Abstract
Acute chest syndrome (ACS) is an important cause of morbidity and mortality in sickle cell disease (SCD). Previous studies reported conflicting pictures of ACS making therapeutic interventions difficult. The Cooperative Study of Sickle Cell Disease prospectively followed 3,751 patients enrolled from birth to 66 years of age for ACS. Data on presenting signs and symptoms, laboratory findings, and hospital course were collected. There were 1,722 ACS episodes in 939 patients. Young children (age 2 to 4 years) presented with fever and cough, a negative physical exam, and rarely had pain. Adults were often afebrile and complained of shortness of breath, chills, and severe pain. Upper lobe disease was more common in children; multilobe and lower lobe disease affected adults more often. Severe hypoxia occurred in 18% of adults tested and could not be predicted by examination or laboratory findings. Bacteremia was documented in 3.5% of episodes, but was strongly influenced by age (14% of infants and 1.8% of patients <10 years). ACS was most common in winter with children having the most striking increase. Transfusion was used less frequently, but earlier in children. Young children were hospitalized for 5.4 days versus 9 days for adults. Fifty percent of adults had a pain event in the 2 weeks preceding ACS and children were more likely to have febrile events. The death rate was four times higher in adults than in children. Fatal cases generally developed rapid pulmonary failure and one third were associated with bacteremia. Age has a striking effect on the clinical picture of ACS. In children, ACS was milder and more likely due to infection, whereas in adults ACS was severe, associated with pain and had a higher mortality rate.
ACUTE CHEST SYNDROME (ACS) is the second most common cause of hospitalization in patients with sickle cell disease (SCD) and is responsible for up to 25% of deaths.1-4 Repeated events have been associated with an increased risk of chronic lung disease and early death.1,5,6 Both infectious and noninfectious etiologies including infarction and fat embolism have been described.7-11 Targeting intervention appropriately is difficult due to the inability to identify high-risk patients early in the course of disease. Recent reports suggest the clinical presentation of ACS may be predictive of etiology and severity.7 Limited studies have found conflicting clinical pictures and suggest aggressive interventions may not be necessary for all patients.5,9,11,12 The Cooperative Study of Sickle Cell Disease has prospectively followed 3,751 patients. The incidence rates for ACS in this population have been previously reported.6 This report defines the clinical and laboratory features of 1,722 ACS episodes in 939 patients and examines whether specific presentations or risk factors are predictive of clinical outcome.
MATERIALS AND METHODS
The Cooperative Study of Sickle Cell Disease13 is a prospective study of the clinical course of SCD with 3,751 patients enrolled from birth to 66 years of age and followed for at least 1 month. Patients were followed at 23 centers from 1979 through 1988. Detailed information was available for 1,722 ACS episodes in 939 SCD patients before January 1987. These patients were followed for a range of 1 month to 8 years (mean, 5.6 years). The number of patients and episodes is less than the number previously reported6 because a less-detailed data form was introduced between July 1985 and December 1986. Episodes on this less detailed data form were not included in this analysis. A majority of patients (537 [57.2%]) experienced only one event during the study period. Of the 939 patients who experienced ACS, there were 738 hemoglobin (Hb) SS, 127 Hb SC, 50 Hb Sβo thalassemia, and 24 Hb Sβ+ thalassemia patients.
Baseline and steady-state studies.Diagnoses of SCD were assigned at the Center for Disease Control, Atlanta, GA, using Hb electrophoresis. Patients received regular laboratory and physical evaluation at their center including a comprehensive annual visit.
Acute event studies.A diagnosis of ACS was defined as a new pulmonary infiltrate on chest x-ray or a perfusion defect demonstrable on a lung radioisotope scan.6 ACS was detected by chest x-ray in 930 of the 939 episodes (99%) and using lung scan in the remaining nine episodes (1%). When a diagnosis of ACS was made, data forms for the event were used as previously described.13 ACS forms included data concerning daily physical exams, laboratory results (including complete blood counts, chest x-ray, and blood, sputum and pleural fluid cultures), room air arterial blood gas results in patients 20 years or older, and follow-up reporting.
Other reported clinical events included pain crises, febrile events, transfusions, anemic events, surgeries, and right upper quadrant syndrome. An associated event was defined as an event that occurred in the 2 weeks before or on the day of presentation with ACS.
Mortality.The case fatality rate was calculated as the number of deaths within 15 days of an event, divided by the number of events. Because some patients may have died of ACS without an ACS event form being completed, all deaths related to ACS were reviewed as previously described.1 14 From this review, 19 additional deaths were identified that were not originally reported as ACS deaths. The reasons these patients were not identified as ACS deaths included: (1) no chest x-ray due to rapid death (ACS confirmed on autopsy), (2) death at a hospital outside the study, and (3) ACS form not completed. These 19 deaths were added to both the numerator and denominator of the case fatality rate.
Statistical methods.Cochran-Mantel Haenszel tests (dichotomous variables) or analysis of variance (ANOVA) (continuous variables) were used to test for differences between Hb SS and Hb SC subjects, controlling for age group, and for differences between 2- to 4-year-olds and 20+-year-olds, controlling for Hb SS or Hb SC.
Summary data for presenting symptoms and clinical signs are presented for the first ACS event for all genotypes combined. Lab values are presented separately for Hb SS and Hb SC patients for the first ACS event per patient. Means are presented for continuous variables, and proportions are presented for dichotomous variables.
Fisher's exact test was used to test for differences between proportions, and a t-test was used to test for differences between means. For subjects with two or more ACS events, logistic regression (SAS, PROC logistic, SAS computer program, SAS Institute, Cary, NC)15 was used to assess the relationship between the occurrence of symptoms on the first event and the second event, adjusting for age.
The seasonal variation analysis was based on the entire follow-up period for all subjects who had at least one ACS event. The ACS rate was then expressed as the number of events per 30 days. Poisson confidence intervals were used to test for differences between ACS rates.
RESULTS
Presenting symptoms.The most common presenting symptoms were fever, cough, and chest pain (Table 1). While less common, many patients experienced shortness of breath, wheezing, hemoptysis, chills, and productive cough. The frequency of presenting symptoms was age-dependent with fever and cough being more common in young children (age 2 to 4 years) and the incidence of chest pain, shortness of breath, chills, productive cough, and hemoptysis increasing with age. There were no significant differences in frequency of presenting symptoms between Hb SS and Hb SC patients except for severe pain, which was more common in Hb SS patients (P < .001).
Presenting Symptoms of Patients With ACS According to Age
| . | Total . | Age (Yr) . | P Value* . | ||||
|---|---|---|---|---|---|---|---|
| . | . | <2 . | 2-4 . | 5-9 . | 10-19 . | 20+ . | . |
| Symptoms . | . | % of Patients . | |||||
| . | . | . | . | . | . | . | |
| Fever | 80 | 97 | 88 | 87 | 76 | 64 | <.001 |
| Cough | 74 | 77 | 86 | 81 | 69 | 63 | <.001 |
| Chest pain | 57 | 6 | 16 | 48 | 78 | 84 | <.001 |
| Shortness of breath | 28 | 14 | 19 | 19 | 26 | 47 | <.001 |
| Productive cough | 24 | 7 | 13 | 15 | 28 | 44 | <.001 |
| Severe chest pain | 22 | 0 | 3 | 8 | 29 | 44 | <.001 |
| Chills | 18 | 4 | 4 | 8 | 22 | 39 | <.001 |
| Wheezing | 11 | 13 | 16 | 7 | 8 | 13 | .769 |
| Hemoptysis | 2 | <1 | <1 | <1 | <1 | 6 | .011 |
| No. of patients | 939 | 124 | 168 | 191 | 204 | 252 | — |
| . | Total . | Age (Yr) . | P Value* . | ||||
|---|---|---|---|---|---|---|---|
| . | . | <2 . | 2-4 . | 5-9 . | 10-19 . | 20+ . | . |
| Symptoms . | . | % of Patients . | |||||
| . | . | . | . | . | . | . | |
| Fever | 80 | 97 | 88 | 87 | 76 | 64 | <.001 |
| Cough | 74 | 77 | 86 | 81 | 69 | 63 | <.001 |
| Chest pain | 57 | 6 | 16 | 48 | 78 | 84 | <.001 |
| Shortness of breath | 28 | 14 | 19 | 19 | 26 | 47 | <.001 |
| Productive cough | 24 | 7 | 13 | 15 | 28 | 44 | <.001 |
| Severe chest pain | 22 | 0 | 3 | 8 | 29 | 44 | <.001 |
| Chills | 18 | 4 | 4 | 8 | 22 | 39 | <.001 |
| Wheezing | 11 | 13 | 16 | 7 | 8 | 13 | .769 |
| Hemoptysis | 2 | <1 | <1 | <1 | <1 | 6 | .011 |
| No. of patients | 939 | 124 | 168 | 191 | 204 | 252 | — |
P value for the difference between age groups 2 to 4 years and 20+ years adjusting for genotype (Hb SS or Hb SC). Results reflect symptoms from only first episode of ACS for each patient.
Presenting symptoms observed during a patient's first ACS event were predictive of symptoms during subsequent events. Even when corrected for age, the person who had pain (P = .04), fever (P = .01), wheezing (P <. 001), chills (P = .023), or productive cough (P = .008) at the first event was significantly more likely to have the same symptoms at the second event.
Physical findings.Vital signs at the time of hospitalization were age-dependent with children experiencing higher temperature, pulse rate, and respiratory rate than adults (Table 2). These findings were similar for both Hb SS and Hb SC subjects. The most frequent physical exam findings were rales and dullness to percussion. Notably, the second most common auscultory finding was a normal lung exam, which occurred in 35% of patients. After adjusting for age differences, patients with an abnormal lung exam were more likely to have a productive cough (P = .035), shortness of breath (P = .001), wheezing (P < .001), fever (P = .007), and multilobe involvement (P < .001) than those with a normal exam. Seventeen percent of patients had lower extremity pain when they presented with ACS. The frequency of extremity pain was age dependent for hemoglobin SS patients (P = .049). Nearly one quarter of adults presenting with ACS had at least one lower extremity finding.
Physical Exam Findings of Patients With ACS According to Age
| . | Total . | Age (yr) . | P Value* . | ||||
|---|---|---|---|---|---|---|---|
| . | . | <2 . | 2-4 . | 5-9 . | 10-19 . | 20+ . | . |
| . | . | % of Patients . | |||||
| . | . | . | . | . | . | . | |
| Vital signs | |||||||
| T > 39°C | 31 | 64 | 42 | 34 | 19 | 15 | <.001 |
| Respirations >40/min | 18 | 43 | 29 | 19 | 8 | 6 | <.001 |
| Pulse > 140 beats/min | 15 | 50 | 28 | 11 | 3 | 1 | <.001 |
| Physical exam | |||||||
| Rales | 46 | 40 | 47 | 45 | 42 | 54 | 0.119 |
| Normal exam | 35 | 40 | 35 | 36 | 39 | 28 | 0.077 |
| Dullness to percussion | 31 | 22 | 22 | 28 | 34 | 40 | <.001 |
| Rhonchi | 16 | 22 | 23 | 15 | 12 | 12 | 0.008 |
| Wheezing | 10 | 15 | 11 | 11 | 7 | 8 | 0.718 |
| No. of patients | 939 | 124 | 168 | 191 | 204 | 252 | — |
| . | Total . | Age (yr) . | P Value* . | ||||
|---|---|---|---|---|---|---|---|
| . | . | <2 . | 2-4 . | 5-9 . | 10-19 . | 20+ . | . |
| . | . | % of Patients . | |||||
| . | . | . | . | . | . | . | |
| Vital signs | |||||||
| T > 39°C | 31 | 64 | 42 | 34 | 19 | 15 | <.001 |
| Respirations >40/min | 18 | 43 | 29 | 19 | 8 | 6 | <.001 |
| Pulse > 140 beats/min | 15 | 50 | 28 | 11 | 3 | 1 | <.001 |
| Physical exam | |||||||
| Rales | 46 | 40 | 47 | 45 | 42 | 54 | 0.119 |
| Normal exam | 35 | 40 | 35 | 36 | 39 | 28 | 0.077 |
| Dullness to percussion | 31 | 22 | 22 | 28 | 34 | 40 | <.001 |
| Rhonchi | 16 | 22 | 23 | 15 | 12 | 12 | 0.008 |
| Wheezing | 10 | 15 | 11 | 11 | 7 | 8 | 0.718 |
| No. of patients | 939 | 124 | 168 | 191 | 204 | 252 | — |
P value for the difference between age groups 2 to 4 years and 20+ years adjusting for genotype (Hb SS or Hb SC). Results reflect symptoms from only first episode of ACS for each patient.
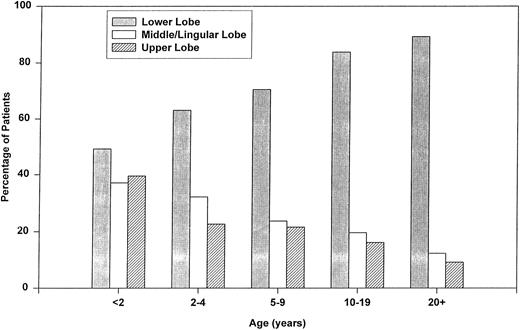
Radiographic findings.Radiographic findings varied by age (Fig 1). Young children had isolated upper (P < .001) and middle lobe (P < .001) disease significantly more often and lower lobe disease (P < .001) less often than adults. Adults presented with multilobe involvement (36% v 24%, P = .027) and pleural effusion (21% v 3%, P < .001) more frequently than children.
Age-specific radiographic findings among SCD patients with first episode of ACS.
Laboratory findings.Blood counts documented during ACS were compared with steady state values (Table 3). Hemoglobin and white blood cell count showed a significant change with Hb dropping an average of 0.7 g/dL and white blood cell count increasing by 69%. The difference between acute and baseline Hb levels was statistically significant (P < .05) for each age group for both Hb SS and Hb SC patients. The degree of Hb drop was consistent in the Hb SS and Hb SC genotypes.
Baseline and ACS Laboratory Values According to Age
| . | Total . | Age (yr) . | ||||
|---|---|---|---|---|---|---|
| . | . | <2 . | 2-4 . | 5-9 . | 10-19 . | 20+ . |
| Hemoglobin SS patients | ||||||
| Baseline Hb (g/dL) | 8.5 ± 1.3 | 8.7 ± 1.1 | 8.5 ± 1.11 | 8.1 ± 1.0 | 8.2 ± 1.0 | 9.0 ± 1.5 |
| ACS Hb (g/dL) | 7.9 ± 1.5 | 8.3 ± 1.5 | 7.8 ± 1.5 | 7.4 ± 1.3 | 7.7 ± 1.3 | 8.2 ± 1.6 |
| Baseline WBC (1,000/μL) | 13.3 ± 3.9 | 17 ± 7.0 | 14.6 ± 4.5 | 13.2 ± 3.3 | 12.4 ± 3.0 | 12.3 ± 2.8 |
| ACS WBC (1,000/μL) | 21.1 ± 8.6 | 21.5 ± 10.0 | 21.5 ± 8.9 | 22.3 ± 9.1 | 21.5 ± 7.6 | 19.4 ± 7.7 |
| PMN (% of differential) | 62 ± 16 | 46 ± 20 | 58 ± 15 | 64 ± 14 | 67 ± 14 | 65 ± 12 |
| Any NRBC (% of patients) | 47 | 36 | 42 | 47 | 47 | 55 |
| No. of patients | 738 | 92 | 136 | 151 | 154 | 205 |
| Hemoglobin SC patients | ||||||
| Baseline Hb (g/dL) | 11.2 ± 1.3 | 10.3 ± 0.4 | 10.6 ± 0.8 | 10.8 ± 0.8 | 11.5 ± 1.2 | 12.2 ± 1.6 |
| ACS Hb (g/dL) | 10.3 ± 1.6 | 9.9 ± 1.2 | 9.7 ± 1.3 | 9.9 ± 1.5 | 10.6 ± 1.8 | 11.2 ± 1.8 |
| Baseline WBC (1,000/μL) | 9.1 ± 2.7 | 10.7 ± 2.7 | 10.1 ± 3.3 | 8.1 ± 1.5 | 7.6 ± 1.9 | 9.6 ± 2.8 |
| ACS WBC (1,000/μL) | 15.8 ± 7.5 | 18.6 ± 8.8 | 16.4 ± 8.2 | 14.6 ± 5.0 | 13.6 ± 6.0 | 16.0 ± 8.3 |
| PMN (% of differential) | 62 ± 15 | 50 ± 18 | 59 ± 15 | 70 ± 13 | 64 ± 11 | 68 ± 11 |
| Any NRBC (% of patients) | 15 | 4 | 7 | 10 | 22 | 30 |
| No. of patients | 127 | 26 | 27 | 20 | 27 | 27 |
| . | Total . | Age (yr) . | ||||
|---|---|---|---|---|---|---|
| . | . | <2 . | 2-4 . | 5-9 . | 10-19 . | 20+ . |
| Hemoglobin SS patients | ||||||
| Baseline Hb (g/dL) | 8.5 ± 1.3 | 8.7 ± 1.1 | 8.5 ± 1.11 | 8.1 ± 1.0 | 8.2 ± 1.0 | 9.0 ± 1.5 |
| ACS Hb (g/dL) | 7.9 ± 1.5 | 8.3 ± 1.5 | 7.8 ± 1.5 | 7.4 ± 1.3 | 7.7 ± 1.3 | 8.2 ± 1.6 |
| Baseline WBC (1,000/μL) | 13.3 ± 3.9 | 17 ± 7.0 | 14.6 ± 4.5 | 13.2 ± 3.3 | 12.4 ± 3.0 | 12.3 ± 2.8 |
| ACS WBC (1,000/μL) | 21.1 ± 8.6 | 21.5 ± 10.0 | 21.5 ± 8.9 | 22.3 ± 9.1 | 21.5 ± 7.6 | 19.4 ± 7.7 |
| PMN (% of differential) | 62 ± 16 | 46 ± 20 | 58 ± 15 | 64 ± 14 | 67 ± 14 | 65 ± 12 |
| Any NRBC (% of patients) | 47 | 36 | 42 | 47 | 47 | 55 |
| No. of patients | 738 | 92 | 136 | 151 | 154 | 205 |
| Hemoglobin SC patients | ||||||
| Baseline Hb (g/dL) | 11.2 ± 1.3 | 10.3 ± 0.4 | 10.6 ± 0.8 | 10.8 ± 0.8 | 11.5 ± 1.2 | 12.2 ± 1.6 |
| ACS Hb (g/dL) | 10.3 ± 1.6 | 9.9 ± 1.2 | 9.7 ± 1.3 | 9.9 ± 1.5 | 10.6 ± 1.8 | 11.2 ± 1.8 |
| Baseline WBC (1,000/μL) | 9.1 ± 2.7 | 10.7 ± 2.7 | 10.1 ± 3.3 | 8.1 ± 1.5 | 7.6 ± 1.9 | 9.6 ± 2.8 |
| ACS WBC (1,000/μL) | 15.8 ± 7.5 | 18.6 ± 8.8 | 16.4 ± 8.2 | 14.6 ± 5.0 | 13.6 ± 6.0 | 16.0 ± 8.3 |
| PMN (% of differential) | 62 ± 15 | 50 ± 18 | 59 ± 15 | 70 ± 13 | 64 ± 11 | 68 ± 11 |
| Any NRBC (% of patients) | 15 | 4 | 7 | 10 | 22 | 30 |
| No. of patients | 127 | 26 | 27 | 20 | 27 | 27 |
Difference between acute and baseline Hb levels and WBC count was significant (P < .05) for each age group for both Hb SS and Hb SC patients. The degree of Hb drop was consistent in the Hb SS and Hb SC genotypes. Results reflect symptoms from only first episode of ACS for each patient.
Abbreviations: ACS, acute chest syndrome; Hb, hemoglobin; WBC, white blood cell; PMN, polymorphonuclear cells; NRBC, nucleated red blood cell.
Room air arterial blood gas sampling was performed in 56% (141 of 252) of first ACS events in adults. Mean PaO2 was 71 mm Hg and mean PaCO2 was 35 mm Hg. Nearly one fifth of the patients presented with a PaO2 less than 60 mm Hg and no clinical finding was predictive for severe hypoxia.
Bacteriology.Bacteremia was documented in 3.5% (33 of 939) of ACS episodes. The most common organisms isolated were Streptococcus pneumoniae (19 of 33 [58%]) and Hemophilus influenza (6 of 33 [18%]). The frequency and etiology of bacteremia was strongly influenced by age. Fourteen percent of children with SCD under 2 years had bacteremia associated with ACS and pneumococcus was the etiology in 78% of these cases. Children aged 0 to 4 years with bacteremia had temperatures ≥39°C (P = .005), elevated pulse rate (P < .001), elevated respiration rate (P = .037), upper lobe involvement (P = .034), and pleural effusion (P = .022) more often than children without bacteremia.
In contrast to young children, only 1.8% of patients aged 10 and older had bacteremia and only 25% were due to pneumococcus. In this group, other causes of bacteremia included Staphylococcus aureus, Salmonella, Enterobacter, Hemophilus influenza, and Clostridia. Eight episodes of bacteremia were detected in Hb SC or Hb Sβ+ thalassemia patients; almost all (7 of 8) were in children age 2 years or younger.
Seasonal factors.The rate of ACS was affected by the season of the year (Fig 2). The seasonal variation in young children was most striking with a lower rate of ACS in summer and an increasing rate into the winter. Other age groups followed a similar pattern, but were less pronounced. ACS rates were significantly higher in children aged 0 to 4 than in adults during the month of December (P < .05).
Therapy.Transfusion was administered in 26% of ACS events. Twenty-nine percent of Hb SS patients were transfused in contrast to only 14% of Hb SC patients (P = .002). Children with sickle cell anemia were transfused less frequently (22% v 39%, P < .001), but earlier than adults (2.2 days v 3.6 days, P < .039).
Seventeen percent of patients received narcotic analgesia for pain. Its use was age-dependent with 36% of adults receiving narcotic analgesia in contrast to 1% of patients under 2 years of age (P < .001).
Hospital course.Ninety-five percent of subjects were hospitalized for their first episode of ACS. The mean length of hospitalization was 6.8 days. Children were hospitalized for less time than adults (mean, 5.4 v 9 days, respectively, P < .001). For adults, hemoptysis (P = .029), expectoration (P = .042), shortness of breath (P = .004), and high respiratory rate (P = .05) predicted a longer hospitalizations (≥8 days) in adults. There were no predictors for a long hospitalization in children. There was no difference between Hb SS and Hb SC patients in the percent of patients hospitalized or the length of hospitalization.
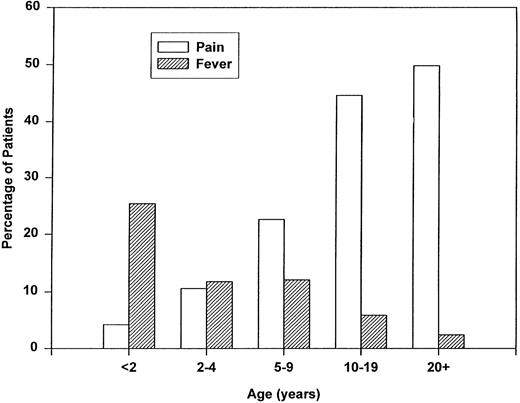
Associated events analysis.Pain and fever were the most common associated events (Fig 3). Overall, 30% of patients experienced a pain event, however, the percentage of adults who had pain before ACS was significantly higher than young children (50% v 11% respectively, P < .001). Fever was seen in 10% of patients before ACS and also showed an age predilection with 25% of infants versus 2% of adults (P < .001) experiencing a preceding febrile event. Surgery in the 2 weeks before the ACS event was seen in 1.5% of patients overall, but in 4.4% of adults versus 0.4 % of children with ACS.
Age-specific associated events within the 2 weeks preceding the first episode of ACS.
Age-specific associated events within the 2 weeks preceding the first episode of ACS.
Deaths.The overall death rate was 1.8% (32 deaths in 1,741 events, 949 patients). However, there was a striking difference in the death rate between adults and children. Of the 32 deaths, 14 occurred in patients less than 20 years of age for a death rate for children of 1.1% (14 deaths in 695 patients/1,322 events). Nine of the 14 deaths (64%) in the pediatric age group were in children less than 3 years of age, and seven of the 14 were associated with septicemia. The death rate in adults was 4.3% (18 deaths in 419 events, 271 patients). The death rate per event was similar for hemoglobin SC patients (1.6%, three deaths in 183 events) and Hb SS patients (1.9%, 27 deaths in 1,447 events).
Of the 32 deaths, detailed information on presenting signs and symptoms and laboratory data was available for 13 deaths identified with ACS forms. Five of the 13 (38%) patients had a history of ACS and three had chronic lung disease. No presenting complaint or physical finding predicted which patients would succumb, however the lowest presenting hemoglobins (mean, 4.9 g/dL) were seen in the patients who died within 24 hours of presentation. Except for multilobe disease, which occurred in the majority of fatal cases, chest radiography results in the patients who died were similar to that for ACS patients as a whole.
From analysis of death reports, four points were notable. First, most patients who would succumb developed respiratory failure within 48 hours after presentation. If these patients were alive after this period, it was with intensive cardiopulmonary support that later failed. Second, associated bacterial sepsis was common with nine patients having blood cultures positive for pneumococcus, three with gram negative rods, and one with group B Streptococcal sepsis. Thirdly, vaso-occlusive crisis in an extremity was present in at least three fourths of patients who died and was often the reason for seeking care. Lastly, autopsy reports demonstrated pulmonary embolus including massive fat embolism in nine of 16 patients.
DISCUSSION
An accurate characterization of the clinical course of ACS becomes necessary considering the morbidity of proposed interventions including steroids, chronic transfusion, and bone marrow transplantation.16-18 This prospective study demonstrates tremendous clinical variability in the presentation of ACS. This is most evident when comparing young children and adults with ACS. Young children commonly present with fever and cough and rarely have pain in association with ACS. Children are also more likely to have upper lobe disease or no chest exam findings at presentation. The seasonal predilection in ACS is most prominent in young children with a threefold higher rate in winter as compared with summer. Acute chest syndrome is generally less severe in children as indicated by less frequent transfusions, shorter hospitalization, and a lower mortality rate.
Adults, in contrast, are often afebrile and complain of shortness of breath, chills, and severe pain. Chest x-ray almost always involves the lower lobes. ACS in adults is generally more severe as evidenced by severe hypoxia, a higher rate of transfusion, longer hospitalization and a higher death rate.
Although this study was not designed to investigate the etiology of ACS, the difference in clinical presentations and severity between young children and adults supports the hypothesis of multiple etiologies. The milder, shorter course of childhood ACS in combination with its prominent seasonal predilection, high rate of bacteremia, and upper lobe disease suggest an infectious cause. Upper lobe disease has previously been associated with bacteremia.19 ACS in adults was characterized by severe pain and lower or multilobe disease and implies that vascular occlusion is a common cause of ACS in adults. The radiographic findings of predominantly lower lobe disease corresponds to autopsy results demonstrating pulmonary thrombosis and fat embolism.20,22 The finding of both pain and lower lobe disease in older patients suggests chest wall infarction may also be a common cause of ACS and is compatible with the clinical picture reported in the study by Bellet.23 Adults, also, are at greater risk for developing ACS in association with surgical procedures. A decrease in oxygenation and ventilation-perfusion mismatching with anesthesia may increase the risk of pulmonary infarction or infection.24
Presenting symptoms during a patient's first episode of ACS were predictive of symptoms during subsequent events. Knowledge of previous symptomatology should allow for earlier diagnosis and intervention in subsequent episodes. It is discouraging, however, that physical exam findings on presentation of ACS were often an unreliable indicator of the presence of disease and clinically there were no predictors of which patients would succumb. This reinforces the need for careful monitoring of all ACS patients.
Recent reports have suggested bacterial pneumonia is an uncommon cause of ACS.9,11,12,25 These studies also relied only on blood and sputum cultures and, therefore, likely underestimate the frequency of bacterial pneumonia. Bronchoscopy is more sensitive method in determining etiology and in the one adult study to use bronchoalveolar lavage, 20% of all episodes were bacterial.26 Due to the insensitivity of bacteremia in detecting bacterial pneumonia, the observation of bacteremia in 3.5% of ACS cases overall and 14% of infants suggests bacterial pneumonia is still a serious problem. The emerging resistance of pneumococcus to antibiotics is concerning and may need to be considered in choosing antibiotic coverage for the patient with severe ACS.27
The presentation and clinical course of ACS were similar in all of the sickling syndromes. Length of hospitalization, bacteremia, and death rates were similar for Hb SS and Hb SC patients. These findings point out the need for aggressive care in all ACS patients, even those with the milder sickling syndromes.
The overall death rate in this study was 1.8%, but adults had a death rate four times that of children. This may partly explain the wide discrepancy in death rates of previous reports on ACS, as these reports have tended to focus on either an adult or a pediatric population.2,5,11 12 Clinical deterioration and death occurred very rapidly, and often quite unexpectedly, in the majority of patients who died. Close surveillance of ACS patients is necessary, especially in the first 48 hours of hospitalization.
In summary, ACS appears to be multifactorial in etiology, and severity varies greatly between young children and adults. It is both a common cause of death and a predictor of shortened survival. ACS in children is generally mild and most likely results from infectious causes. In contrast, adults seem to suffer more from pulmonary thrombotic events, which lead to severe hypoxia, longer hospitalization, and a higher death rate.
ACKNOWLEDGMENT
We are indebted to Jolene Edwards for editorial assistance.
APPENDIX
The following are members of the Cooperative Study of Sickle Cell Disease.
Participating investigators: R. Johnson, Alta Bates Hospital (Berkeley, CA); L. McMahon, Boston City Hospital (Boston, MA); O. Platt, Children's Hospital (Boston, MA); F. Gill and K. Ohene Frempong, Children's Hospital (Philadelphia, PA); G. Bran, J.F. Kelleher, and S. Leikin, Children's Hospital National Medical Center (Washington, DC); E. Vichinsky and B. Lubin, Children's Hospital Oakland (Oakland, CA); A. Bank and S. Piomelli, Columbia-Presbyterian Hospital (New York, NY); W. Rosse, J. Falletta, and T. Kinney, Duke University (Durham, NC); L. Lessin, George Washington University (Washington, DC); J. Smith and Y. Khakoo, Harlem Hospital (New York, NY); R.B. Scott, O. Castro, and C. Reindorf, Howard University (Washington, DC); H. Dosik, S. Diamond, and R. Bellevue, Interfaith Medical Center (Brooklyn, NY); W. Wang and J. Wilimas, LeBonheur Children's Hospital (Memphis, TN); P. Milner, Medical College of Georgia (Augusta, GA); A. Brown, S. Miller, R. Rieder, and P. Gillette, State University of New York Downstate Medical Center (Brooklyn, NY); W. Lande, S. Embury, and W. Mentzer, San Francisco General Hospital (San Francisco, CA); D. Wethers and R. Grover, St Luke's-Roosevelt Medical Center (New York, NY); M. Koshy and N. Talishy, University of Illinois (Chicago, IL); C. Pegelow and P. Klug, University of Miami (Miami, FL); M. Steinberg, University of Mississippi (Jackson, MS); A. Kraus, University of Tennessee (Memphis, TN); H. Zarkowsky, Washington University (St Louis, MO); C. Dampier, Wyler Children's Hospital (Chicago, IL); and H. Pearson and A.K. Ritchey, Yale University (New Haven, CT).
Statistical coordinating centers: P. Levy, D. Gallagher, A. Koranda, Z. Flournoy-Gill, and E. Jones, University of Illinois School of Public Health (Chicago, IL), 1978-1989; S. McKinlay, O. Platt, D. Gallagher, B. Thorington and D. Brambilla, New England Research Institute (Watertown, MA), 1989-1990; and M. Espeland, Bowman-Gray School of Medicine (Winston-Salem, NC).
Program administration: M. Gaston, C. Reid, D. Bonds, and J. Verter, National Heart, Lung and Blood Institute (Bethesda, MD).
Supported by The Cooperative Study of Sickle Cell Disease, a program of the Sickle Cell Disease Scientific Research Group of the National Heart, Lung and Blood Institute, Bethesda, MD.
Address reprint requests to Elliott P. Vichinsky, MD, Children's Hospital Oakland, 747 - 52nd St, Oakland, CA 94609.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal