Abstract
Human immunodeficiency virus-1 (HIV-1) Tat protein can be released by infected cells and activates mesenchymal cells. Among these, monocytes respond to Tat by migrating into tissues and releasing inflammatory mediators. In the present study, we have examined the molecular mechanism of monocyte activation by Tat, showing that this viral protein signals inside the cells through the tyrosine kinase receptor for vascular endothelial growth factor encoded by fms-like tyrosine kinase gene (VEGFR-1/Flt-1). Subnanomolar concentrations of Tat induced monocyte chemotaxis, which was inhibited by cell preincubation with vascular-endothelial growth factor-A (VEGF-A). This desensitisation was specific for VEGF-A, because it not was observed with FMLP. In addition, the soluble form of VEGFR-1 specifically inhibited polarization and migration induced by Tat and VEGF-A, thus confirming the common use of this receptor. Binding studies performed at equilibrium by using radiolabeled Tat showed that monocytes expressed a unique class of binding site, with a kd of approximately 0.2 nmol/L. The binding of radiolabeled Tat to monocyte surface and the cross-linking to a protein of 150 kD was inhibited specifically by an excess of cold Tat or VEGF-A. Western blot analysis with an antibody anti–VEGFR-1/Flt-1 performed on monocyte phosphoproteins immunoprecipitated by an monoclonal antibody antiphosphotyrosine showed that Tat induced a rapid phosphorylation in tyrosine residue of the 150-kD VEGFR-1/Flt-1. Taken together, these results suggest that biologic activities of HIV-1 Tat in human monocytes may, at least in part, be elicited by activation of VEGFR-1/Flt-1.
MONOCYTES AND macrophages have been implicated in a variety of relevant roles in human human immunodeficiency virus–type 1 (HIV-1) infection. During the latent phase of the disease, these cells represent an important viral reservoir for the transmission of virus to sensitive target cells.1,2 Productive HIV-1 replication in monocytes and macrophages is triggered by interleukin-6 (IL-6),3 granulocyte-macrophage colony-stimulating factor (GM-CSF ),4 lipopolysaccharide (LPS),5 or contact with endothelial cells6 and often predicts the clinical manifestation of the disease.2 Furthermore, HIV-1–infected monocytes/macrophages participate in tissue injury by releasing inflammatory cytokines and autacoids.7-9 The mechanism by which HIV-1 alters monocyte/macrophage gene expression and function is unknown, and a putative effector is the product of the regulatory viral gene tat. Tat protein transactivates viral genes, increases the replication rate of virion,10 and regulates host gene expression, including tumor necrosis factor α and β (TNF ), IL-2, IL-6, major histocompatibility complex of class I , IL-2 receptor, p53 tumor-suppressor gene, and superoxide dismutase in lymphoid cells11-17 and matrix proteins in glioblastoma cells.18
Furthermore, Tat can be released by infected cells19 and acts extracellularly in the microenvironment regulating functions of immunocompetent and mesenchymal cells.20,21 One of the most striking effects of Tat is the induction of a functional program in endothelial cells related to angiogenesis and inflammation (migration, proliferation, expression of plasminogen activator inhibitor-1 and E selectin, in vivo angiogenesis22-25 ). Subnanomolar concentrations of Tat promote the release of IL-6, transforming growth factor β (TGFβ) and metalloproteinase-9 by human monocytes11,26 increase their motility27 and adhesion to endothelial cells.26
Given the similarity of Tat basic sequence (aminoacids 42-64) to basic sequence of growth factor (fibroblast growth factor, vascular-endothelial growth factor-A [VEGF-A], hepatocyte growth factor, and heparin binding epidermal growth factor),24 we have recently shown that the tyrosine kinase VEGF receptor-2 (VEGFR-2)28,29 mediates Tat-induced migration of endothelial cells and in vivo angiogenesis.30 VEGF-A is a dimeric glycoprotein mitogenic for endothelial cells and able to induce angiogenesis and vessel permeability (reviewed in Ferrara et al31 and Kenneth32 ). Two high-affinity tyrosine kinase receptor for VEGF-A have been identified so far on vascular endothelial cells: VEGFR-1, encoded by the fms-like tyrosine kinase (Flt-1) gene,33 and VEGFR-2, encoded by kinase insert domain-containing receptor (KDR) gene.28,29 However, the activities of VEGF-A are not restricted to endothelial cells, because it also activates the induction of procoagulant activity by monocytes34 and their migration by acting through VEGFR-1.35 36
In light of these similarities between Tat and VEGF, we have investigated the chemotactic properties of Tat on human monocytes. This report shows that monocytes express a high-affinity binding site for Tat that induces a rapid (within minutes) cell shape change and migration. Monocytes activation by Tat is associated to phosphorylation of a 150-kD protein that cross-reacted with an anti–VEGFR-1 antibody. Finally, VEGF-A165 cross-desensitized with Tat in chemotaxis assay and competed with labeled Tat to monocytes, suggesting the promiscuous use of a common receptor by the two proteins.
MATERIALS AND METHODS
Tat protein.Recombinant full-length Tat protein (rTat) was obtained by Intracel (London, UK; batch R12.3). rTat (50 ng) gave a unique band of 14 kD (12%) detected by silver staining (Gelcode; Pierce, Europe B.V., Oud Beijerland, The Netherlands). Synthetic Tat (sTat), constituted by 86 aminoacid residues, was from Tecnogen (Piana di Monte Verna, Italy). The purity of this molecule was controlled by reverse-phase high-pressure liquid chromatography and was more than 97%. sTat was eluted from a Spherisorb C18 column (100 × 1 mm; Applied Biosystem, Foster City, CA) for 5 minutes under isocratic gradient (100% solvent A: 0.1% trifluoroacetic acid in water), for 40 minutes under a linear gradient from 0% to 95% solvent B (0.1% trifluoroacetic acid in CH3CN2 ), for 5 minutes under isocratic gradient (B 95%), and finally for 2 minutes under linear gradient to 5% solvent B. rTat and sTat were stored at −80°C in phosphate-buffered saline (PBS) containing 0.1 % human serum albumin (HSA; Farma Biagini, Lucca, Italy) and 1 mmol/L dithiothreitol. To label Tat, rTat (2 μg) was dissolved in 200 μL of sodium phosphate buffer at 20 mmol/L, pH 7.4, without dithiothreitol and transferred in iodogen-coated tubes (50 μg/mL; Pierce), where Tat was iodinated (5 minutes at 4°C) with 0.2 mCi 125I (Amersham, Buchs, UK). Twenty microliters of phosphate buffer at 20 mmol/L, pH 7.2, containing 1% HSA, 0.4 mol/L NaCl, and 0.1% Chaps (Pierce) was added, and the reaction products were separated on Sephadex-G10. The specific activity of the tracer was 1 mCi/127 fmol. [125I] Tat retained its biologic activity as migration of Kaposi's sarcoma cells.34
Cloning and expression of the soluble form of VEGFR-1/Flt-1.Human Flt-1 cDNA was cloned from a placental cDNA library. A 2-kb HindIII fragment of Flt-1 cDNA coding for the first five extracellular Ig-like domains was transfected into Spodoptera frugiperda clone 158 (Sf158) cells using a liposome transfection system (Life Technologies GmbH, Eggenstein, Germany) along with purified and Bsu-36I–linearized baculovirus DNA (Pharmigen, Hamburg, Germany). After infection, cells were maintained in serum-free medium SF900 II (Life Technologies). Positive baculovirus plaques were identified by RNA expression of Flt-1. The soluble form of VEGFR-1/Flt-1 (sFlt-1) was purified, initially by addition of ammonium sulphate to a final concentrations of 35%, and the resulting precipitate was discarded and the sFlt-1 in the supernatant was precipitated by the addition of ammonium sulphate to a final concentration of 65%. After precipitation, the samples were centrifuged at 2°C. The pellet was dissolved in 10 mmol/L sodium phosphate buffer (pH 6. 2) containing 0.15 mol/L NaCl and applied to heparin-Sepharose CL-6B column (1.5 × 4 cm; Pharmacia Biotech, Uppsala, Sweden). The column was washed with the same buffer containing 0. 3 or 0.6 mol/L NaCl and sFlt-1 was eluted as a single peak with 1.2 mol/L NaCl. The purity of the protein was analyzed using sodium dodecyl sulphate-polyacrilamide gel electrophoresis (SDS-PAGE) under denaturating and nondenaturating conditions and, after silver staining, there appeared a unique band of 70 kD. Purified protein was identified by NH2 -terminal sequence analysis using Edman degradation on an automatic gas sequencer (Applied Biosystems, Weiterstand, Germany).
Mononuclear cell preparation.Peripheral blood mononuclear cells (PBMCs) were obtained from buffy coats of healthy blood donors through the courtesy of Centro Trasfusionale, Ospedale Sacco (Milan, Italy). Blood was washed once with saline at 400g to remove plasma and platelets and then centrifuged on Ficoll (Biochrom; Seromed, Berlin, Germany) at 600g for 30 minutes at room temperature. PBMCs were collected at the interface, washed twice with saline, and resuspended in RPMI 1640 with 1% fetal calf serum (FCS; Irvine, Santa Ana, CA). In some experiments, monocytes were further purified (>85% pure) by centrifugation at 450g on a 46% iso-osmotic Percoll (Pharmacia Biotech) gradient, as previously described.37
Migration assay.Cell migration was evaluated using a chemotaxis microchamber technique,38 as previously described,37 using a 5-μm pore size polycarbonate filter (Neuroprobe, Pleasanton, CA). Polycarbonate (migrated cell count at the end point) or nitrocellulose (leading front distance as the end point) were used. sTat, rTat, VEGF-A165 ,35 and FMLP (Sigma, St Louis, MO) were used as stimuli. Twenty-seven microliters of chemoattractant solution or control medium (RPMI 1640 with 0.1% HSA, 1 U/mL heparin) was added to the lower wells of a chemotaxis chamber (Neuroprobe, Bethesda, MD). Fifty microliters of cell suspension (1.5 × 106/mL monocytes) was seeded in the upper chamber. At the end of the incubation (37°C in air with 5% CO2 for 90 minutes), filters were removed and stained with Diff-Quik (Baxter Spa, Rome, Italy) and five high-power oil-immersion fields were counted. Cross desensitization studies were performed by preincubating cells (2 × 106/mL monocytes in PBMCs) with the different agonists at 37°C in constant agitation for 30 minutes. The cells were then washed in RPMI 1% FCS, counted, and tested for their ability to respond to agonist stimulation.37 Recombinant pertussis toxin (PTox) was from Calbiochem (San Diego, CA). Cells were preincubated with different concentrations of the toxins at 37°C for 90 minutes. At the end of the incubation, cells were washed twice with RPMI and tested for their ability to migrate in response to chemotactic agonists.37 Ptox treatment did not alter cell viability (>90%) as evaluated by trypan blue dye exclusion.
Polarization assay.Monocyte polarization assay was performed as previously described39 with minor modifications. Briefly, monocytes (106/mL) were prewarmed in polypropilene tubes at 37°C for 5 minutes and then exposed to different concentrations of the agonists for 10 minutes. The reaction was stopped by adding an equal volume (1 mL) of ice-cold phosphate-buffered formaldehyde (10% vol/vol; pH 7. 2). The percentage of cells with bipolar configuration (front-tail) was determined in at least 200 cells for each tube by phase-contrast microscopy at 400× magnification. Each experiment was performed in duplicate.
Binding studies.For binding studies, monocytes (1 × 106) were incubated for 2 hours at 22°C in 0.2 mL RPMI 1640 medium containing 20 mmol/L (N-[2-hydroxyethyl]piperazine-N′[2-ethanesulfonicacid]) pH 7.4, 0.1% HSA, 0.2 U heparin, 100 μg/mL soybean trypsin inhibitor, and bacitracin (binding buffer) and 0.7 pmol [I125]Tat without or with the indicated concentrations of unlabeled polypeptides. Alternatively, monocytes were incubated in the same way with different concentrations of [125I]Tat in the presence of 100-fold excess of cold ligand. At the end of incubation, cell suspension were overlayered on 0.5 mL of dibutylphtalate (Sigma) and centrifuged for 10 minutes at 13,000g, and the pellet was solubilized with 2% SDS in PBS was counted in a γ counter. Specific binding, which was calculated by subtracting from the total the cpm bound after incubation with a 100-fold excess of unlabeled ligand, was approximately 80%. The kd was estimated by Scatchard plot using Ligand program (Elseviere-Biosoft, Cambridge, UK).
Cross-linking.Monocytes (2 × 107) were incubated at 22°C in 0.5 mL of binding buffer with 0.5 nmol/L [125I]Tat with or without a 50-fold excess of unlabeled rTat or VEGF-A165 for 90 minutes and then cross-linked with 1 mmol/L disuccimidyl suberate (Pierce) for 30 minutes. Cells were washed twice with PBS and separated by denaturating SDS-PAGE (8%). Radiolabeled proteins were visualized by autoradiography.
Tyrosine phosphorylation and immunoprecipitation experiments.Human monocytes (1 × 107) were washed three times with RPMI 1640 and then twice with PBS containing 1 mmol/L Na orthovanadate. Cells were stimulated for different times with rTat (10 ng/mL) at 4°C. At the end of incubation, cells were centrifuged (1 minute at 130,000g ) and then cells were lysed in 1 mL at 4°C in a 20 mmol/L Tris-HCl buffer, pH 7.2, containing 158 mmol/L NaCl, 1% Triton X-100, 1% sodium deoxycholate, 1 mmol/L ethylendiaminetetraacetic acid, 5 mmol/L ethylene glycol-bis(β-aminoethyl ether)N,N,N′,N′ tetracetic acid, and protease and phosphatase inhibitors (50 μg/mL pepstatin, 50 μg/mL leupeptin, 10 μg/mL aprotinin, 2 mmol/L phenyl-methyl-sulphonyl fluoride, 500 μg/mL soybean trypsin inhibitor, 100 μmol/L ZnCl2 , 1 mmol/L Na orthovanadate, 1 mmol/L NaF, and 2 mmol/L sodium pirophosphate). After centrifugation (20 minutes for 10,000g ), supernatants were precleared by incubation for 1 hour with rabbit antimouse IgG-agarose and then incubated for 1 hour with monoclonal antibody 4G10 antiphosphotyrosine (10 μg/mL; UBI, Salt Lake City, UT). Immune complexes were recovered on rabbit antimouse IgG-agarose. Immunoprecipitates were washed four times with lysis buffer, twice with the same buffer without detergents, and once with Tris-buffered saline. Proteins were separated by SDS-PAGE (8%), blotted onto nitrocellulose, and probed with polyclonal antibody anti–Flt-1 (Santa Cruz, Santa Cruz, CA). Immunoreactivity was determined by using the enhanced chemiluminescence reaction (Amersham).
Effect of rTat on human monocyte chemotaxis and polarization. (A) Human monocytes (1.5 × 106/mL in PBMCs) were tested for their ability to migrate across a 5-μm pore-size polycarbonate filter in response to different rTat concentrations (•). Results of one experiment (performed in triplicate and representative of at least 3 independent experiments) are shown. In the same experiment, migration to an optimal concentration (10 nmol/L) of FMLP was 149 ± 9 monocytes. **P < .001; *P < .01 versus control (no rTat) by paired Dunnett's test. The effect of Ptox on rTat-induced migration was evaluated by preincubating cells with 1 μg/mL PTox for 90 minutes (○). **P < .001 versus respective Ptox-untreated cells. Results of one experiment (performed in triplicate and representative of at least 3 independent experiments, mean ± SD) are shown. (B) Percoll-purified human monocytes (106/mL) were incubated with different concentrations of sTat for 10 minutes (○). The reaction was stopped and the percentage of cells with bipolar configuration (front-tail) was determined. Data are expressed as the percentage of increase over control (0%). In the absence of the agonist (control), 23.6% ± 4% (n = 3) monocytes were in the polarized configuration. Results are the mean ± SD of three independent experiments performed in duplicates. In the same experimental conditions, the effect of 10 nmol/L FMLP was 133% ± 19% (n = 3) increase over control. **P < .001; *P < .01 versus control (no rTat) by paired Dunnett's test. The effect of sFlt-1 on rTat-induced polarization was evaluated by preincubating cells with 210 ng/mL for 10 minutes (•). **P < .001 versus sFlt-1–untreated cells. Results of one experiment (performed in triplicate and representative of at least 3 independent experiments, mean ± SD) are shown.
Effect of rTat on human monocyte chemotaxis and polarization. (A) Human monocytes (1.5 × 106/mL in PBMCs) were tested for their ability to migrate across a 5-μm pore-size polycarbonate filter in response to different rTat concentrations (•). Results of one experiment (performed in triplicate and representative of at least 3 independent experiments) are shown. In the same experiment, migration to an optimal concentration (10 nmol/L) of FMLP was 149 ± 9 monocytes. **P < .001; *P < .01 versus control (no rTat) by paired Dunnett's test. The effect of Ptox on rTat-induced migration was evaluated by preincubating cells with 1 μg/mL PTox for 90 minutes (○). **P < .001 versus respective Ptox-untreated cells. Results of one experiment (performed in triplicate and representative of at least 3 independent experiments, mean ± SD) are shown. (B) Percoll-purified human monocytes (106/mL) were incubated with different concentrations of sTat for 10 minutes (○). The reaction was stopped and the percentage of cells with bipolar configuration (front-tail) was determined. Data are expressed as the percentage of increase over control (0%). In the absence of the agonist (control), 23.6% ± 4% (n = 3) monocytes were in the polarized configuration. Results are the mean ± SD of three independent experiments performed in duplicates. In the same experimental conditions, the effect of 10 nmol/L FMLP was 133% ± 19% (n = 3) increase over control. **P < .001; *P < .01 versus control (no rTat) by paired Dunnett's test. The effect of sFlt-1 on rTat-induced polarization was evaluated by preincubating cells with 210 ng/mL for 10 minutes (•). **P < .001 versus sFlt-1–untreated cells. Results of one experiment (performed in triplicate and representative of at least 3 independent experiments, mean ± SD) are shown.
Statistical analysis.Chemotaxis experiments were performed in triplicate. Results are presented as mean ± SD of a representative experiment or as a mean (±SE) of several experiments. Statistical significance was assessed using the Student's t-test.
RESULTS
Induction of human monocyte migration and polarization by Tat.Figure 1A shows that rTat induced a concentration-dependent migration of human monocytes that was statistically significant at the concentration of 1 ng/mL and reached maximal values at 10 ng/mL (5 and 500 pmol/L, respectively). These concentrations are in the range of the active concentrations described for monocytes26,27 and endothelial cells.30 At the optimal concentration of 10 ng/mL, the number of monocytes migrating to Tat was 87% ± 13% (n = 3) of that observed in the presence of an optimal (10 nmol/L) concentration of FMLP, a reference chemoattractant. Statistical analysis of checkerboard experiments performed with nitrocellulose filters showed that monocyte migration in response to rTat was mainly directional migration (chemotaxis), with a minor but consistent chemokinetic component (data not shown). Boiled rTat was inactive (data not shown). Identical results were obtained with sTat (data not shown). Monocytes exposed to rTat or sTat acquired a polarized morphology that was maximal at 10 minutes (data not shown) and was concentration-dependent, with a peak observed at 10 ng/mL (Fig 1B). These data suggest a direct effect of rTat on monocyte cytoskeleton reorganization, a prerequisite for cell movement.40 Tat-induced monocyte migration was sensitive to the action of PTox (Fig 1A and data not shown). At the highest concentration tested (1 μg/mL PTox), monocyte migration to 10 ng/mL rTat was inhibited 79% ± 9% (n = 3; P < .01; Fig 1A). In the same assay conditions, chemotaxis to FMLP was inhibited 81% ± 7% (n = 3), as previously reported.37 These data are consistent with the sensitivity to PTox of most chemotactic agonists,41 42 including those using tyrosine kinase receptors (ie, VEGF-A35165 ).
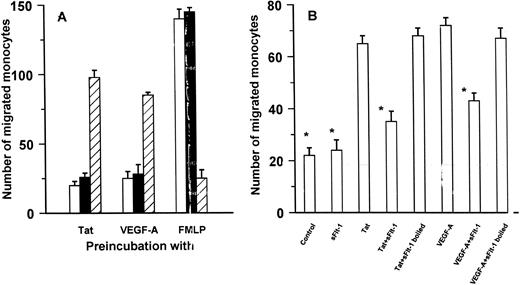
Interaction of Tat with Flt-1 on human monocytes.On the basis of previous observations in endothelial cells,30 the ability of rTat to cross-desensitize with VEGF-A165 in terms of chemotaxis was investigated. As shown in Fig 2A, pretreatment of monocytes with either 10 ng/mL rTat or 10 ng/mL VEGF-A165 resulted in more than 75% (P < .01 by Dunnett' test) of inhibition of a subsequent response to the two agonists. On the contrary, there was no (<20% inhibition) heterologous deactivation between rTat or VEGF-A165 and FMLP (Fig 2A).
rTat and VEGF-A165 activate human monocytes through the same receptor. (A) Homologous and heterologous desensitization of monocyte migration by rTat (□), VEGF-A165 (▪), and FMLP (▨). Monocytes were preincubated with optimal chemotactic concentrations of the agonists (10 ng/mL rTat, 10 ng/mL VEGF-A165 , and 10 nmol/L FMLP) at 37°C for 30 minutes. The cells were then washed and assayed for their migration toward homologous of heterologous stimuli used at the same concentrations. Results are expressed as percent of activity with respect to relative control group (cell incubated with medium and tested against the 4 single agonists). The mean numbers (±SE) of three separate experiments performed in triplicate are reported. (B) Monocyte migration by optimal chemotactic concentrations of the agonists (10 ng/mL rTat and 10 ng/mL VEGF-A165 ) was evaluated in the absence or in the presence of 210 ng/mL sFlt-1. Results (mean ± SD) of one experiment (performed in triplicate and representative of at least 3 independent experiments) are shown. In the same experiment, migration to an optimal concentration (10 nmol/L) of FMLP was 114 ± 7 monocytes. *P = .05 or lower versus rTat or VEGF-A165 .
rTat and VEGF-A165 activate human monocytes through the same receptor. (A) Homologous and heterologous desensitization of monocyte migration by rTat (□), VEGF-A165 (▪), and FMLP (▨). Monocytes were preincubated with optimal chemotactic concentrations of the agonists (10 ng/mL rTat, 10 ng/mL VEGF-A165 , and 10 nmol/L FMLP) at 37°C for 30 minutes. The cells were then washed and assayed for their migration toward homologous of heterologous stimuli used at the same concentrations. Results are expressed as percent of activity with respect to relative control group (cell incubated with medium and tested against the 4 single agonists). The mean numbers (±SE) of three separate experiments performed in triplicate are reported. (B) Monocyte migration by optimal chemotactic concentrations of the agonists (10 ng/mL rTat and 10 ng/mL VEGF-A165 ) was evaluated in the absence or in the presence of 210 ng/mL sFlt-1. Results (mean ± SD) of one experiment (performed in triplicate and representative of at least 3 independent experiments) are shown. In the same experiment, migration to an optimal concentration (10 nmol/L) of FMLP was 114 ± 7 monocytes. *P = .05 or lower versus rTat or VEGF-A165 .
Of the two VEGF-A165 receptors, human monocytes express on their surface only VEGFR-1/Flt-1.35,36 The soluble, truncated product of VEGFR-1/Flt-1 (sFlt-1) is a competitor of VEGF-A37 and has been used to verify the role of this receptor on the biologic response of Tat on monocytes. sFlt-1 inhibited both monocyte polarization (Fig 1B) and monocyte chemotaxis (Fig 2B) to rTat. The effect was concentration dependent, with an IC50 of 12 ng/mL for chemotaxis (not shown). At the highest concentration of sFlt-1 tested (210 ng/mL), inhibition of chemotaxis to rTat was 71% ± 10% (n = 4; P < .01). In the same experimental conditions, also monocyte migration to VEGF-A165 was significantly inhibited (56% ± 9%; n = 3; P < .01), whereas chemotactic response to FMLP was not affected (120% of control; n = 2; not shown). sFlt-1 did not change monocyte basal migration and boiled sFlt-1 was inactive (Fig 2B). These data strongly suggest the Tat and VEGF-A165 share common receptors on human monocytes.
To test the presence of high-affinity binding sites for Tat on human monocytes, cells were incubated with increasing concentrations of [125I]Tat in the presence of 100-fold amounts of cold molecule. The binding evaluated at equilibrium indicated the presence of a high-affinity binding site and the calculated kd was 280 ± 78 pmol/L (n = 3) with 320 ± 100 receptors per cell (n = 3; Fig 3A). As reported in chemotaxis experiments (Fig 2A), VEGF-A165 competed with [125I]Tat for the binding to monocytes (Fig 3B), and a concentration of 100 ± 12 pmol/L (n = 4) of unlabeled VEGF-A165 blocked half of the binding of the labeled ligand. Figure 3B shows that unlabeled rTat also inhibits the binding of [125I]Tat to monocytes, with a IC50 of 204 ± 67 pmol/L.
Specific binding of [125 I]Tat to monocytes and curve displacement with cold rTat and VEGF-A165 . (A) Specific [125I]Tat binding curve to monocyte at equilibrium and Schatchard analysis. For specific binding studies of Tat, human monocytes (1 × 106/ 0.2 mL of binding buffer) were incubated with increasing concentrations of [125I]Tat in the presence of a 100-fold excess of unlabeled ligand at 22°C for 120 minutes. At the end of incubation, cell suspension were overlayered on 0.5 mL of dibutylphtalate and centrifuged for 10 minutes at 13,000g, and the pellet was solubilized with 2% SDS in PBS was counted in a γ counter. (B) Ligand displacement curve at equilibrium of [125I]Tat binding to monocytes by rTat and VEGF-A165 . Human monocytes (1 × 106) were treated in the same way as in (A) with 0.7 pmol of [125I] without or with the indicated concentrations of unlabeled rTat (○) or VEGF-A165 (•). Each experiment was repeated at least three times.
Specific binding of [125 I]Tat to monocytes and curve displacement with cold rTat and VEGF-A165 . (A) Specific [125I]Tat binding curve to monocyte at equilibrium and Schatchard analysis. For specific binding studies of Tat, human monocytes (1 × 106/ 0.2 mL of binding buffer) were incubated with increasing concentrations of [125I]Tat in the presence of a 100-fold excess of unlabeled ligand at 22°C for 120 minutes. At the end of incubation, cell suspension were overlayered on 0.5 mL of dibutylphtalate and centrifuged for 10 minutes at 13,000g, and the pellet was solubilized with 2% SDS in PBS was counted in a γ counter. (B) Ligand displacement curve at equilibrium of [125I]Tat binding to monocytes by rTat and VEGF-A165 . Human monocytes (1 × 106) were treated in the same way as in (A) with 0.7 pmol of [125I] without or with the indicated concentrations of unlabeled rTat (○) or VEGF-A165 (•). Each experiment was repeated at least three times.
The affinity binding site for Tat was further characterized by cross-linking experiments (Fig 4A). Two [125I]Tat-containing major complexes with molecular weights of 92, 163, and 180 kD were detected when the ligand was cross-linked to the intact cells and displaced by 50-fold excess of unlabeled rTat or VEGF-A165 , indicating the interaction specificity and the cross-competition between the two polypeptides for the same protein (Fig 4A), confirming the biologic observation that VEGF-A165 has a competitive effect on Tat-induced monocyte movement (Fig 2A). A third complex of 90 to 100 kD was present, but was not displaced by an excess of cold ligands.
Cross-linking of [125I]Tat to monocytes and effect of rTat on tyrosine autophosphorylation of the VEGFR-1/Flt-1. (A) Monocytes (2 × 107) were incubated at 22°C in 0.5 mL of binding buffer with 0.5 nmol/L [125I]Tat with or without 50-fold excess of unlabeled rTat or VEGF-A165 for 90 minutes and then cross-linked with 1 mmol/L disuccimidyl suberate for 30 minutes. Solubilized proteins were separated by denaturating SDS-PAGE (8%) and visualized by autoradiography. (B) Human monocytes (1 × 107) were stimulated with rTat (10 ng/mL) in RPMI 1640 medium containing 1 mmol/L Na orthovanadate at 4°C. Cell lysates were immunoprecipitated with antiphosphotyrosine monoclonal antibody. After SDS-PAGE (8%) separation, proteins were immunoblotted with an anti–VEGFR-1/Flt-1 antibody and visualized by enhanced chemiluminescence technique. Each experiment was repeated at least two times.
Cross-linking of [125I]Tat to monocytes and effect of rTat on tyrosine autophosphorylation of the VEGFR-1/Flt-1. (A) Monocytes (2 × 107) were incubated at 22°C in 0.5 mL of binding buffer with 0.5 nmol/L [125I]Tat with or without 50-fold excess of unlabeled rTat or VEGF-A165 for 90 minutes and then cross-linked with 1 mmol/L disuccimidyl suberate for 30 minutes. Solubilized proteins were separated by denaturating SDS-PAGE (8%) and visualized by autoradiography. (B) Human monocytes (1 × 107) were stimulated with rTat (10 ng/mL) in RPMI 1640 medium containing 1 mmol/L Na orthovanadate at 4°C. Cell lysates were immunoprecipitated with antiphosphotyrosine monoclonal antibody. After SDS-PAGE (8%) separation, proteins were immunoblotted with an anti–VEGFR-1/Flt-1 antibody and visualized by enhanced chemiluminescence technique. Each experiment was repeated at least two times.
To further confirm that Tat uses VEGFR-1/Flt-1, we studied the tyrosine phosphorylation of the receptor after stimulation. rTat or VEGF-A165 induced the phosphorylation in tyrosine residues of a 150-kD protein recognized by anti–VEGFR-1/Flt-1 antibody (Fig 4B). Heat-inactivated rTat was inactive, showing the specificity of the phenomenon (data not shown).
DISCUSSION
Tat protein of HIV-1 activates biologic functions of mesenchymal cells, including monocytes and endothelial cells.11,13,20,21-27 We have previously shown that Tat binds VEGFR-2/KDR expressed on endothelial cells and activates its tyrosine kinase activity.30 Furthermore, antiserum neutralizing VEGFR-2/KDR or an excess of soluble form of KDR inhibited in vitro and in vivo activation of endothelial cells triggered by Tat, suggesting that this viral protein mimics VEGF-A.30
Besides migration and proliferation of endothelial cells,31,32 VEGF induces directed migration of monocytes and expression of procoagulant activity.34 This effect is mediated by a specific interaction with a single class of binding site with an apparent kd of 300 to 500 pmol/L.44 In contrast to endothelial cells, which express both VEGFR-1/Flt-1 and VEGFR-2/KDR,28,29,33 human monocytes express only VEGFR-1/Flt-1, which accounts for the observed biologic activities.35 36
Indeed, the observations that VEGF-A activates the migration of monocytes by a specific receptor-dependent mechanism31,35,36 and that rTat activates VEGFR-2/KDR in endothelial cells raise the possibility that a VEGFR is also essential for the migration and the invasiveness properties of human monocytes triggered by this HIV-1 protein.26 27
Human monocytes stimulated by subnanomolar concentrations of rTat start to migrate. This biologic effect shows a cross-desensitization with VEGF-A165 , but not with FMLP. Furthermore, monocyte migration elicited both by rTat and VEGF-A165 is blocked by Ptox, and as a whole these data suggest that the two molecules use an overlapping pathway to induce monocyte motility. sFlt-1, which lacks in transmembrane and kinase domains and inhibits biologic activities of VEGF-A on endothelium,43 has been used to support this hypothesis. sFlt-1 inhibited the polarization and the migration induced by rTat and VEGF-A165 , but was ineffective on the activity of FMLP. This suggests that sFlt-1 competes with membrane spanning VEGFR-1/Flt-1, as shown in endothelial cells,43 and thus can function as specific antagonist. However, it is possible to speculate that sFlt-1 makes a heterodimer with the membrane receptor that cannot signal inside monocytes upon rTat or VEGF-A165 binding.
Based on radioligand binding experiments, monocytes expressed a single class of Tat binding sites with a kd of approximately 0.2 nmol/L, similar to that reported for VEGF-A on monocytes.43 The displacement of [125I]Tat by an excess of VEGF-A165 again confirms the hypothesis that Tat uses a VEGFR. [125I]Tat was specifically cross-linked into two major complexes with molecular weights of 163 and 180 kD. These complexes were displaced by an excess of Tat or VEGF-A165 . Because the molecular mass of VEGFR-1/Flt-1 in monocytes is 150 kD (Fig 4), we can speculate that these complexes correspond to VEGFR-1/Flt-1 containing, respectively, one and two molecules of labeled Tat. A third complex of 90 to 100 kD was observed, but not displaced by an excess of cold rTat, suggesting an unspecified interaction due to the cross-linking procedure. However, we cannot exclude that the last complex is the result of a specific binding with a membrane protein that binds Tat with low affinity and high capacity (ie, an integrin45 ). The lack of displacement of [125I]Tat by the addition of cold rTat could be explained by the fact that experimental conditions have been standardized to study the high-affinity binding sites
The tyrosine kinase domain of VEGFR-1/Flt-1 is implicated in the ligand-dependent signal transduction in intact cells and the same receptor can be weakly phosphorylated on tyrosine residues upon VEGF-A challenge.33,46 47 Therefore, the rapid tyrosine phosphorylation of VEGFR-1/Flt-1 in rTat-stimulated monocytes suggests that the complex Tat/receptor transduces motogenic signals.
Experimental evidences have indicated that some extracellular activities of Tat can be mediated through specific interactions between the RGD-containing region and integrin receptors.25,27,45 48 Indeed, we cannot exclude a cooperation between VEGFR-1/Flt-1 and integrins in monocyte response to Tat.
The data presented in this report suggest that HIV-1-Tat could play an active role in monocyte extravasation and subsequent tissue damage in AIDS patients. Furthermore, the activation of monocyte VEGFR-1/Flt-1 by Tat present in serum or tissues20,25 could participate to the generalized impairment of immune system in infected subjects. A previous report has shown that monocytes from patients with AIDS have a selective defect of chemotaxis.49 Our preliminary data demonstrate that Tat desensitizes monocytes to unrelated chemotactic stimuli such as monocyte chemotactic peptide-1 (MCP-1; monocytes preincubated for 2 hours in control medium [% over the basal migration; n = 3]: Tat [10 ng/mL], 75 ± 12; VEGF-A165 [10 ng/mL], 72 ± 9; MCP-1 [50 ng/mL], 102 ± 15; and monocytes preincubated for 2 hours with 10 ng/mL rTat [% of the basal migration; n = 4]: Tat [10 ng/mL],10 ± 3; VEGF-A165 [10 ng/mL], 12 ± 2; MCP-1 [50 ng/mL], 15 ± 3]) and suggest that continued exposure to this viral protein could induce a loss in monocyte functions. We can also presume that Tat-VEGFR-1/Flt-1 interaction is instrumental in the defective function of professional antigen-presenting cells, including monocytes, as showed for VEGF-A.50
ACKNOWLEDGMENT
The authors thank A. Mantovani for helpful suggestions and criticisms.
S.M. and S.S. equally contributed to this work.
Supported by grants of Istituto Superiore di Sanità (AIDS Project and Italy-USA Program on Tumor Therapy), Associazione italiana per la Ricerca sul Cancro (Angiogenesis Special Project), and European Union (Biomed-2: BMHL-CT96-0669). S.M. and L.P. were supported, respectively, by a fellowship of “Fondazione Bossolasco” and of Associazione italiana per la Ricerca sul Cancro.
Address reprint requests to Federico Bussolino, MD, Dipartimento di Genetica, Biologia e Chimica Medica, Via Santena 5bis, 10126 Torino, Italy.


![Fig. 3. Specific binding of [125 I]Tat to monocytes and curve displacement with cold rTat and VEGF-A165 . (A) Specific [125I]Tat binding curve to monocyte at equilibrium and Schatchard analysis. For specific binding studies of Tat, human monocytes (1 × 106/ 0.2 mL of binding buffer) were incubated with increasing concentrations of [125I]Tat in the presence of a 100-fold excess of unlabeled ligand at 22°C for 120 minutes. At the end of incubation, cell suspension were overlayered on 0.5 mL of dibutylphtalate and centrifuged for 10 minutes at 13,000g, and the pellet was solubilized with 2% SDS in PBS was counted in a γ counter. (B) Ligand displacement curve at equilibrium of [125I]Tat binding to monocytes by rTat and VEGF-A165 . Human monocytes (1 × 106) were treated in the same way as in (A) with 0.7 pmol of [125I] without or with the indicated concentrations of unlabeled rTat (○) or VEGF-A165 (•). Each experiment was repeated at least three times.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/4/10.1182_blood.v90.4.1365/3/m_bl_0040f3.jpeg?Expires=1766110399&Signature=nhE993B2g3tvRAXJNXp5ASlbwHQpNuDbUVS1d87hk--neTLKAWc83WU~zkhAuj5M7k-7VsTf0ooaFBOITu3hZdWnhBm4mh5cpSRlDVWRNMKrH2oSzRoB5acbFIR5-Z6TMoggoaHy4y6QyElHuadXOT0FArfO1jnZ~MZDLhrei9dBEHwt3XBBvdWo95-n9FKotwbsbTKhFbSxQHhUTA94G~xP1emNFP1sLvuaDYDsAH1HkaAf6JQ5RscAv~wMNfPgAQqwMSobzE7FtmkVXng1UBoTJ0E9fq0YkdsNoDvPW6sTASP5gCnfDAXNkXizQNLABDAUgnXwmLJThq9P80PFTQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 4. Cross-linking of [125I]Tat to monocytes and effect of rTat on tyrosine autophosphorylation of the VEGFR-1/Flt-1. (A) Monocytes (2 × 107) were incubated at 22°C in 0.5 mL of binding buffer with 0.5 nmol/L [125I]Tat with or without 50-fold excess of unlabeled rTat or VEGF-A165 for 90 minutes and then cross-linked with 1 mmol/L disuccimidyl suberate for 30 minutes. Solubilized proteins were separated by denaturating SDS-PAGE (8%) and visualized by autoradiography. (B) Human monocytes (1 × 107) were stimulated with rTat (10 ng/mL) in RPMI 1640 medium containing 1 mmol/L Na orthovanadate at 4°C. Cell lysates were immunoprecipitated with antiphosphotyrosine monoclonal antibody. After SDS-PAGE (8%) separation, proteins were immunoblotted with an anti–VEGFR-1/Flt-1 antibody and visualized by enhanced chemiluminescence technique. Each experiment was repeated at least two times.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/4/10.1182_blood.v90.4.1365/3/m_bl_0040f4.jpeg?Expires=1766110399&Signature=dMAbOSQD2bg2lNmPEuQKY4H3BuXCuVEeg8O6o1stweo~6WT3F5Sp08jv5KedAJyGqoWlLb6CMjLgXqHmhVsiDT4KhsdQUu4vId6CwGqTRUEyxUnm4qipY7k5lhNnL1gl0Pcgs6wVfuFrVYgUNo4fsJ5eRunQm8JhJ0auTbD6Qmxh3BGw9zDTGYVVPmDpMiktAy1KUtmrZkV3cUtQnMivPW92fApHk8SpSJFcm86o7zC52BnCsf6hcu9sEshmx~zeJW0BxHtN6-oJe25ksjOBl1g4yi1AMmFncLx~g4ISVB9i6LWIg8wYpdhZdWHzvbYUACbfo-03Hs5r3J5H-v1H4g__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal