Abstract
Alveolar macrophages, which form a principal line of defense against a variety of pulmonary pathogens, may themselves be infected by viruses like human immunodeficiency virus-1 (HIV-1), which impair their defensive functions. Interleukin-13 (IL-13), a multifunctional cytokine, has been considered for therapeutic use based on its potent inhibition of HIV-1 in these cells. We have further examined the effects of IL-13 on alveolar macrophages under conditions that reflect those seen in acquired immune deficiency syndrome, where this cell type is often infected by the opportunistic pathogen human cytomegalovirus (HCMV). Alveolar macrophages exposed to both HCMV and HIV-1 consistently exhibited higher levels of HIV-1 replication than cells exposed to HIV-1 alone. HIV-1 production was strongly suppressed in alveolar macrophages treated with IL-13 regardless of whether or not the cultures were coinfected with HCMV. However, IL-13 treatment markedly enhanced the expression of HCMV in otherwise latently infected macrophages in a dose dependent manner. These unexpected differential effects of IL-13 on host-virus interactions are important considerations in guiding its potential therapeutic applications.
THERE IS considerable interest in identifying cytokines that may be used therapeutically against human immunodeficiency virus-1 (HIV-1) infection to augment immune function and/or inhibit retroviral production in infected cells. Interleukin-13 (IL-13) is a recently described cytokine secreted by activated T cells,1 which has been shown to be a potent in vitro modulator of human monocyte and B-cell functions.2,3 Among its pleiotropic activities, IL-13 induces significant changes in the phenotype of human monocytes, upregulating their expression of multiple cell surface molecules and increasing their antigen presenting capabilities. Exposure to IL-13 inhibits the transcription of many other cytokines in monocytes, including interleukin-1α (IL-1α), IL-1β, IL-6, tumor necrosis factor-β (TNF-β), and granulocyte-macrophage colony-stimulating factor (GM-CSF ),3 all of which have been implicated in enhancing HIV-1 replication in vitro.4-9
Recently, IL-13 has been shown to inhibit HIV-1 replication in blood-derived monocytes and mature lung macrophages, but not in T cells.1,10 The mechanism of this inhibition is not yet defined. However, in light of recent evidence that infection of monocytic lineages by HIV-1 may play important roles in the progression to AIDS,11-16 these findings suggest that this cytokine may have a therapeutic use against HIV-1.
Human cytomegalovirus (HCMV) is a major pathogen responsible for serious illness and death in acquired immune deficiency syndrome (AIDS) patients, as well as in newborns and in patients receiving immunosuppressive treatment following organ transplantation.17-19 HCMV is the most prevalent viral copathogen in HIV-infected individuals and, while overt HCMV pneumonia in AIDS patients is relatively rare, the virus can routinely be cultured from bronchoalveolar lavage samples from infected individuals.20 The lung has been identified as a principal organ site for murine CMV latency and recurrence, and appears to be a key reservoir in its life cycle.21 HCMV is known to infect alveolar macrophages,22-25 a key cell type mediating the normal immune responses against lung pathogens, including several bacterial and fungal organisms that are ubiquitous in AIDS.26 The increased susceptibility to these opportunistic pulmonary organisms is indicative that this local lung immune function is also disrupted in AIDS.
HIV-1 has also been shown to productively infect alveolar macrophages both in vitro and in vivo.27-31 Several reports have documented abnormal production of TNF-α, IL-6, and GM-CSF by alveolar macrophages isolated from HIV-1 infected individuals, indicating an autocrine mechanism for the augmentation of HIV-1 production by these mediators.32-36 Increased levels of HIV-1 infection of this cell type may also correlate with the progression to overt disease.37 38 Moreover, infection may lead to impaired host defense in this cell type in the lung.
Therefore, alveolar macrophages represent a clinically important cell population where coinfection by these two very different viruses may occur naturally. Numerous reports have documented interactions between HCMV and HIV-1.24,25,39-44 Although some conflicting data exist in the literature, the majority of the data indicate that coinfection with HCMV upregulates HIV-1 production in coinfected monocytes.43 44 Against this background, we examined the impact of IL-13 on the course of HCMV infection of alveolar macrophages and the effect of HCMV on HIV-1 replication in coinfected cultures treated with IL-13. Our studies show that while IL-13 treatment effectively inhibited HIV-1 even in cultures coinfected with HCMV, it also enhanced HCMV expression in human alveolar macrophages.
MATERIALS AND METHODS
Alveolar macrophage culture conditions.Alveolar macrophages were obtained by bronchoalveolar lavage from normal nonsmoking donors, and plated at a density of 1 × 106 cells per well in 24-well plates. The cells were allowed to adhere to the wells for 3 days before manipulation, and were maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% human AB serum, 1% sodium pyruvate, 1% nonessential amino acids, and antibiotics. The culture medium was changed completely every 3 days during experiments, and sampled for assays at these times. For exposure of alveolar macrophages to IL-13, the cells were pretreated with the indicated amount of the cytokine for 48 hours before challenging them with either HIV-1 or HCMV. The cultures were then supplemented again with the same concentration of fresh IL-13 at the time of each feeding.
Virus preparation.The HCMV strains Davis and RC256 were obtained from the American Type Culture Collection (Rockville, MD). HCMV RC256 is a recombinant strain of HCMV Towne, which has incorporated the lacZ gene of Escherichia coli under the regulation of the HCMV major early gene promoter.45 Both HCMV strains were propagated in human foreskin fibroblasts (HFF ) maintained in DMEM supplemented with 10% fetal bovine serum. Virus strains were passed at low virus to cell ratios to achieve a high titer of virus. HCMV stocks were generated by sonication of infected HFF cells after the appearance of overt cytopathic effects, and supernatants were cryopreserved at −80°C until use. The titer of infectious virus was determined by plaque assay.46
All HIV-1 virus isolates used in these studies were obtained from the AIDS Research and Reference Reagent Program (Rockville, MD). Monocytotropic HIV-1 isolates (HIV-1BAL , HIV-1JR-FL ) were propagated by passage through peripheral blood-derived macrophages (PBDM), and the lymphotropic HIV-1IIIB was propagated in Jurkat T cells.47 Culture medium from HIV-1 infected cultures was collected every 3 days, clarified by centrifugation at 1,500 RPM for 5 minutes, quantitated for reverse transcriptase (RT) activity and p24 antigen, and stored as cell-free virus at −80°C until use.
Virus infections.Four days after isolation, cultures to be exposed to HIV-1 alone were challenged with 0.25 mL of either HIV-1BAL , HIV-1JR-FL , or HIV-1IIIB (1 to 2 × 105 counts per minute [cpm]/mL by RT assay) for 4 hours. HIV-1 viral supernatants were then removed, and the cultures were washed 3× with Hanks' Balanced Salt Solution (HBSS) and maintained in 1 mL growth medium.
Macrophage cultures exposed to HCMV alone were challenged with 1 × 106 plaque forming units (PFU) HCMV (Davis or RC256 strain) at a multiplicity of infection (MOI) of 1.0 for 4 hours at 37°C. The virus inoculum was then removed and the cells were washed 3× with HBSS before being refed with fresh medium. Cultures exposed to both HIV-1 and HCMV were simultaneously challenged with 0.25 mL of HIV-1BAL or HIV-1JR-FL (1 to 2 × 105 cpm/mL by RT assay) and 1 × 106 PFU HCMV (Davis or RC256 strain) at an MOI of 1.0 for 4 hours, then refed with fresh growth medium. All cultures were maintained in 1 mL growth medium, which was changed and sampled every 3 days. Controls consisted of cells exposed to growth medium only for mock exposure. All experiments were performed in triplicate wells.
An HIV-1 antigen capture enzyme-linked immunosorbent assay (ELISA) (New England Nuclear, Boston, MA) was used to quantitate p24 gag protein concentration in cell-free supernatants and was reported as picograms of p24 per milliliter of sample. RT activity in cell-free supernatants from the HIV-1 exposed alveolar macrophage cultures was assayed as outlined by Hoffman et al48 for 2 hours at 37°C. In this assay, an activity level 5 times over background is considered positive.
Immunocytochemistry.Detection of HCMV infected cells was accomplished using methods adapted from Mattiace et al.49 Cultures were fixed in cold 80% methanol in phosphate-buffered saline (PBS) for 30 minutes, washed with TRIS buffered saline (TBS), and incubated 30 minutes at 4°C in a solution containing 0.25% Triton X-100 (Sigma Inc, St Louis, MO) and 3% H2O2 in TBS to quench endogenous peroxidase activity. Cells were then washed 3× with TBS and incubated with 2.5% nonfat dry milk/2.5% bovine serum albumin in TBS to block nonspecific antigens. After 1 hour, the blocking solution was removed and the cells were incubated with one of the following primary antibodies at the indicated dilutions in blocking solution overnight at 4°C; (1) anti-HCMV immediate early antigen (IEA; DuPont) at a dilution of 1:40; (2) anti-HCMV late nuclear antigen (DuPont) at a dilution of 1:25.
For chromogen staining, cells were washed 3 × 1 mL with TBS and incubated with a 1:200 dilution of a biotinylated goat antimouse secondary antibody (Vector Laboratories, Inc, Burlingame, CA) for 1 hour. Cells were then washed 3× with TBS and incubated with avidin coupled horseradish peroxidase (Vector Laboratories, Inc) for 60 minutes. The cells were then washed 3× with TBS, and the antigen was localized by incubation with 0.5 mg diaminobenzidine/mL, 100 mmol/L TRIS pH 7.6, and 0.075% H2O2 . HCMV antigens, localized by a dark brown precipitate, were observed using inverted light microscopy. Controls consisted of HCMV infected HFF, alveolar macrophages not exposed to HCMV stained with anti-HCMV IEA antibody, and alveolar macrophages exposed to HCMV stained with secondary antibody alone.
β-Galactosidase (β-gal) staining.Five days postinfection with the RC256 recombinant HCMV virus, the cells were washed twice with HBSS and fixed in cold glutaraldehyde/formaldehyde for 5 minutes. Cells were then incubated with X-gal stain (1 mg X-gal in dimethylsulfoxide, 2.1 mg potassium ferrocyanide, 1.6 mg potassium ferricyanide, and 2 mmol/L magnesium chloride) for 18 hours at 37°C. Cells were then washed 2× with HBSS, observed, and quantitated by light microscopy.
DNA polymerase chain reaction (PCR) for detection of lacZ sequences.Chromosomal DNA for the PCR analyses was isolated from the alveolar macrophages using the Puregene DNA isolation kit (Gentra Systems, Inc, Research Triangle Park, NC). DNA PCR was performed as previously described using primers to amplify β-gal.50 A positive control for a β-gal signal was generated from serial dilutions of Hela-CD4-LTR-β-gal DNA, which has one integrated β-gal copy per cell.51 This cell line was obtained through the AIDS Research and Reference Reagent Program (Rockville, MD) from Dr Michael Emerman. Each alveolar macrophage PCR reaction was set up using 25 ng of DNA. The PCR reaction was performed in a volume of 100 μL with 50 mmol/L KCl, 10 mmol/L Tris/HCl pH 8.3, 2.5 mmol/L MgCl2 , 200 μmol/L of the four deoxynucleotide triphosphate (dNTPs), 250 nmol/L of each primer, and 2.5 U of Amplitaq DNA polymerase (Perkin-Elmer Cetus, Branchburg, NJ). Template DNA was denatured at 95°C for 5 minutes before the addition of the amplification reagents. Cycling conditions for the PCR reactions were as follows: (1) 94°C × 30 seconds, (2) 57°C × 30 seconds, (3) 72°C × 60 seconds for 28 cycles in a Perkin-Elmer 9600 thermal cycler. A single 7 minute extension following amplification was performed at 72°C for all PCRs.
Liquid hybridization/gel retardation analyses.Following amplification, 30 μL of the amplified DNA was denatured by boiling for 5 minutes and then subjected to liquid hybridization (10 minutes at 55°C)51 with an internal 32P end-labeled oligonucleotide probe for β-gal.52 The hybridization conditions consisted of 200,000 cpm of the 32P-labeled probe and 1.5 mol/L NaCl/25 mmol/L EDTA, in a total volume of 20 μL per reaction. Hybridization products were subjected to gel retardation analyses through an 8% polyacrylamide gel in 1× TBE for 70 minutes at 220 V, and visualized overnight by autoradiography.
Statistics.Each cytochemical experiment quantitating the numbers of cells expressing HCMV antigens involved counting 3 separate wells per time point and/or cytokine dose. The data reported were compiled from three separate experiments and represented graphically as the mean number of positive cells per data point. Error bars indicate one standard deviation from the mean.
RESULTS
IL-13 blocks HIV-1 production in alveolar macrophages even in cultures coinfected with HCMV.Freshly isolated human alveolar macrophages from healthy seronegative donors were exposed to one of several strains of HIV-1, including the monocytotropic strains HIV-1BAL and HIV-1JR-FL or the T-cell tropic strain HIV-1IIIB , under the conditions described in Materials and Methods. The alveolar macrophages were readily infected by both monocytotropic strains as shown by the levels of HIV-1 core antigen (p24) and RT activity in culture supernatants. When virus-producing alveolar macrophage cultures were observed by light microscopy, large multinucleated giant cells were routinely detected by 30-days postinfection with either monocytotropic HIV-1 strain. No productive infection by the HIV-1IIIB virus or cytopathic effects resulting from the virus challenge were detectable, although the challenged cultures did become positive for HIV-1 sequences as determined by PCR (data not shown).
In multiple independent experiments, productive infection of the alveolar macrophages by HIV-1 was profoundly inhibited by continuous treatment of the cells with IL-13 beginning 48 hours before the virus challenge (a typical result employing the HIV-1BAL strain is shown in Fig 1). RT activity was not detected, and the levels of p24 antigen were close to or below the limits of detectability. This suppression could be maintained throughout the lifetime of the cultures (40 days) when IL-13 was continuously present. However, even at the highest doses of cytokine used, HIV-1 proviral sequences were still detectable by PCR in the virus-challenged cultures. Cessation of IL-13 treatment on day 7 or 11 postinfection resulted in breakthrough HIV-1 replication and detectable p24 antigen within 5 days (data not shown). Titration of the suppressive effect indicated that a partial inhibition was detectable at cytokine doses as low as 10 ng/mL, and was maximal at about 200 ng/mL. No enhanced cell toxicity was noted at any concentration of the cytokine tested as monitored by the trypan blue exclusion method.
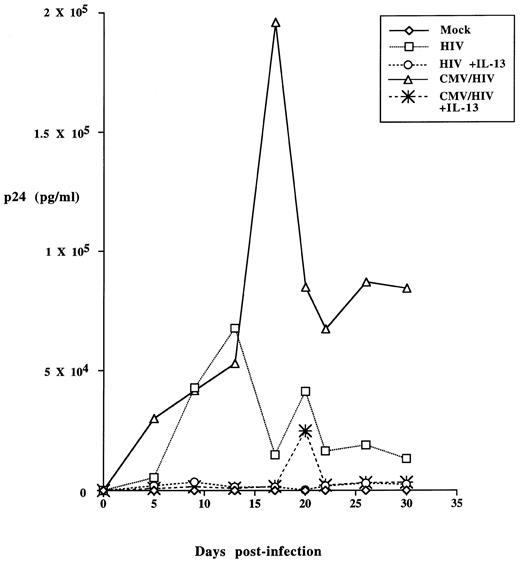
IL-13 blocks HIV-1 replication in alveolar macrophages coinfected with HCMV. Alveolar macrophages were cultured at a density of 1 × 106 cells per well for 7 days before exposure to HCMV and/or HIV-1. Half the cultures also received 50 ng/mL IL-13 for 2 days before virus challenge. Different cultures were then incubated with HIV-1BAL (5 × 105 cpm RT activity) with or without HCMV Davis (MOI of 1) as indicated. After 4 hours, the viruses were removed and the cultures were washed three times with 1 mL HBSS, then refed either with macrophage growth medium alone or supplemented with the same concentration of IL-13. Mock exposed alveolar macrophages received growth medium alone. Culture supernatants were removed and replaced with fresh growth medium at 5, 8, 13, 17, 20, 22, 26, and 30-days postinfection and stored at −20°C until assayed for HIV-1 p24 antigen production by ELISA (DuPont).
IL-13 blocks HIV-1 replication in alveolar macrophages coinfected with HCMV. Alveolar macrophages were cultured at a density of 1 × 106 cells per well for 7 days before exposure to HCMV and/or HIV-1. Half the cultures also received 50 ng/mL IL-13 for 2 days before virus challenge. Different cultures were then incubated with HIV-1BAL (5 × 105 cpm RT activity) with or without HCMV Davis (MOI of 1) as indicated. After 4 hours, the viruses were removed and the cultures were washed three times with 1 mL HBSS, then refed either with macrophage growth medium alone or supplemented with the same concentration of IL-13. Mock exposed alveolar macrophages received growth medium alone. Culture supernatants were removed and replaced with fresh growth medium at 5, 8, 13, 17, 20, 22, 26, and 30-days postinfection and stored at −20°C until assayed for HIV-1 p24 antigen production by ELISA (DuPont).
Both positive and negative effects of exposure to HCMV on the course of HIV-1 coinfection have been reported in other cell types and systems.42-44 Alveolar macrophages challenged with either of the two monocytotropic HIV-1 strains together with the Davis strain of HCMV consistently exhibited a greater production of HIV-1 compared to parallel cultures infected with HIV-1 alone (see representative data in Fig 1). However, simultaneous treatment of the cultures with IL-13 virtually eliminated p24 production by the dually infected cultures. HCMV was not able to overcome the suppression of HIV-1 replication mediated by IL-13. Coinfection with HCMV did not enhance the replication of the T-cell tropic HIV-1IIIB strain in alveolar macrophages under any conditions tested.
IL-13 treated alveolar macrophages exhibit enhanced expression of HCMV antigens.Based on these results, it appeared possible that the cytokine was affecting the course of the HCMV infection of the alveolar macrophages. To compare the extent of HCMV infection in alveolar macrophages in the presence or absence of the cytokine, cultures were exposed to the Davis strain of the virus after 2 days of pretreatment with IL-13, and then maintained in the presence of the cytokine until collection. Five days postinfection, the alveolar macrophages were analyzed by immunocytochemistry to detect the different classes of viral proteins. The genes of HCMV fall into three distinct classes, the immediate early (IE) and early (E) regulatory genes, and the late (L) structural genes.53 HCMV replication is characterized by the programmed sequential transcription of these three distinct genetic cassettes, with the IE cassette being expressed first.
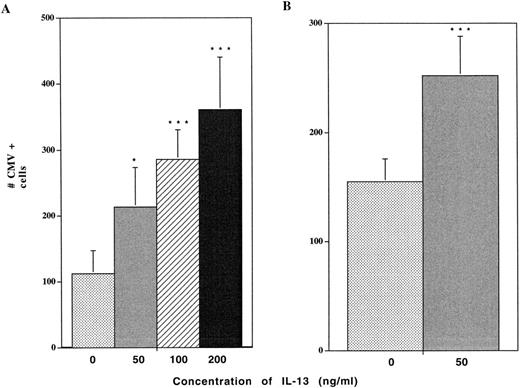
Treatment of alveolar macrophages with 100 ng/mL IL-13 consistently resulted in marked (fourfold to fivefold) increases in the numbers of cells expressing HCMV IE antigens in the cultures following exposure to the virus (Fig 2A). Titration experiments performed over a concentration range of 50 to 200 ng/mL IL-13 established a dose response relationship between the level of IL-13 and the number of cells expressing IE antigens postchallenge with the virus. Similar increases in the numbers of cells expressing a late nuclear antigen were also noted (Fig 2B). No positive cells were detected either in the control uninfected macrophages or in infected cultures probed with secondary antibody alone.
IL-13 increases HCMV antigen production in alveolar macrophages. Alveolar macrophages cultured at a density of 1 × 106 cells/well for 7 days were pretreated with the indicated concentrations of IL-13 for 2 days before virus challenge. The cells were then exposed to HCMV Davis (MOI of 1) for 4 hours, washed 3× with PBS and refed with growth medium containing the same concentrations of IL-13. Cells were fixed at day 5 postinfection and probed by immunocytochemical staining using antibodies against either HCMV immediate early (IE) antigen (A), or late nuclear antigen (LNA) (B). The numbers of antigen-expressing cells were quantitated by light microscopy at 200× magnification. Data are derived from 3 wells from 3 different experiments. Statistically significant differences from the control were determined by employing the Student's t-test. * = P < .05; ** = P < .01; *** = P < .001.
IL-13 increases HCMV antigen production in alveolar macrophages. Alveolar macrophages cultured at a density of 1 × 106 cells/well for 7 days were pretreated with the indicated concentrations of IL-13 for 2 days before virus challenge. The cells were then exposed to HCMV Davis (MOI of 1) for 4 hours, washed 3× with PBS and refed with growth medium containing the same concentrations of IL-13. Cells were fixed at day 5 postinfection and probed by immunocytochemical staining using antibodies against either HCMV immediate early (IE) antigen (A), or late nuclear antigen (LNA) (B). The numbers of antigen-expressing cells were quantitated by light microscopy at 200× magnification. Data are derived from 3 wells from 3 different experiments. Statistically significant differences from the control were determined by employing the Student's t-test. * = P < .05; ** = P < .01; *** = P < .001.
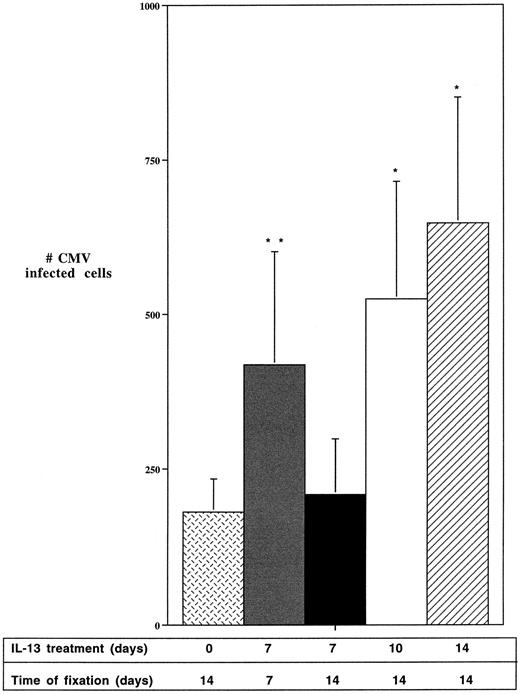
IL-13 treatment affects the HCMV lifecycle post entry.The enhancing effect of IL-13 on HCMV infection in macrophages could be the result of either increased numbers of cells initially infected, increased HCMV gene expression, or both. To discriminate among these mechanisms, kinetic experiments were performed to monitor the numbers of cells expressing HCMV antigens in virus-exposed cultures following the removal of IL-13 at various times over a 2-week period (Fig 3). Continuous inclusion of IL-13 for the 2-weeks postinfection resulted in the expected several-fold increase in the numbers of viral antigen-expressing cells as detected by immunocytochemistry. In contrast, alveolar macrophages infected with the HCMV Davis strain and treated with IL-13 for only 7 days postinfection, then cultured without IL-13 for an additional 7 days, exhibited similar numbers of IE antigen-expressing cells as infected cultures that were not treated with IL-13. HCMV infected macrophages exposed to IL-13 for 10 days postinfection and then cultured for an additional 4 days in the absence of the cytokine showed intermediate increases in the numbers of HCMV antigen-expressing cells. Together these data indicated that the enhanced numbers of macrophages expressing HCMV antigens as a result of IL-13 treatment were due primarily to the increased expression of the viral genes in cells harboring the virus, rather than increases in the numbers of infected cells resulting from the enhanced viral gene expression.
IL-13 enhances HCMV gene expression in infected macrophages. Day 7 alveolar macrophages, cultured at a density of 1 × 106 cells/well, were pretreated for 2 days with 50 ng/mL IL-13, unless indicated otherwise, and then exposed to HCMV Davis (MOI of 1). The virus was removed after 4 hours and the cells were maintained as follows: Mock: No IL-13. Cells fixed at day 14; 7/0: IL-13 for 7 days after HCMV infection. Cells fixed at day 7; 7/7: IL-13 for 7 days after HCMV infection. Cultures maintained for an additional 7 days without IL-13 and fixed on day 14; 10/4: IL-13 for 10 days after HCMV infection. Cultures maintained for an additional 4 days without IL-13 and fixed on day 14. 14/0: IL-13 for 14 days after HCMV infection, until the cells were fixed on day 14. All cells were fixed in methanol and probed with anti-CMV IE antibody. HCMV (+) cells were quantified under a light microscope at 200× magnification. Data are derived from 3 wells from 3 different experiments. Statistically significant differences from the control were determined by employing the Student's t-test. * = P < .05; ** = P < .01; *** = P < .001.
IL-13 enhances HCMV gene expression in infected macrophages. Day 7 alveolar macrophages, cultured at a density of 1 × 106 cells/well, were pretreated for 2 days with 50 ng/mL IL-13, unless indicated otherwise, and then exposed to HCMV Davis (MOI of 1). The virus was removed after 4 hours and the cells were maintained as follows: Mock: No IL-13. Cells fixed at day 14; 7/0: IL-13 for 7 days after HCMV infection. Cells fixed at day 7; 7/7: IL-13 for 7 days after HCMV infection. Cultures maintained for an additional 7 days without IL-13 and fixed on day 14; 10/4: IL-13 for 10 days after HCMV infection. Cultures maintained for an additional 4 days without IL-13 and fixed on day 14. 14/0: IL-13 for 14 days after HCMV infection, until the cells were fixed on day 14. All cells were fixed in methanol and probed with anti-CMV IE antibody. HCMV (+) cells were quantified under a light microscope at 200× magnification. Data are derived from 3 wells from 3 different experiments. Statistically significant differences from the control were determined by employing the Student's t-test. * = P < .05; ** = P < .01; *** = P < .001.
IL-13 treatment enhances HCMV gene expression.A replication-competent recombinant of the HCMV Towne strain, RC256, was employed to provide additional information regarding IL-13's effect on HCMV replication in the context of the live virus. In RC256, the E coli lacZ gene coding sequence has been substituted for nonessential sequences within the early gene cassette of the virus45 where its expression is regulated together with that of the other early genes. Expression of the early gene cassette following successful infection results in the production of β-gal, which is readily detectable using standard techniques.
Alveolar macrophages were infected with the RC256 virus either in the presence or absence of IL-13. Parallel cultures were set up for detection of either the β-gal enzyme itself or for isolation of cellular DNA for the purpose of detecting the lacZ gene. Staining of infected alveolar macrophage cultures with the chromogenic substrate X-gal 5-days postchallenge with RC256 clearly showed the presence of blue chromogen localized to the nuclear and perinuclear regions of the infected cells. X-gal assay of uninfected cells or cultures infected with the HCMV Davis strain, which does not contain the lacZ gene, exhibited no background staining under these conditions.
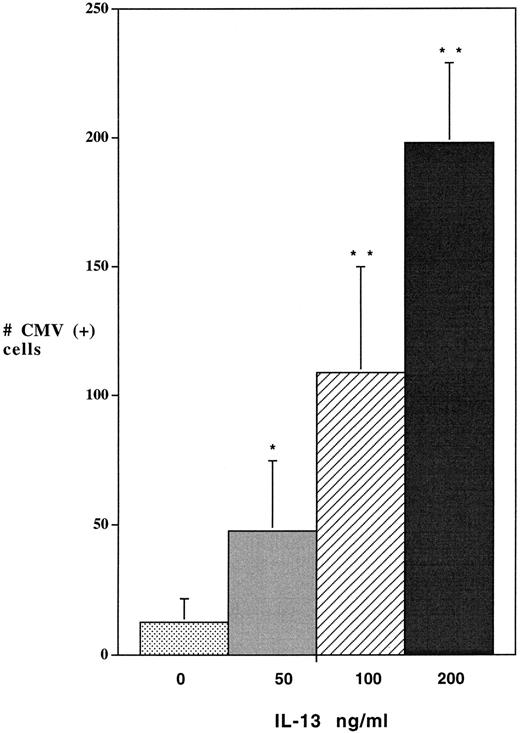
As with the previous immunocytochemical detection of IE and late nuclear antigens from the HCMV Davis strain, dramatic (up to 10-fold) increases in the numbers of cells expressing β-gal were produced in virus-infected macrophages by treatment with IL-13 (Fig 4). However, when DNA from these cells was used as a template for quantitative PCR amplification of the lacZ gene sequence, no significant differences in the relative copy number of the lacZ gene were detected between the IL-13 treated and control cultures (Fig 5). This finding indicated that the order of magnitude increase in the population of macrophages in which the β-gal protein could be detected in the IL-13 treated cultures was the result of active transcription of the viral genes in additional cells harboring the viral genome, rather than an absolute increase in the number of cells carrying the RC256 genome.
IL-13 enhances lacZ production by a recombinant HCMV virus. Alveolar macrophages, cultured at a density of 1 × 106 cells/ well for 7 days, were pretreated with different doses of IL-13 for 2 days before infection with HCMV. Cultures were then exposed to CMV RC256 (MOI of 1) for 4 hours, washed 3× with PBS, and refed with growth medium containing the noted concentrations of IL-13. Cells were fixed at day-5 postinfection and assayed by X-gal staining for β-gal activity. The numbers of cells expressing β-gal were quantitated by light microscopy at 200× magnification. Data are derived from 3 wells from 3 different experiments. Statistically significant differences from the control were determined by employing the Student's t-test. * = P < .05; ** = P < .01; *** = P < .001.
IL-13 enhances lacZ production by a recombinant HCMV virus. Alveolar macrophages, cultured at a density of 1 × 106 cells/ well for 7 days, were pretreated with different doses of IL-13 for 2 days before infection with HCMV. Cultures were then exposed to CMV RC256 (MOI of 1) for 4 hours, washed 3× with PBS, and refed with growth medium containing the noted concentrations of IL-13. Cells were fixed at day-5 postinfection and assayed by X-gal staining for β-gal activity. The numbers of cells expressing β-gal were quantitated by light microscopy at 200× magnification. Data are derived from 3 wells from 3 different experiments. Statistically significant differences from the control were determined by employing the Student's t-test. * = P < .05; ** = P < .01; *** = P < .001.
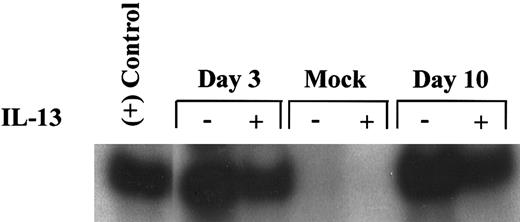
Detection of the lacZ gene sequence in macrophages infected with CMV RC256. Alveolar macrophages, cultured at a density of 1 × 106 cells/well for 7 days, were either pretreated with 50 ng/mL of IL-13 or received growth medium alone for 2 days before infection with HCMV. Cultures were then exposed either to the lacZ recombinant CMV, RC256 (MOI of 1), or growth medium (Mock) for 4 hours, washed 3× with PBS and refed with growth medium supplemented with IL-13 (+) or growth medium alone (−). The growth medium was changed every 3 days. Cells were collected at days 3 and 10 postvirus exposure, and 25 ng of cellular DNA was used as a template for amplification with β-gal primers by PCR under conditions described in Materials and Methods. An equal amount of DNA from the Hela-CD4-LTR β-gal cell line was amplified as a positive control. Amplified DNA was subjected to liquid hybridization with a 32P-labeled β-gal probe, electrophoresed through 8% polyacrylamide gel electrophoresis, and visualized overnight by autoradiography.
Detection of the lacZ gene sequence in macrophages infected with CMV RC256. Alveolar macrophages, cultured at a density of 1 × 106 cells/well for 7 days, were either pretreated with 50 ng/mL of IL-13 or received growth medium alone for 2 days before infection with HCMV. Cultures were then exposed either to the lacZ recombinant CMV, RC256 (MOI of 1), or growth medium (Mock) for 4 hours, washed 3× with PBS and refed with growth medium supplemented with IL-13 (+) or growth medium alone (−). The growth medium was changed every 3 days. Cells were collected at days 3 and 10 postvirus exposure, and 25 ng of cellular DNA was used as a template for amplification with β-gal primers by PCR under conditions described in Materials and Methods. An equal amount of DNA from the Hela-CD4-LTR β-gal cell line was amplified as a positive control. Amplified DNA was subjected to liquid hybridization with a 32P-labeled β-gal probe, electrophoresed through 8% polyacrylamide gel electrophoresis, and visualized overnight by autoradiography.
DISCUSSION
This study reports on the potent and opposing effects of the cytokine IL-13 on infection of alveolar macrophages by two clinically relevant viruses, HIV-1 and HCMV. The mechanism by which IL-13 inhibits HIV-1 is not yet clear. It has been suggested that the cytokine may act to block HIV-1 replication at the level of transcription. Montaner et al1 reported that IL-13 treatment of PBDM did not reduce either cell surface CD4 expression or thymidine incorporation into uninfected cells. They also reported no apparent difference between control and IL-13 treated macrophages at the levels of entry or reverse transcription of HIV-1 as determined by PCR for gag and long-terminal repeat sequences (LTR). Our findings of persistent low levels of proviral DNA subsequent to challenge with HIV-1 even in alveolar macrophages pretreated with IL-13, as well as a rapid rebound in virus replication following the removal of the cytokine, are fully consistent with these reports. The augmented HIV-1 replication we noted following coinfection of alveolar macrophages with HCMV, which did not occur in macrophages exposed to IL-13, has further implications for the activity of this cytokine. A major component of the enhancement of HIV-1 by HCMV has been attributed to interactions between the HIV LTR and HCMV IE proteins.39 The action of the cytokine presumably precedes such transcriptional interactions between the two viruses, because it abrogates their impact.
We then sought to determine if the suppressive effects of IL-13 also extended to HCMV infection of alveolar macrophages. Unexpectedly, IL-13 enhanced the expression of HCMV proteins in these macrophages in a dose dependent manner. This enhancement appeared to be principally at the level of expression of the viral genes, rather than in the susceptibility of the macrophages to HCMV infection. However, this enhanced activity may in turn have produced a change in the status of the virus infection in certain instances. Transfer experiments onto highly susceptible HFF cells indicated that challenge of untreated alveolar macrophages with either the HCMV Davis strain or the RC256 recombinant typically resulted in nonproductive infections, in agreement with other findings employing these particular strains in PBDM.54 However, in cultures infected with the HCMV Davis strain, low levels of productive infection were sometimes detectable in IL-13 treated cultures (data not shown). The heat liability of HCMV, with a half-life of less than 1 hour at 37°C,55 56 argues against this output virus representing the initial inoculum. A component of the enhanced detection of HCMV antigens in the cytokine-treated macrophage cultures may therefore have been the result of virus spread between 1- and 2-weeks postinfection. This would be consistent with the findings from the kinetic experiments. In the case of the RC256 virus, an isolate derived from the more attenuated Towne strain, no signs of productive infection were detected in either cytokine-treated or untreated cultures, consistent with the PCR analysis (data not shown).
Coinfection of cells with HIV-1 and HCMV in vivo has been reported in several tissues, including the brain,24 retina,25 and lung.41 The lung in particular represents a unique environment where frequent interactions between these two agents have ample opportunity to occur. As noted earlier, recent reports suggest a major role for the lung as a site of CMV latency and reactivation. The finding that IL-13 treatment increases the activity of HCMV in infected alveolar macrophages is a cause for concern when considering the therapeutic potential of this cytokine, especially in HIV-1 infected individuals, many of whom already suffer severe clinical syndromes due to HCMV. Our observations highlight the complex interactions that may occur between immune modulating cytokines and viruses, the latter having developed numerous mechanisms to thwart immune surveillance and clearance57 58 and, in some cases, to subvert the immune system to enhance the infection of host cells.
ACKNOWLEDGMENT
We thank Dr Alan Ezekowitz (Massachusetts General Hospital, Boston) and Rhone Poulenc, Inc (Los Angeles, CA) for generously providing the IL-13 used in these studies, and Drs Henry Koziel and Paula Pinkston (Beth Israel Deaconess Medical Center, Boston, MA) as well as Dr Hardy Kornfeld (Boston University Medical Center, MA) for supplying alveolar macrophages. Additional thanks are owed to Patricia Price for her technical assistance, and to Janet Delahanty for her graphics work and manuscript preparation.
Supported by National Heart, Lung, and Blood Institute Grant No. 5P01 HL435 10-05.
Address reprint requests to Ernest F. Terwilliger, PhD, Beth Israel Deaconess Medical Center, Harvard Institutes of Medicine, One Deaconess Rd, Boston, MA 02215.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal