Key Points
In patients with diffuse large B-cell lymphoma progressing after CAR-T therapy, odronextamab showed a 48% ORR and 32% CR rate.
Odronextamab had a generally manageable safety profile, with 48% low-grade cytokine release syndrome and 20% grade ≥3 infections.
Visual Abstract
Patients with relapsed/refractory diffuse large B-cell lymphoma progressing after chimeric antigen receptor T-cell (CAR-T) therapy have dismal outcomes. The prespecified post–CAR-T expansion cohort of the ELM-1 study investigated the efficacy and safety of odronextamab, a CD20×CD3 bispecific antibody, in patients with disease progression after CAR-Ts. Sixty patients received IV odronextamab weekly for 4 cycles followed by maintenance until progression. The primary end point was objective response rate (ORR) by independent central review. The median number of prior lines of therapy was 3 (range, 2-9), 71.7% were refractory to CAR-Ts, and 48.3% relapsed within 90 days of CAR-T therapy. After a median follow-up of 16.2 months, ORR and complete response (CR) rate were 48.3% and 31.7%, respectively. Responses were similar across prior CAR-T products and time to relapse on CAR-T therapy. Median duration of response was 14.8 months and median duration of CR was not reached. Median progression-free survival and overall survival were 4.8 and 10.2 months, respectively. The most common treatment-emergent adverse event was cytokine release syndrome (48.3%; no grade ≥3 events). No cases of immune effector cell–associated neurotoxicity syndrome were reported. Grade ≥3 infections occurred in 12 patients (20.0%), 2 of which were COVID-19. Odronextamab monotherapy demonstrated encouraging efficacy and generally manageable safety, supporting its potential as an off-the-shelf option for patients after CAR-T therapy. This trial was registered at www.clinicaltrials.gov as #NCT02290951.
Introduction
CD19-directed chimeric antigen receptor (CAR) T-cell (CAR-T) therapy is an established treatment option for relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL). Axicabtagene ciloleucel (axi-cel), tisagenlecleucel (tisa-cel), and lisocabtagene maraleucel (liso-cel) demonstrated clinically meaningful efficacy improvements in their pivotal studies with objective response rates (ORRs) of 52% to 82% and complete responses (CR) rates of 40% to 55%, leading to their approvals in the United States and Europe in the third-line or later setting.1-5 More recently, axi-cel and liso-cel received approval for use for patients who are refractory to, or relapse within 12 months of, first-line chemoimmunotherapy.
Despite the efficacy observed with CD19-targeting CAR-Ts, approximately half of patients with R/R DLBCL will relapse, after which median overall survival (OS) is 5 months.6,7 Furthermore, many patients do not receive subsequent treatment after CAR-Ts,6-8 highlighting a significant unmet medical need in this population.
One potential mechanism of resistance or relapse to CAR-Ts is CD19 antigen loss, which is observed in ∼30% of patients with DLBCL at the time of progression after CAR-T therapy.8-10 Treatment options targeting alternate B-cell antigens are thus an attractive option; however, there is no consensus on the standard of care in this population.
Odronextamab is an Fc-silenced, human CD20×CD3 bispecific antibody that induces T-cell–mediated cytotoxicity by simultaneously engaging CD20 on malignant B cells and CD3 on cytotoxic T cells.11 In the phase 1, dose-escalation/expansion ELM-1 study, odronextamab monotherapy demonstrated encouraging efficacy and generally manageable safety in heavily pretreated patients with R/R DLBCL and other B-cell malignancies.12 Here, we report results from the primary analysis of the dedicated post–CAR-T expansion cohort of ELM-1.
Methods
Study design and patients
ELM-1 is a phase 1, single-arm, multicenter, dose-escalation and dose-expansion study of odronextamab monotherapy for patients with R/R CD20+ B-cell non-Hodgkin lymphoma (ClinicalTrials.gov identifier: NCT02290951). Details of the ELM-1 study design and inclusion/exclusion criteria have been described previously.12
The prespecified post–CAR-T expansion cohort enrolled patients aged ≥18 years who had documented DLBCL with disease progression after CAR-Ts and had recovered from the toxicities of lymphodepletion therapy and CAR-T infusion. Prior CAR-T therapy was not required to be the most recent line of therapy before enrollment. Patients who had received at least 1 prior treatment with an anti-CD20 antibody, had an Eastern Cooperative Oncology Group performance status of 0 or 1, and adequate hematological and organ function were eligible. Patients with primary central nervous system (CNS) lymphoma or known or suspected CNS involvement by nonprimary CNS non-Hodgkin lymphoma were excluded. Measures to facilitate diverse and inclusive enrollment are in the supplemental Methods, available on the Blood website.
All patients provided written informed consent. The study protocol was approved by relevant institutional review boards and ethics committees at each site. The study was conducted in accordance with the International Conference on Harmonization good clinical practice guidelines and the Declaration of Helsinki.
Treatment
Patients received IV odronextamab in 21-day cycles, with premedication and cycle 1 (C1) step-up dosing (optimized from 1/20 mg to 0.7/4/20 mg during the study, as previously described12) to help mitigate the risk of cytokine release syndrome (CRS). Patients received odronextamab 160 mg on days 1, 8, and 15 of C2 to C4, then 320 mg once every 2 weeks until disease progression or unacceptable toxicity. Patients with a CR lasting ≥9 months transitioned to 320 mg dosing once every 4 weeks (further details on the dosing regimen are in the supplemental Methods).
Prophylaxis for Pneumocystis jirovecii pneumonia was recommended for all patients during the study. Antiviral prophylaxis was recommended for patients with a history of herpes simplex virus and cytomegalovirus infections, or with prior hepatitis B infection. For patients with hypogammaglobulinemia, IV immunoglobulin G was recommended according to local institutional guidelines (supplemental Methods).
End points and assessment
The primary analysis was performed when all patients had completed the 36-week tumor assessment or withdrawn from the study. The primary end point was ORR assessed by independent central review according to Lugano criteria.13 Secondary end points included duration of response (DOR), progression-free survival (PFS), OS, safety, pharmacokinetics, and immunogenicity. Biomarkers, including T-cell and B-cell dynamics and function, as well as cytokine profiles, were exploratory end points.
Radiologic assessment for efficacy was performed using computed tomography (CT) or magnetic resonance imaging (MRI; if CT was not feasible) at baseline, at the end of week 5 and week 12, then every 12 weeks up to the end of year 2, and every 24 weeks thereafter. Fluorodeoxyglucose positron emission tomography assessments were required at weeks 12, 24 (only if no complete metabolic response on prior scan), 36 at the end of year 2, and annually thereafter.
Safety and tolerability were assessed until 90 days after the last dose of study drug or initiation of another antilymphoma therapy. Adverse events (AEs) were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE; version 4.03), and CRS according to the American Society for Transplantation and Cellular Therapy criteria.14 The immune effector cell-associated encephalopathy (ICE) score was not used because the study began before the ICE score was established for grading immune effector cell–associated neurotoxicity syndrome (ICANS). Instead, neurological events were described according to the CTCAE terms. Anti-infection prophylaxis was recommended as part of a protocol amendment.
CRS and infection rates were analyzed using the CAR-HEMATOTOX (HT) model.15,16 Infection rates were analyzed by HT score for different periods: events occurring during the first 30, 60, and 90 days of treatment, and until end of study. CRS rates were analyzed by HT score for events occurring within the first 30 days.
Biomarker analysis
Tumor biopsies were obtained at baseline if clinically feasible. CD19/CD20 expression levels were analyzed using fully automated multiplex immunofluorescence assays on the Ventana Discovery ULTRA platform (Ventana Medical System), and CD20 expression was also assessed using fully automated chromogenic immunohistochemistry assays at a central laboratory (detailed methods have been described previously17). Peripheral immune cell populations were quantified using fresh whole blood by flow cytometry performed by CellCarta. To provide comparison of T-cell expansion and function in patients who did not receive prior CAR-T, immune cell counts from CAR-T–naïve patients enrolled in the ELM-2 R/R DLBCL cohort (ClinicalTrials.gov identifier: NCT03888105) were obtained. Cytokines in serum were quantified using the Luminex and Simoa platforms, performed by Rules-Based Medicine. Details on ex vivo assessments are in the supplemental Methods.
Statistical analysis
There were no formal statistical hypotheses in this study. A sample size of 60 patients was determined for the dose-expansion cohort on the basis that this would be adequate to observe an ORR that is significantly different to ≥35%. Duration of efficacy follow-up was calculated based on reverse Kaplan-Meier PFS (investigator assessment). ORR and CR rate were summarized with the 2-sided 95% confidence intervals (CIs) using the Clopper-Pearson methods. DOR, PFS, and OS were estimated using the Kaplan-Meier method. Data were analyzed using SAS version 9.4 (SAS Institute).
Results
From 17 May 2020 to 19 April 2023, 60 patients (20 on the 1/20 mg and 40 on the 0.7/4/20 mg step-up regimens) with R/R DLBCL progressing after CAR-T were enrolled and evaluable for efficacy and safety. At data cutoff (22 January 2024), 55 (91.7%) patients completed the first cycle of treatment, and 31 (51.7%) completed ≥4 treatment cycles. Five patients did not complete C1 (4 because of progressive disease and 1 because of an AE). Median duration of exposure to odronextamab was 12.07 weeks (range, 0.7-154.1) and 2 patients remained on treatment (supplemental Figure 1). The most common reasons for discontinuation were disease progression (55.0%), physician or patient decision (26.7%; 12 physician decisions, 4 patient decisions; of these, 5 patients proceeded to stem cell transplantation), and AEs (11.7%).
Four (6.7%) patients received cytomegalovirus prophylaxis, 58 (96.7%) received herpes simplex virus prophylaxis, 46 (76.7%) received Pneumocystitis jirovecii pneumonia prophylaxis, and 6 (10.0%) received IV immunoglobulin G prophylaxis.
Baseline characteristics
The median age was 63 years (range, 27-82; Table 1). Patients were heavily pretreated, with a median of 3 prior lines of therapy (range, 2-9); 58.3% were primary refractory, 76.7% were refractory to their most recent line of therapy, and 71.7% were refractory to CAR-Ts in any prior line.
Patient demographics and baseline characteristics
| . | All patients, N = 60 . |
|---|---|
| Age, median (range), y | 63 (27-82) |
| ≥65 y, n (%) | 27 (45.0) |
| ≥75 y, n (%) | 6 (10.0) |
| Male, n (%) | 39 (65.0) |
| Race, n (%) | |
| White | 46 (76.7) |
| Black or African American | 2 (3.3) |
| Asian | 5 (8.3) |
| American Indian or Alaska Native | 1 (1.7) |
| Other | 1 (1.7) |
| Unknown/not reported | 5 (8.3) |
| Ethnicity, n (%) | |
| Hispanic/Latino | 6 (10.0) |
| Not Hispanic/Latino | 52 (86.7) |
| Not reported | 2 (3.3) |
| Geographic region, n (%) | |
| North America | 44 (73.3) |
| Europe | 12 (20.0) |
| Rest of world | 4 (6.7) |
| ECOG PS, n (%) | |
| 0 | 14 (23.3) |
| 1 | 46 (76.7) |
| Ann Arbor stage III/IV, n (%) | 44 (73.3) |
| IPI 3-5, n (%) | 35 (58.3) |
| Bulky disease (investigator assessment), n (%) | 24 (40.0) |
| Prior lines of therapy, median (range), n | 3 (2-9) |
| ≥3 prior lines, n (%) | 51 (85.0) |
| ≥4 prior lines, n (%) | 27 (45.0) |
| ≥5 prior lines, n (%) | 19 (31.7) |
| Cell of origin, n (%) | |
| GCB | 25 (41.7) |
| Non-GCB | 21 (35.0) |
| Unknown | 14 (23.3) |
| Site-reported cytogenetic status, n (%) | |
| HGBL with MYC and BCL2/BCL6 rearrangements | 16 (26.7) |
| NOS | 21 (35.0) |
| Missing | 23 (38.3) |
| Tumor burden, n (%) | |
| SPD low | 28 (46.7) |
| SPD high | 29 (48.3) |
| Missing | 3 (5.0) |
| HT score, n (%) | |
| 0-1 (HTlow) | 16 (26.7) |
| ≥2 (HThigh) | 28 (46.7) |
| Missing | 16 (26.7) |
| Prior CAR-T therapy, n (%) | |
| Axi-cel | 36 (60.0) |
| Tisa-cel | 10 (16.7) |
| Liso-cel | 6 (10.0) |
| Investigational CD19-directed CAR-Ts | 8 (13.3) |
| CAR-Ts as most recent line of therapy | 43 (71.7) |
| Best response to CAR-Ts, n (%) | |
| CR | 21 (35.0) |
| PR | 12 (20.0) |
| SD | 5 (8.3) |
| PD | 22 (36.7) |
| Time since prior CAR-T therapy, median, mo | 6.5 |
| Time to CAR-T relapse, n (%) | |
| ≤30 d | 4 (6.7) |
| ≤90 d | 29 (48.3) |
| ≤180 d | 40 (66.7) |
| R/R subgroup, n (%) | |
| Primary refractory to first-line therapy | 35 (58.3) |
| Refractory to most recent line of therapy | 46 (76.7) |
| Refractory to CAR-T therapy in any line | 43 (71.7) |
| . | All patients, N = 60 . |
|---|---|
| Age, median (range), y | 63 (27-82) |
| ≥65 y, n (%) | 27 (45.0) |
| ≥75 y, n (%) | 6 (10.0) |
| Male, n (%) | 39 (65.0) |
| Race, n (%) | |
| White | 46 (76.7) |
| Black or African American | 2 (3.3) |
| Asian | 5 (8.3) |
| American Indian or Alaska Native | 1 (1.7) |
| Other | 1 (1.7) |
| Unknown/not reported | 5 (8.3) |
| Ethnicity, n (%) | |
| Hispanic/Latino | 6 (10.0) |
| Not Hispanic/Latino | 52 (86.7) |
| Not reported | 2 (3.3) |
| Geographic region, n (%) | |
| North America | 44 (73.3) |
| Europe | 12 (20.0) |
| Rest of world | 4 (6.7) |
| ECOG PS, n (%) | |
| 0 | 14 (23.3) |
| 1 | 46 (76.7) |
| Ann Arbor stage III/IV, n (%) | 44 (73.3) |
| IPI 3-5, n (%) | 35 (58.3) |
| Bulky disease (investigator assessment), n (%) | 24 (40.0) |
| Prior lines of therapy, median (range), n | 3 (2-9) |
| ≥3 prior lines, n (%) | 51 (85.0) |
| ≥4 prior lines, n (%) | 27 (45.0) |
| ≥5 prior lines, n (%) | 19 (31.7) |
| Cell of origin, n (%) | |
| GCB | 25 (41.7) |
| Non-GCB | 21 (35.0) |
| Unknown | 14 (23.3) |
| Site-reported cytogenetic status, n (%) | |
| HGBL with MYC and BCL2/BCL6 rearrangements | 16 (26.7) |
| NOS | 21 (35.0) |
| Missing | 23 (38.3) |
| Tumor burden, n (%) | |
| SPD low | 28 (46.7) |
| SPD high | 29 (48.3) |
| Missing | 3 (5.0) |
| HT score, n (%) | |
| 0-1 (HTlow) | 16 (26.7) |
| ≥2 (HThigh) | 28 (46.7) |
| Missing | 16 (26.7) |
| Prior CAR-T therapy, n (%) | |
| Axi-cel | 36 (60.0) |
| Tisa-cel | 10 (16.7) |
| Liso-cel | 6 (10.0) |
| Investigational CD19-directed CAR-Ts | 8 (13.3) |
| CAR-Ts as most recent line of therapy | 43 (71.7) |
| Best response to CAR-Ts, n (%) | |
| CR | 21 (35.0) |
| PR | 12 (20.0) |
| SD | 5 (8.3) |
| PD | 22 (36.7) |
| Time since prior CAR-T therapy, median, mo | 6.5 |
| Time to CAR-T relapse, n (%) | |
| ≤30 d | 4 (6.7) |
| ≤90 d | 29 (48.3) |
| ≤180 d | 40 (66.7) |
| R/R subgroup, n (%) | |
| Primary refractory to first-line therapy | 35 (58.3) |
| Refractory to most recent line of therapy | 46 (76.7) |
| Refractory to CAR-T therapy in any line | 43 (71.7) |
Refractory was defined as disease progression during prior therapy or relapsed within 6 months from completion of any line of therapy. Relapse was defined as disease that recurred after a response lasting >6 months from completion of any line of therapy.
CD, cluster of differentiation; ECOG PS, Eastern Cooperative Oncology Group performance status; GCB, germinal center B cell; HGBL, high-grade B-cell lymphoma; HTlow, low HT score; IPI, International Prognostic Index; mo, months; NOS, not otherwise specified; SPD, sum of product diameter.
Prior CAR-T therapies included axi-cel (60.0%), tisa-cel (16.7%), liso-cel (10.0%), and investigational CD19-directed CAR-Ts (13.3%). CAR-T was the most recent line of therapy in 71.7% of patients. Time to relapse on CAR-T therapy was ≤90 days in 48.3% of patients and ≤180 days in 66.7% of patients. The median time from prior CAR-Ts to odronextamab treatment was 6.5 months (range, 1.2-46.2). The best ORR to CAR-Ts was 55.0% (33/60; CR, 35.0% [21/60]). Baseline HT scores were available in 44 patients; of these, 63.6% had a high HT score (HThigh).
Most patients had high-risk disease; 73.3% had Ann Arbor stage III/IV, 26.7% had high-grade B-cell lymphoma with MYC and BCL2 and/or BCL6 rearrangements, and the median sum of product diameter was 2430 mm2. Baseline characteristics were broadly similar between patients on the 1/20 mg and 0.7/4/20 mg regimens (supplemental Table 1).
Efficacy
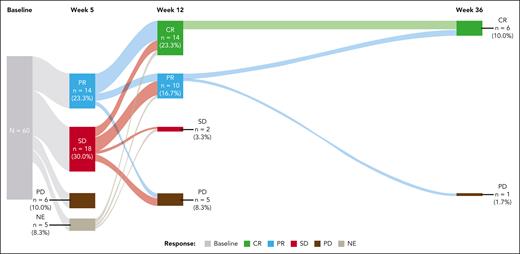
Median follow-up was 16.2 months (95% CI, 8.0-17.6) and median time to response was 1.8 months (range, 0.9-5.4). The initial postbaseline tumor assessment at week 5 using CT/MRI imaging demonstrated an ORR of 23.3% (14/60), all of which were partial responses (PRs). Subsequent tumor assessments at week 12, including fluorodeoxyglucose positron emission tomography scan and CT/MRI, demonstrated an ORR of 40.0% (24/60) and a CR rate of 23.3% (14/60). Among patients who achieved a PR at week 5 (n = 14), the majority had deeper response or maintained response at week 12 (57.1% [n = 8] achieved a CR and 21.4% [n = 3] remained at PR; Figure 1). The best ORR by independent central review was 48.3% (29/60; 95% CI, 35.2-61.6) and CR rate was 31.7% (19/60; 95% CI, 20.3-45.0; Table 2). Responses were similar in patients who received the 1/20 mg and 0.7/4/20 mg regimens, with overlapping CIs for ORR, CR, and PR (supplemental Table 2).
Sankey plot of responses by independent central review at weeks 5, 12, and 36. Deeper responses were observed at week 12, with 8 of 14 patients with PR at week 5 achieving CR, and 3 of 14 maintaining PR; 4 of 18 patients with SD at week 5 achieving CR and 6 of 18 achieving PR. Additionally, 2 patients with PR at week 12 achieved CR at week 36.
Sankey plot of responses by independent central review at weeks 5, 12, and 36. Deeper responses were observed at week 12, with 8 of 14 patients with PR at week 5 achieving CR, and 3 of 14 maintaining PR; 4 of 18 patients with SD at week 5 achieving CR and 6 of 18 achieving PR. Additionally, 2 patients with PR at week 12 achieved CR at week 36.
Best overall response per independent central review in patients with response assessment at 12 weeks
| n (%) . | All patients, N = 60 . |
|---|---|
| ORR | 29 (48.3) (95% CI, 35.2-61.6) |
| CR | 19 (31.7) (95% CI, 20.3-45.0) |
| PR | 10 (16.7) (95% CI, 8.3-28.5) |
| SD | 9 (15.0) |
| n (%) . | All patients, N = 60 . |
|---|---|
| ORR | 29 (48.3) (95% CI, 35.2-61.6) |
| CR | 19 (31.7) (95% CI, 20.3-45.0) |
| PR | 10 (16.7) (95% CI, 8.3-28.5) |
| SD | 9 (15.0) |
Response rates were generally similar across high-risk subgroups and across different prior CAR-T products. Responses were observed in patients who were refractory to prior CAR-Ts (ORR, 34.9% [15/43]; CR, 18.6% [8/43]; supplemental Figure 2). Responses in patients with stable disease (SD)/progressive disease (PD) to prior CAR-Ts, and those relapsing within 90 days of CAR-T therapy, were as follows: SD: ORR and CR, 20.0% (1/5); PD: ORR, 27.3% (6/22); CR, 13.6% (3/22); relapsed within 90 days of CAR-Ts: ORR, 20.7% (6/29); CR, 10.3% (3/29; supplemental Figure 3). Patients who relapsed within 90 days of CAR-Ts had a lower ORR than those who relapsed after >90 days (ORR, 63.6%-84.6%). Similarly, patients with SD/PD on prior CAR-T therapy had a lower ORR (20.0%-27.3%) than those with CR/PR on prior CAR-T therapy (ORR, 50.0%-76.2%).
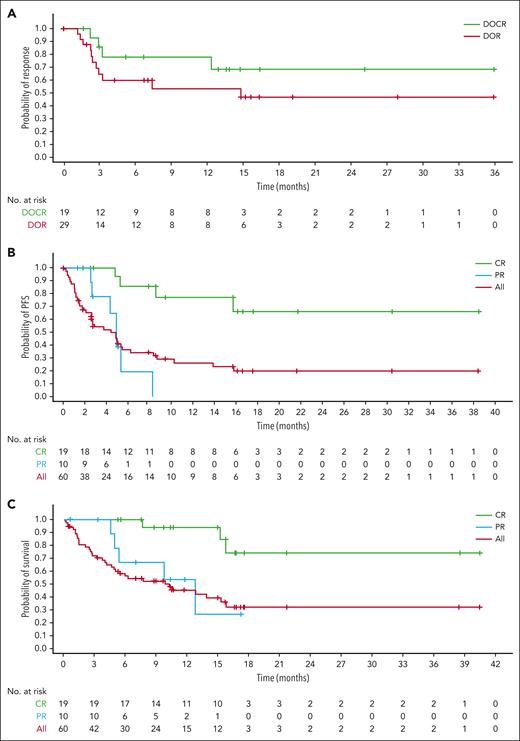
Median DOR was 14.8 months (95% CI, 2.8 to not estimable [NE]) and median duration of CR was not reached (NR; 95% CI, 3.3 to NE; Figure 2). Median DOR in patients who discontinued treatment for reasons other than disease progression or death (n = 23) was 3.7 months (range, 0.5-35.9), and 16 patients had ongoing responses at the time of discontinuation. Three patients who maintained a CR for 9 months transitioned to every-4-week dosing and responses were maintained throughout the study period.
Time-to-event outcomes. DOR and DOCR by independent central review (ICR) (A), PFS by response by ICR (B), and OS by response (C). DOCR, duration of complete response.
Time-to-event outcomes. DOR and DOCR by independent central review (ICR) (A), PFS by response by ICR (B), and OS by response (C). DOCR, duration of complete response.
Median PFS (mPFS) was 4.8 months (95% CI, 2.6-5.4) with a 12-month PFS rate of 26.5% (95% CI, 14.6-39.9). Median OS (mOS) was 10.2 months (95% CI, 4.6-15.8) with a 12-month OS rate of 45.5% (95% CI, 31.7-58.3). Patients with a CR had longer mPFS and mOS than those with a PR (mPFS: NR [95% CI, 8.6 to NE] vs 5.0 months [95% CI, 2.6 to NE]; hazard ratio, 0.07 [95% CI, 0.01-0.37]; mOS: NR [95% CI, 15.2 to NE] vs 12.8 months [95% CI, 4.6 to NE]; hazard ratio, 0.15 [95% CI, 0.03-0.67]; Figure 2). The 12-month PFS and OS rates in patients with CR were 77.1% (95% CI, 44.2-92.1) and 93.8% (95% CI, 63.2-99.1), respectively.
The 12-month PFS and OS in patients relapsing within 90 days of prior CAR-Ts (12-month PFS and OS: 10.3% and 20.7%, respectively) were shorter than those relapsing in >90 days (relapsed in 91-180 days: NE and 58.3%; relapsed in 181 days to 1 year: 54.8% and 84.6%; relapsed in >1 year: 50.0% and 66.7%, respectively).
Safety
All 60 patients had at least 1 treatment-emergent AE (TEAE) and 90.0% had at least 1 treatment-related AE (Table 3). Grade ≥3 TEAEs occurred in 76.7% of patients and the most common (≥10%) were neutropenia (16.7%), anemia (13.3%), neutrophil count decreased (10.0%), and white blood cell count decreased (10.0%). Treatment-related TEAEs led to treatment discontinuation in 8.3% of patients (COVID-19, pneumonia, encephalopathy, epilepsy, and gait disturbance [n = 1 each]). Grade 5 AEs occurred in 2 (3.3%) patients: 1 event of brain herniation on day 140 that occurred in the setting of hyperammonemia and ongoing COVID-19 infection, considered unrelated to treatment; and 1 event of COVID-19 pneumonia on day 35 of the study, considered treatment related.
Safety summary and TEAE profile
| n (%) . | All patients, N = 60 . |
|---|---|
| Patients with any TEAE | 60 (100.0) |
| Patients with any treatment-related TEAE | 54 (90.0) |
| TEAEs occurring in ≥15% of all patients | |
| CRS | 29 (48.3) |
| Fatigue | 21 (35.0) |
| Pyrexia | 19 (31.7) |
| Anemia | 18 (30.0) |
| Hypotension | 15 (25.0) |
| Cough | 15 (25.0) |
| Thrombocytopenia | 14 (23.3) |
| Diarrhea | 13 (21.7) |
| Nausea | 12 (20.0) |
| Hypophosphatemia | 11 (18.3) |
| Neutropenia | 11 (18.3) |
| Chills | 10 (16.7) |
| Insomnia | 10 (16.7) |
| Neutrophil count decreased | 10 (16.7) |
| Decreased appetite | 10 (16.7) |
| Peripheral edema | 9 (15.0) |
| Hypomagnesemia | 9 (15.0) |
| Abdominal pain | 9 (15.0) |
| Dyspnea | 9 (15.0) |
| Grade ≥3 TEAE | 46 (76.7) |
| Treatment-related grade ≥3 TEAE | 31 (51.7) |
| Serious TEAE | 33 (55.0) |
| Treatment-related serious TEAE | 24 (40.0) |
| TEAE leading to treatment discontinuation | 8 (13.3) |
| Treatment-related TEAE leading to treatment discontinuation | 5 (8.3) |
| TEAE leading to dose interruption/delay | 35 (58.3) |
| Treatment-related TEAE leading to dose interruption/delay | 28 (46.7) |
| TEAE leading to death | 2 (3.3) |
| Treatment-related TEAE leading to death | 1 (1.7) |
| n (%) . | All patients, N = 60 . |
|---|---|
| Patients with any TEAE | 60 (100.0) |
| Patients with any treatment-related TEAE | 54 (90.0) |
| TEAEs occurring in ≥15% of all patients | |
| CRS | 29 (48.3) |
| Fatigue | 21 (35.0) |
| Pyrexia | 19 (31.7) |
| Anemia | 18 (30.0) |
| Hypotension | 15 (25.0) |
| Cough | 15 (25.0) |
| Thrombocytopenia | 14 (23.3) |
| Diarrhea | 13 (21.7) |
| Nausea | 12 (20.0) |
| Hypophosphatemia | 11 (18.3) |
| Neutropenia | 11 (18.3) |
| Chills | 10 (16.7) |
| Insomnia | 10 (16.7) |
| Neutrophil count decreased | 10 (16.7) |
| Decreased appetite | 10 (16.7) |
| Peripheral edema | 9 (15.0) |
| Hypomagnesemia | 9 (15.0) |
| Abdominal pain | 9 (15.0) |
| Dyspnea | 9 (15.0) |
| Grade ≥3 TEAE | 46 (76.7) |
| Treatment-related grade ≥3 TEAE | 31 (51.7) |
| Serious TEAE | 33 (55.0) |
| Treatment-related serious TEAE | 24 (40.0) |
| TEAE leading to treatment discontinuation | 8 (13.3) |
| Treatment-related TEAE leading to treatment discontinuation | 5 (8.3) |
| TEAE leading to dose interruption/delay | 35 (58.3) |
| Treatment-related TEAE leading to dose interruption/delay | 28 (46.7) |
| TEAE leading to death | 2 (3.3) |
| Treatment-related TEAE leading to death | 1 (1.7) |
Treatment-related TEAEs led to hospitalization in 5 patients (device-related infection, pneumonia, encephalopathy, epilepsy, and acute pancreatitis; n = 1 each).
CRS was the most commonly reported TEAE (48.3%), and all CRS events were low grade (grade 1: 25.0%; grade 2: 23.3%; supplemental Table 3). The median time to onset of CRS was 17.7 hours (range, −2.4 to 143.5), with a median duration of 2 days (range, 1-8). CRS was treated with tocilizumab in 21.7% of patients and with corticosteroids in 16.7% of patients. Rates of any grade CRS were generally similar regardless of prior CAR-T product used (41.7% [axi-cel]; 50.0% [liso-cel]; and 75.0% [tisa-cel]), and regardless of time to relapse on CAR-T therapy (48.3% [≤90 days]; 54.5% [<90 to ≤180 days]; and 42.1% [>180 days]).
The rate of grade 2 CRS was higher in patients with HThigh than in those with low CAR-HEMATOTOX score (HTlow; 28.6% vs 6.3%). There was no association between baseline T-cell counts and markers of T-cell activation and rates of CRS (supplemental Figure 4).
Any-grade neurologic AEs occurred in 30 (50.0%) patients, the most common were headache (13.3%), anxiety (10.0%), and encephalopathy (8.3%). No cases of ICANS, as measured by the CTCAE, were reported. ICE score was not systemically collected, but details on 8 (13.3%) patients who experienced grade 3 neurologic AEs are as follows: 4 cases of encephalopathy on days 30, 97, 138, and 217 of the study, respectively; aphasia on day 58; transient ischemic attack on day 77; peripheral motor neuropathy on day 8; and cognitive disorder on day 12 (n = 1 each). Each case was accompanied by confounding factors (supplemental Results) and no grade 5 events were reported. No cases of tumor pain or tumor lysis syndrome were reported.
Infections occurred in 50.0% of patients, with 20.0% experiencing grade ≥3 infections. Infections led to treatment discontinuation in 5.0% (n = 3) of patients (COVID-19, device-related infection, and pneumonia [n = 1 each]). Infections by pathogen are provided in supplemental Table 4. The rate of grade ≥3 infections was 21.4% in patients with HThigh and 18.8% in those with HTlow during the entire study period. Compared with patients with HTlow, patients with HThigh had higher rates of grade ≥3 infections within 30, 60, and 90 days of the first dose (supplemental Table 5). Grade 5 COVID-19 pneumonia occurred in 1 (1.7%) patient and was considered related to treatment.
Pharmacokinetics and immunogenicity
Pharmacokinetic exposure parameters of odronextamab in the post–CAR-T cohort over the treatment period are shown in supplemental Table 6. Odronextamab exposure observed in this study was similar to that in CAR-T–naïve patients from the ELM-2 study over a period of 12 weeks. No anti-odronextamab antibodies were identified.
Biomarker analysis
Of 21 patients with baseline tumor biopsies available for multiplex immunofluorescence, 10 were CD19− (supplemental Figure 5A). The proportion of CD20+ cells among total cells in baseline biopsies was assessed using chromogenic immunohistochemistry, which demonstrated that 24 of 31 patients had >20% CD20+ cells (CD20-high patients) and 7 of 31 had ≤20% CD20+ cells (CD20-low patients). Objective responses were observed in patients regardless of baseline CD20 expression level (ORR in CD20-high vs CD20-low patients: 50.0% [95% CI, 29.1-70.9] vs 57.1% [95% CI, 18.4-90.1]; supplemental Figure 5B). Peripheral CD4/CD8 T-cell counts were obtained from 42 patients at baseline. Compared with CAR-T–naïve patients from the ELM-2 study, patients who had received CAR-Ts in this study had lower baseline T-cell counts (supplemental Figure 6A). Baseline T-cell counts were not associated with time since last CAR-T therapy (supplemental Figure 7).
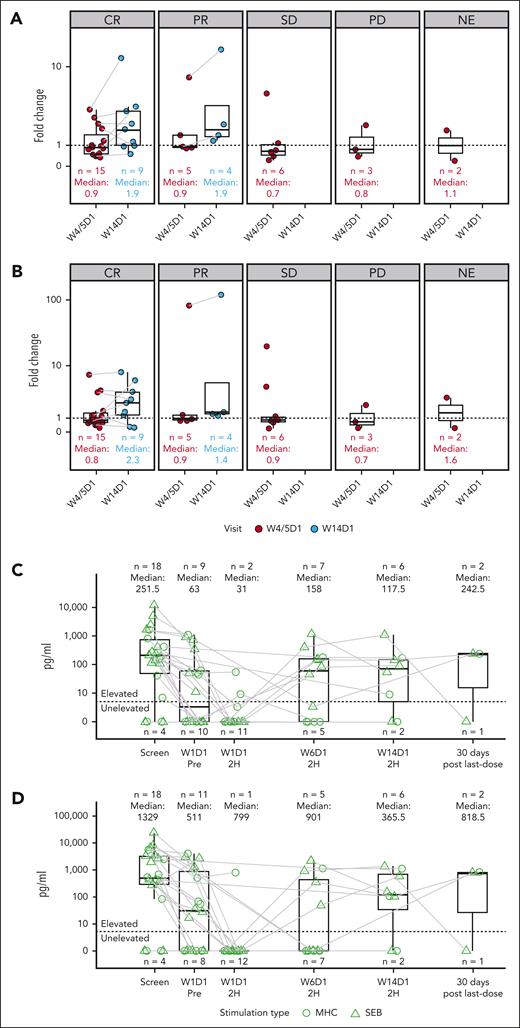
After repeated postdose decreases of CD4/CD8 T-cell counts during the step-up dosing period, a robust increase in CD4/CD8 T-cell counts was observed at week 14 (Figure 3A-B), with similar T-cell expansion observed in post–CAR-T and CAR-T–naïve patients (supplemental Figure 6B). This initial effect during step-up dosing was accompanied by a temporary increase in CD69 and HLA-DR–activated CD8 T cells at week 1 day 3, whereas inducible costimulator–positive CD8 T cells remained low. Programmed cell death protein 1–positive CD8 T cells initially increased but returned to baseline level by week 14 (supplemental Figure 8), coinciding with early T-cell activation.
T-cell counts and fitness on odronextamab treatment. Fold changes in CD4 T-cell (A) and CD8 T-cell (B) counts from baseline to week 4/5 and week 14 by best overall response. Changes in IFN-γ (C) and TNF-α (D) upon ex vivo stimulation. 2H, 2 hours; D, day; NA, not available; W, week.
T-cell counts and fitness on odronextamab treatment. Fold changes in CD4 T-cell (A) and CD8 T-cell (B) counts from baseline to week 4/5 and week 14 by best overall response. Changes in IFN-γ (C) and TNF-α (D) upon ex vivo stimulation. 2H, 2 hours; D, day; NA, not available; W, week.
Peripheral blood samples were collected for ex vivo stimulation as a measure of T-cell fitness. Analysis of baseline samples demonstrated adequate T-cell fitness at screening, with 18 of 22 samples showing cytokine release, including interferon gamma (IFN-γ) and tumor necrosis factor α (TNF-α), after stimulation. A decrease in cytokine release corresponding to dexamethasone administration was observed, with only 9 of 19 samples releasing IFN-γ before the first dose of odronextamab at week 1 day 1, and only 2 of 13 samples showing cytokine release at 2 hours after infusion. T-cell fitness returned to baseline level at week 14, with most samples collected 2 hours after odronextamab infusion showing cytokine release upon stimulation (Figure 3C-D).
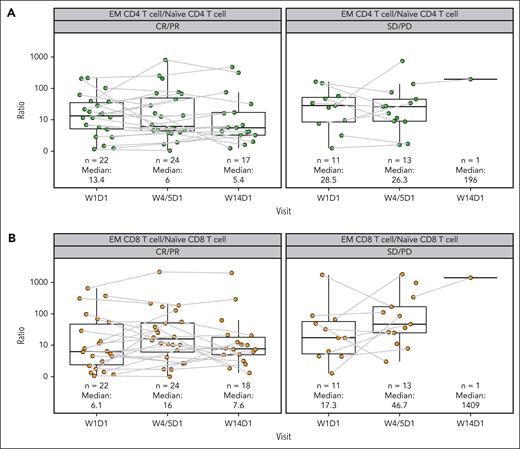
The percentage of naïve CD4 and CD8 T cells doubled from baseline to week 14, whereas the percentage of CD4 and CD8 effector memory T cells (TEM) remained stable (supplemental Figure 9). The median ratio of TEM/naïve T cells trended lower at baseline and on treatment in patients with CR/PR than in those with SD/PD (TEM/naïve CD8 T cells at week 4/5 on treatment in CR/PR vs SD/PD: 16 vs 46.7; Figure 4).
Ratio of TEM to naïve T cells at baseline and on treatment by best overall response. Ratio of TEM CD4 to naïve CD4 T cells (A) and TEM CD8 to naïve CD8 T cells (B).
Ratio of TEM to naïve T cells at baseline and on treatment by best overall response. Ratio of TEM CD4 to naïve CD4 T cells (A) and TEM CD8 to naïve CD8 T cells (B).
No association between clinical CRS rates and T-cell counts, inducible costimulator–positive CD8 T cells, or programmed cell death protein 1–positive CD8 T cells at baseline was observed. Baseline IFN-γ and TNF-α levels were generally low and not predictive of clinical CRS. Peak cytokine levels measured on odronextamab treatment showed a population-level association between clinical CRS and peak levels of IFN-γ and TNF-α (supplemental Figure 4E-F).
Discussion
This is, to our knowledge, the largest study to prospectively evaluate the use of a CD20×CD3 bispecific antibody in R/R DLBCL after CAR-T therapy in a dedicated expansion cohort. Odronextamab demonstrated encouraging responses (CR: 31.7%; median DOR: 14.8 months) with similar response rates observed irrespective of the CAR-T product used. Responses were seen early (week 5) and deepened by week 12. Responses were observed in patients with disease refractory to prior CAR-Ts (ORR, 34.9%) and in those with relapse within 90 days of CAR-Ts (ORR, 20.7%), highlighting the potential for odronextamab to address a significant unmet medical need.
Almost half of all patients enrolled in this study had disease progression within 3 months of CAR-Ts, and two-thirds progressed within 6 months, indicating a highly refractory patient population with a significant unmet need. Odronextamab demonstrated clinically meaningful responses across subgroups with a trend for improved rates in patients who relapsed after 180 days on CAR-Ts. Median OS with odronextamab in the overall post–CAR-T population was 10.2 months, with a 12-month OS rate of 45.5%. This is particularly notable in a setting in which the historical median OS is 5 months and the 12-month OS rate is 26.9%.6,7 Additionally, odronextamab demonstrated clinically important long-term outcomes in patients who achieved a CR, with a 12-month CR rate of 53.2%, and 12-month PFS and OS rates of 77.1% and 93.8%, respectively.
Although cross-study comparisons should be interpreted with caution as potential differences in population and study design have not been accounted for, the CR rate in the subgroup analysis of post–CAR-T patients treated with glofitamab was 35% and with epcoritamab was 34%.18,19 In the DLBCL cohort of the BICAR study (n = 44), post–CAR-T patients receiving glofitamab had a complete metabolic response of 36.4%.20
Odronextamab administration was feasible, with 91.7% of patients completing C1, 51.7% completing C4, and a low discontinuation rate (8.3%) because of treatment-related TEAEs. Although odronextamab administration included 3 step-up doses and split dosing, it resulted in a lower incidence of grade 3 CRS than that of other bispecific antibody regimens with 2 step-up doses for DLBCL, while also exhibiting a manageable safety profile that permits administration with close monitoring without the need for mandatory hospitalization.18,19,21
In this study, CRS events occurred in 48.3% of patients treated with odronextamab, and all were grade 1 or 2. HT scores at baseline appeared to predict the risk of CRS, with patients with HThigh showing higher rates of grade 2 CRS than those with HTlow. Elevated baseline levels of inflammatory markers such as C-reactive protein and ferritin may suggest a proinflammatory state, potentially predisposing patients to CRS. Reduction in cytokine release observed with ex vivo stimulation of samples from patients pretreated with dexamethasone further supported this hypothesis; however, additional studies are warranted.
No ICANS events were reported with odronextamab; however, ICANS has been reported with glofitamab (7.8%; CTCAE criteria) and epcoritamab (6.4%; American Society for Transplantation and Cellular Therapy criteria).18,19 Neurologic events reported in this study mostly occurred at day 30 or later, whereas ICANS events typically occur early in the treatment period.22-24 The 4 grade 3 encephalopathy events observed in this study also occurred much later in the treatment period and were confounded by other etiologies.
Infections are common among patients receiving CAR-Ts and have been identified as the leading cause of nonrelapse mortality in this population.25-29 Grade ≥3 infections occurred in 20.0% of patients receiving odronextamab. The rate of grade ≥3 infections reported with glofitamab was 14.9% and with epcoritamab was 14.6% in studies enrolling CAR-T–naïve and post–CAR-T patients.18,19 We observed higher rates of grade ≥3 infections in patients with HThigh occurring within ≤90 days, which is similar to the findings with CAR-T therapies.15 This suggests that HT scoring at baseline may potentially predict the risk of severe infections with bispecific antibodies in DLBCL, although prospective studies are needed.
Nonrelapse mortality due to infection has been reported in ∼3% of patients treated with CAR-Ts within 1 year,26,27 and this rate increased to 7.6% within 5 years.28,29 The rate of fatal infections with odronextamab in this study was 1.7%, suggesting nonrelapse mortality due to infection was similar despite patients having been exposed to CAR-Ts and a bispecific antibody. The rate of fatal infections with epcoritamab was 1.3% and with glofitamab was 4.5%; however, rates in the post–CAR-T subgroups have not been disclosed.18,19 These observations provide potential support for the use of anti-infection prophylaxis and HT scoring to help improve patient outcomes.
As expected in the post–CAR-T setting, CD19 loss was observed in 50% of patients at baseline, although the number of patients with available tumor biopsies was low. Odronextamab demonstrated similar responses regardless of baseline CD20 expression, which is consistent with results observed in ELM-2 in patients with R/R DLBCL and R/R follicular lymphoma.30 In contrast, lower activity in patients with a low CD20 expression level has been reported with glofitamab.31,32
The use of odronextamab after CAR-Ts did not appear to have a negative effect on T-cell count and fitness in this study. T-cell proliferation was similar in the post–CAR-T population in this study and in the CAR-T–naïve population from the ELM-2 study. We observed a typical pattern of T-cell dynamics seen with other T-cell engagers, in which T-cell counts decreased at the start of treatment, followed by an increase during the treatment period.33-35 No changes in T-cell fitness with ongoing odronextamab treatment were found, with functional T-cells observed at week 14. Findings from the ex vivo stimulation further confirmed the maintenance of T-cell fitness throughout treatment, as shown by the recovery of cytokine-releasing capability at weeks 6 and 14. Studies of the long-term effect of odronextamab on B-cell and T-cell counts and fitness are ongoing.
In conclusion, odronextamab monotherapy demonstrated an ORR of 48.3% and a CR rate of 31.7%, with a generally manageable safety profile in patients with difficult-to-treat, highly refractory R/R DLBCL after prior CAR-T therapy. Responses were durable, regardless of time since prior CAR-Ts, and T-cell fitness was maintained during odronextamab treatment. Given the lack of treatment options in this patient population, the results of this study support the potential use of odronextamab as an off-the-shelf treatment option after CAR-T therapy.
Acknowledgments
The authors thank the patients, their families, the ELM-1 study team, all other investigators, and all investigational site members involved in this study, especially during the challenging times of the COVID-19 pandemic. Medical writing support was provided by Ida Darmawan and Claire Stoker of Oberon, OPEN Health Communications, and funded by Regeneron Pharmaceuticals, Inc., in accordance with Good Publication Practice guidelines.
Authorship
Contribution: M.S.T., M.M., J.N.A., S.M.A., J.A.B., J.E.A., J.-M.M., N.G., S.M.O., U.A., I.A., and J.L.C. participated in the acquisition of clinical data from their study sites; Y.C. performed clinical data analysis; J.B.-V. oversaw biomarker strategy implementation, sample collection, and analysis; M.Z. designed pharmacokinetic assessment schedule, oversaw PK sample collection, and performed pharmacokinetic analysis; A.C., H.M., S.A., and D.M.F. participated in the design of the study, including defining patient selection criteria, response definitions, follow-up methodology, and data collection; and all authors contributed to the development of the first and subsequent drafts, and approved the final submission draft of the manuscript.
Conflict-of-interest disclosure: M.S.T. reports consultancy for AbbVie, AstraZeneca, BeiGene, Genmab, Incyte, Janssen, and Kite; and research funding from AstraZeneca, Kite, Regeneron Pharmaceuticals, Inc., and Roche. M.M. reports consultancy for Bayer, Genentech, GM Biosciences, Johnson & Johnson, Pharmacyclics, Roche, and Seattle Genetics; research funding from Bayer, Genentech, GM Biosciences, ImmunoVaccine Technologies, Johnson & Johnson, Pharmacyclics, Roche, and Seattle Genetics; honoraria from ADC Therapeutics, AstraZeneca, Bayer, Bristol Myers Squibb, Celgene, Epizyme, Genentech, IMV Therapeutics, Johnson & Johnson, Kite, Pharmacyclics, Regeneron Pharmaceuticals, Inc., Roche, Pfizer, Seattle Genetics, and Takeda; and membership on an entity’s board of directors or advisory committees for Allogene, Genentech, Genmab, and Merck. J.N.A. reports consultancy for AbbVie, Adaptive Biotechnologies, ADC Therapeutics, AstraZeneca, BeiGene, Genentech, Janssen, Eli Lilly, NeoGenomics, and Pharmacyclics; and research funding from BeiGene, Celgene/Bristol Myers Squibb, Genentech, and Janssen. S.M.A. reports trial research funding from ADC Therapeutics, Affimed, AstraZeneca, Bristol Myers Squibb, Pfizer, Regeneron Pharmaceuticals, Inc., and Takeda. J.E.A. reports speaker’s fee from Regeneron Pharmaceuticals, Inc. J.-M.M. reports consultancy for Ideogen, GlaxoSmithKline, Gilead, Merck, Regeneron Pharmaceuticals, Inc., and Therakos/Mallinckrodt; research funding from Astex Pharmaceuticals; and travel support from Beigene, Bristol Myers Squibb, GlaxoSmithKline, and Regeneron Pharmaceuticals Inc. S.M.O. reports consultancy for AbbVie, AstraZeneca, Autolus, BeiGene, Bristol Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen Oncology, Johnson & Johnson, Loxo Oncology, Merck, Pfizer, and Pharmacyclics; and research funding from Alliance, Caribou Biosciences, Nurix Therapeutics, and Regeneron Pharmaceuticals, Inc. Y.C., D.M.F., M.Z., J.B.-V., A.C., H.M., and S.A. hold stock or stock options for, and are employees of, Regeneron Pharmaceuticals, Inc. J.L.C. reports consultancy for Genmab, Incyte, Karyopharm Therapeutics, Kite Pharma, MorphoSys, Regeneron Pharmaceuticals, Inc., and Seagen; funding from AbbVie, Bayer, Genentech/Roche, and Merck; and employment with the Dana-Farber Cancer Institute. The remaining authors declare no competing financial interests.
Correspondence: Max S. Topp, Medizinische Klinik und Poliklinik 2, Universitätsklinikum Würzburg, Oberdürrbacher Str 6-8, 97080 Würzburg, Germany; email: topp_m@ukw.de.
References
Author notes
Presented in abstract form at the 66th annual meeting of the American Society of Hematology, San Diego, CA, 7 to 10 December 2024.
Qualified researchers may request access to study documents (including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan) that support the methods and findings reported in this manuscript. Individual anonymized participant data will be considered for sharing (1) once the product and indication have been approved by major health authorities (eg, US Food and Drug Administration, European Medicines Agency, Pharmaceuticals and Medical Devices Agency), or development of the product has been discontinued globally for all indications on or after April 2020 and there are no plans for future development; (2) if there is legal authority to share the data; and (3) if there is not a reasonable likelihood of participant reidentification. Submit requests at https://vivli.org/.
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal