) Tumor load in the blood of a xenograft model of AML. Immunodeficient NSG-HuIL-15 mice were engrafted with control or PVR/B7-H6 double-KO HEL (luciferase positive) cell lines. Mice were treated with DOT cells intravenously or left untreated. Three weeks after tumor injection, mice were euthanized and tumor load in the blood was quantified by luminescence. Data were generated in 2 independent experiments. (F) Representative images of image-flow cytometry data of immunological synapses established between DOT cells and different HEL cell lines. (G) Quantification of filamentous actin (F-actin) signal within the area of interaction between the DOT cell and the tumor cell. Data are representative of 2 independent experiments. Statistical analysis was performed using 1-sample t test (hypothetical value: 100 for panel A, or 0 for panel B), 1-way analysis of variance followed by Šídák multiple comparisons test for panel E, or Kruskal-Wallis test followed by Dunn multiple comparisons test for panels D and G. WT, wild type.
PVR and B7-H6 are essential for formation of robust immunological synapses between DOT cells and AML cells. (A) Summary of killing assays performed against the indicated KO HEL cell lines, compared with the mock-transfected cell line. Each dot represents a different DOT-cell donor. Data were generated in 3 independent experiments. (B) Killing assay performed against the mouse breast cancer cell line E0771 previously transfected to express human PVR, nectin-2, or B7-H6 compared with the mock-transfected cell line. DOT cells were selected for high expression of the respective counterreceptors (DNAM-1 or NKp30). Each dot represents a technical replicate. Data were generated in 2 independent experiments. (C) Representative flow cytometry plots of granzyme B (GzmB) and perforin (Prf) expression on DOT cells, when in contact with different cell lines or in the presence of interleukin-15 (IL-15) only. (D) Summary of the flow cytometry data depicted in panel C. Each dot represents a DOT-cell donor. Data were generated in 2 independent experiments. (E) Tumor load in the blood of a xenograft model of AML. Immunodeficient NSG-HuIL-15 mice were engrafted with control or PVR/B7-H6 double-KO HEL (luciferase positive) cell lines. Mice were treated with DOT cells intravenously or left untreated. Three weeks after tumor injection, mice were euthanized and tumor load in the blood was quantified by luminescence. Data were generated in 2 independent experiments. (F) Representative images of image-flow cytometry data of immunological synapses established between DOT cells and different HEL cell lines. (G) Quantification of filamentous actin (F-actin) signal within the area of interaction between the DOT cell and the tumor cell. Data are representative of 2 independent experiments. Statistical analysis was performed using 1-sample t test (hypothetical value: 100 for panel A, or 0 for panel B), 1-way analysis of variance followed by Šídák multiple comparisons test for panel E, or Kruskal-Wallis test followed by Dunn multiple comparisons test for panels D and G. WT, wild type.
Finally, we investigated the impact of NKR ligands on DOT-cell targeting of primary AML cells. Unlike AML cell lines (Figure 1B), primary AML showed variable levels of PVR, nectin-2, and B7-H6, and mostly lacked NKG2D ligand expression (Figure 3A; supplemental Figure 3A-B). This led us to segregate the patient samples into those that expressed (n = 9) or did not express (n = 4) any of the 3 ligands (Figure 3B), and to demonstrate clearly distinct extents of DOT-cell cytotoxicity (Figure 3C). By focusing on each of the ligands, we found PVR and nectin-2, which followed similar expression patterns (Figure 3B), but not B7-H6 or NKG2D ligands to predict DOT-cell cytotoxicity (Figure 3D; supplemental Figure 3C) with significant receiver operating characteristic curves (area under the curve, >0.75; Figure 3E; supplemental Figure 3D). Moreover, we found that incubation of DOT cells with AML samples that express PVR led to an upregulation of CD69 (Figure 3F), a T-cell activation marker, corroborating that PVR expression promotes DOT-cell activation. Altogether, our data indicate that PVR uniquely combines functional relevance, as assessed by genetic manipulation in AML cell lines, with predictive capacity with patient-derived AML samples, and we thus propose that its expression should be considered as a potential biomarker of response to Vδ1/DOT cell–based therapies in AML.
Expression of PVR but not B7-H6 predicts cytotoxic targeting of primary AML samples by DOT cells. (A) Representative flow cytometry histograms of the expression of PVR, nectin-2, and B7-H6. Each row depicts a different AML sample donor. Gray histograms, isotype; black lines, sample. (B) Distribution of PVR, nectin-2, and B7-H6 expression on all primary AML samples analyzed. (C) Targeting of primary AML samples by DOT cells based on the expression of 1 of 3 ligands shown in panels A-B. (D) Targeting of primary AML samples by DOT cells based on the expression of PVR, nectin-2, or B7-H6. (E) Receiver operating characteristic curves that illustrate the predictive ability of the expression of each of the depicted ligand(s) to discriminate whether primary AML samples are targeted by DOT cells. (F) Fold change of percentage of CD69+ cells (left) or CD69 MFI (right) on DOT cells that were cultured alone, or in the presence of PVR− or PVR+ primary AML samples. Each dot represents a different DOT cell/AML combination. Statistical analysis was performed using the Mann-Whitney U test for panels C-D, and Kruskal-Wallis followed by Dunn multiple comparisons test for panel E. AUC, area under the curve; w/o, without.
Expression of PVR but not B7-H6 predicts cytotoxic targeting of primary AML samples by DOT cells. (A) Representative flow cytometry histograms of the expression of PVR, nectin-2, and B7-H6. Each row depicts a different AML sample donor. Gray histograms, isotype; black lines, sample. (B) Distribution of PVR, nectin-2, and B7-H6 expression on all primary AML samples analyzed. (C) Targeting of primary AML samples by DOT cells based on the expression of 1 of 3 ligands shown in panels A-B. (D) Targeting of primary AML samples by DOT cells based on the expression of PVR, nectin-2, or B7-H6. (E) Receiver operating characteristic curves that illustrate the predictive ability of the expression of each of the depicted ligand(s) to discriminate whether primary AML samples are targeted by DOT cells. (F) Fold change of percentage of CD69+ cells (left) or CD69 MFI (right) on DOT cells that were cultured alone, or in the presence of PVR− or PVR+ primary AML samples. Each dot represents a different DOT cell/AML combination. Statistical analysis was performed using the Mann-Whitney U test for panels C-D, and Kruskal-Wallis followed by Dunn multiple comparisons test for panel E. AUC, area under the curve; w/o, without.
From a conceptual point of view, this study highlights the potential of NKRs to provide broad innate sensing mechanisms to T-cell products, such as DOT cells, for adoptive immunotherapy. This is especially relevant for diseases like AML in which it is difficult to define a safe CAR T-cell antigen, and even in B-cell malignancies, in which target antigen loss can drive relapses that limit the 1-year progression-free survival rates to 50%.16,17 Furthermore, the high DNAM-1 expression on DOT cells may be important to compensate for the decreased DNAM-1 expression in endogenous, and functionally impaired, NK cells of patients with AML.18 Furthermore, DOT cells display the key advantages over NK cells of lacking the expression of inhibitory killer cell immunoglobulin-like receptors5 and the additional activation via the TCR, allowing for high yields during in vitro expansion protocols.
In contrast, T-cell dysfunctionality or exhaustion has also been demonstrated in patients with relapsed AML,19 as well as in patients with B-cell leukemia/lymphoma treated with autologous CAR T cells.20 This favors allogeneic T cells, which are neither affected by cancer nor chemotherapy, and may be further boosted in vitro, alike DOT cells.5 Moreover, by being major histocompatibility/human leukocyte antigen independent, DOT cells are particularly well suited for allogeneic use, without need of genetic edition of the endogenous TCR, as for αβ T cells,21 to prevent graft-versus-host disease.22 Importantly, DOT cells are derived from peripheral blood Vδ1+ γδ T cells, which were shown to correlate with long-term disease-free survival of patients with leukemia after allogeneic hematopoietic stem cell transplantation,23 and were recently associated with a CD19 CAR T-cell expansion in a patient with chronic lymphocytic leukemia who was disease free 10 years after treatment.24 We therefore expect DOT cells to constitute a distinctive platform for adoptive immunotherapy of AML and potentially other hematological (and solid) malignancies.
Acknowledgments
The authors thank André Simões, Daniel V. Correia, Simão Rocha, Natacha Gonçalves-Sousa and the Flow Cytometry, Bioimaging and Rodent Facilities of Instituto de Medicina Molecular João Lobo Antunes for their technical support and assistance. The authors also thank the patients, and their families, for donating AML samples.
The authors acknowledge financing by national funds through Fundação para a Ciência e a Tecnologia, I.P. (PTDC/MED-ONC/6829/2020, DOI: 10.54499/PTDC/MED-ONC/6829/2020 to B.S.-S. and L.B.; and 2021.01953.CEECIND, DOI: 10.54499/2021.01953.CEECIND/CP1673/CT0007 to S.M.). The authors also acknowledge funding from Takeda Development Center Americas, Inc (B.S.-S., S.M., C.C., and R.B.-D.); “la Caixa” Foundation (ID 100010434, under agreement LCF/PR/HR19/52160011 [B.S.-S. and P.M.]); “Ayudas Merck de Investigación” from Health Merck Foundation; grant PID2022-136554OA-I00 funded by MICIU/AEI 10.13039/501100011033 and the European Regional Development Fund (ERDF)/EU (D.S.-M.); Centres de Recerca de Catalunya/Generalitat de Catalunya and Fundació Josep Carreras-Obra Social la Caixa for core support; the Spanish Ministry of Economy and Competitiveness/European Union NextGenerationEU (grants CPP2021-008508 and CPP2022-009759); and the scientific foundation of the Spanish Association Against Cancer (grants AECC,PRYGN234975MENE and PRYGN211192BUEN) to P.M.
Authorship
Contribution: S.M. and C.C. performed most of the experiments and analyzed the data; D.S.-M., S.S., R.M.L.C., N.T., and M.V. contributed to the generation of k cell lines; S.S., R.M.L.C., R.B.-D., and L.B. assisted in some experiments and provided critical feedback; B.G., N.C., and M.G.d.S. supplied primary AML samples; B.S.-S., S.M., and P.M. designed the study and wrote the manuscript; and D.S.-M. and R.B.-D. revised the manuscript.
Conflict-of-interest disclosure: B.S.-S., S.M., C.C., and R.B.-D. received funding from a sponsored research agreement with Takeda Development Center Americas, Inc, Lexington, MA. P.M. is cofounder of OneChain ImmunoTherapeutics, a spin-off from the Josep Carreras Leukemia Research Institute, Barcelona, Spain. The remaining authors declare no competing financial interests.
Correspondence: Sofia Mensurado, Instituto de Medicina Molecular João Lobo Antunes, Avenida Prof Egas Moniz, 1649-028 Lisbon, Portugal; email: sofiamensurado@medicina.ulisboa.pt; and Bruno Silva-Santos, Instituto de Medicina Molecular João Lobo Antunes, Avenida Prof Egas Moniz, 1649-028 Lisbon, Portugal; email: bssantos@medicina.ulisboa.pt.
References
Author notes
S.M. and C.C. contributed equally to this study.
P.M. and B.S.-S. contributed equally to this study.
Data are available on request from corresponding authors, Sofia Mensurado (sofiamensurado@medicina.ulisboa.pt) and Bruno Silva-Santos (bssantos@medicina.ulisboa.pt).
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.




This feature is available to Subscribers Only
Sign In or Create an Account Close Modal