Abstract
This study shows that human postthymic T cells express CD10 when undergoing apoptosis, irrespective of the signal responsible for initiating the apoptotic process. Cells from continuous T-cell lines did not normally express CD10, but became CD10+ when induced into apoptosis by human immunodeficiency virus (HIV) infection and exposure to CD95 monoclonal antibody, etoposide, or staurosporin. Inhibitors of caspases blocked apoptosis and CD10 expression. Both CD4+ and CD8+ T cells purified from normal peripheral blood expressed CD10 on apoptotic induction. CD10 was newly synthesized by the apoptosing cells because its expression was inhibited by exposure to cycloheximide and CD10 mRNA became detectable by reverse transcription-polymerase chain reaction in T cells cultured under conditions favoring apoptosis. To show CD10 on T cells apoptosing in vivo, lymph node and peripheral blood T cells from HIV+ subjects were used. These suspensions were composed of a substantial, although variable, proportion of apoptosing T cells that consistently expressed CD10. In contrast, CD10+ as well as spontaneously apoptosing T cells were virtually absent in peripheral blood from normal individuals. Collectively, these observations indicate that CD10 may represent a reliable marker for identifying and isolating apoptosing T cells in vitro and ex vivo and possibly suggest novel functions for surface CD10 in the apoptotic process of lymphoid cells.
CD10 (NEUTRAL endopeptidase peptidase 24.11 [NEP]) is a 100-kilodalton kD, type II integral membrane protein characterized by a single hydrophobic sequence that functions as a signal peptide and a transmembrane region.1-4 CD10 was initially discovered on the surface of acute lymphoblastic leukemia cells and was considered to be a tumor-specific antigen.5 Later, it became clear that a variety of normal cells at particular maturational or functional stages are able to express CD10.3,4 These include cells of nonhematopoietic origin such as epithelial cells from kidney, liver, breast, and lung, as well as fibroblasts and cells of the central nervous system.6-13 Among the hematopoietic cells, CD10 is expressed by immature T and B cells,14-18 by the B cells of the germinal centers of lymphoid follicles,19 by granulocytes,8 and by the cells of a number of lymphoid malignancies.20
Although the neutral endopeptidase activity of CD10 is well documented,3,4 its function in the physiology of CD10-expressing lymphoid cells is poorly understood. However, some evidence, obtained primarily on mature B cells, suggests that CD10 expression may be related to apoptosis. For example, CD10 is found on germinal center B cells that are particularly prone to apoptosis and absent on other subsets of mature B cells that do not spontaneously undergo apoptosis.21-23 In addition, Burkitt’s lymphoma (BL) cells, which readily undergo spontaneous apoptosis both in vivo and in vitro, express abundant surface CD10.24,25Furthermore, B cells from lymphoblastoid B-cell lines transfected with c-myc–carrying episomes, concomitantly acquire the capacity to express CD10 and an increased propensity to spontaneous apoptosis in vitro.26,27 Finally, B cells induced into apoptosis by human immunodeficiency virus (HIV) infection in vitro express CD10.28
In the present study, we investigated whether the correlation between apoptosis and CD10 expression noticed in B cells was also true for T cells. Indeed, we found that mature T cells induced into apoptosis in a variety of manners in vitro, or spontaneously undergoing apoptosis in vivo, invariably expressed CD10. These findings indicate that CD10 may represent a valuable marker for apoptosing T cells and suggest a number of hypotheses for the physiological function of CD10.
MATERIALS AND METHODS
Patients and tissue or peripheral blood specimens.
Peripheral blood was obtained from 10 HIV-seropositive patients. Three were A2, 3 B2, 2 B3, and 2 C3 stage according to the Centers for Disease Control and Prevention staging system.29 Ten normal volunteers matched for sex and age were used as controls. Mononuclear cells (MNC) were separated by Ficoll-Hypaque density gradients and used for surface marker analysis.22 A fragment of a lymph node was obtained from an HIV+ patient undergoing biopsy for a suspected lymphoma. Subsequent histologic examination excluded the presence of malignant tissue. The specimen was minced to a fine suspension22 and analyzed by immunofluorescence. Informed consent for the use of tissue specimens was obtained from all patients.
Cells and cell cultures.
The H9 cell line was used for HIV infection. These CD4+ T cells were infected with HIV according to the method of Tersmette et al.30 T cells were purified from the peripheral blood of normal individuals by rosetting with neuraminidase-treated sheep red blood cells.22 CD4-positive cells were purified by removing CD8- and CD16-positive cells from these suspensions with immunomagnetic beads.23 Purified CD4+ T cells were stimulated with SEB (Staphylococcal Enterotoxin B; Sigma Aldrich, Milan, Italy) and/or CD4 MoAb according to the method of Westendorp et al.31 Briefly, CD4+ T cells were exposed to CD4 monoclonal antibodies (MoAb) (Leu3a; Becton Dickinson, Sunnyvale, CA) (1 μg/106 cells) in the cold for 30 minutes. The cells were washed, rosetted with goat anti-mouse immunoglobulin G (IgG) in the cold for 30 minutes and subsequently incubated in culture (5 × 105 cells/mL). SEB (50 ng/mL) was added to the cultures after 3 hours and the cells obtained at intervals. In control preparations an irrelevant MoAb (anti-CD19, Leu12; Becton Dickinson) was substituted for the anti-CD4 MoAb or SEB was omitted from the cultures. LAM was a BL cell line stabilized from an HIV patient.32
Continuous T-cell lines were generated from peripheral blood MNC of normal subjects. 1 × 106 MNC were stimulated with phytohemagglutinin (PHA) (1 μg/mL; Dakopatts, Glostrup, Denmark). Interleukin-2 (IL-2) (50 U/mL; Glaxo, Geneva, Switzerland) was added after 24 hours, and the cells were expanded in vitro with subsequent additions of IL-2 at the concentration of 20 U/mL. The culture medium used throughout was RPMI 1640 (Seromed, Biochrom KG, Berlin, Germany) supplemented with 10% fetal calf serum (FCS) (Seromed, Biochrom KG).
Immunofluorescence.
The following MoAb were used for immunofluorescence staining: anti-CD3 (Leu-4); anti-CD4 (Leu-3a); anti-CD8 (Leu-2a); anti-CD16 (Leu-11b); anti-CD25 (anti–IL-2 R); anti–HLA-DR (Becton Dickinson); anti-CD10 (J5) (Coulter Corp, Hialeah, FL); and anti-gp120 (NEN Life Science Products, Boston, MA). All of these MoAb were used in indirect immunofluorescence. The second fluorescein isothiocyanate (FITC)- or phycoerythrin (PE)-conjugated antibodies to the appropriate murine Ig isotype were from Southern Biotechnology (Birmingham, AL). The cells were analyzed by flow cytometry (Becton Dickinson) and no fixative was added to the stained cells. In the case of HIV-seropositive patients, the flow cytometer was washed extensively with sodium hypochlorite solution after the analyses. Triple staining was performed using the following reagents: CD3-ECD (Coulter Corp), CD10-Pe (J5) (Coulter Corp), and Annexin-V conjugated with FITC (1 μg/mL) (Apolert™ Apoptosis Kit; Clontech Laboratories Inc, Palo Alto, CA) or CD10 (J5) followed by a second step reaction antibody (anti-mouse IgG2A-PerCP; Becton Dickinson), together with Annexin-V-FITC and propidium iodide (PI) (Sigma Aldrich) 50 μg/mL in isotonic solution (phosphate-buffered saline). The cells were analyzed by flow cytometry (Epics-Elite flow cytometer; Coulter Corp). CD10-positive and -negative cells were physically separated by cell sorting (Epics-Elite). The 2 populations were gated on the basis of CD10 expression and forward light scatter parameter.
Reverse transcription-polymerase chain reaction (RT-PCR).
CD10 and glucose 3-phosphate dehydrogenase (GAPDH) transcripts were detected using RT-PCR as previously reported.22 The following synthetic primers33 were used: CD10 sense: 5′-TTGTAAGCAGCCTCAGCCG-3′; CD10 antisense: 5′-TTGTCCACCTTTTCTCGGAG-3′ (94°C for 1 minute, 50°C for 1 minute, 72°C for 1 minute; 35 cycles); GAPDH sense: 5′-ACATCgCTCAgAACACCTATgg-3′; GAPDH antisense: 5′-gggTCTACATggCAACTgTgAg-3′ (94°C for 1 minute, 59°C for 1 minute, 72°C for 2 minutes; 27 cycles).
After amplification, 45 μL of the PCR sample were run on a 2% agarose gel in the presence of the appropriate molecular-weight markers. The PCR products were detected by ethidium bromide staining and the gel was photographed with T55 film (Polaroid Corp, Cambridge, MA).
Amplification products were digested with specific restriction endonucleases (Alu I, TIB MOLBIOL; Genoa, Italy) as recommended by the manufacturer and the fragments were electrophoresed on 2.5% agarose gel (Metaphor; FMC Bioproduct, Rockland, ME).
The PCR products were purified by precipitation with ethanol and sequenced by the dideoxy-chain termination method, automatically using fluorescent-labeled ddNTP and Taq polymerase (PE Applied Biosystem, Foster City, CA). Data obtained were compared with the CD10 sequence obtained from the European Molecular Biology Laboratory (EMBL) database.
Apoptosis assays.
Cells were induced into apoptosis by exposure to either CD95 MoAb (200 ng/5 × 105 cells/mL) (FAS Immunotech, Marseille, France), etoposide (50 μmol/L) (VEPESID; Bristol-Myers Squibb, Rome, Italy), staurosporin (10 μmol/L) (Boehringer Mannheim, BmbH, Mannheim, Germany), SEB and CD4 MoAb, IL-2 starvation or infection with HIV depending on the design of the individual experiments. Apoptosis was measured by staining with Annexin-V conjugated with FITC (1 μg/mL) (Apolert Apoptosis Kit; Clontech Laboratories Inc) or by PI staining (Sigma Aldrich) and analyzed by flow cytometry (Becton Dickinson).
To inhibit protein synthesis during induction of apoptosis by CD95 MoAb, H9 T cells were preincubated for 2 hours with cycloheximide (50 μg/mL) (Sigma Aldrich).
To test the effect of caspase inhibitors on CD10 expression, H9 T cells were exposed to CD95 MoAb in the presence of VAD-FMK (10 μmol/L final concentration, Apolert ICE family protease inhibitor; Clontech Laboratories Inc). The inhibition of caspases was shown by using the CPP32/Caspase 3 Colorimetric Assay Kit (Clontech Laboratories Inc).
RESULTS
Expression of CD10 by apoptotic H9 T cells.
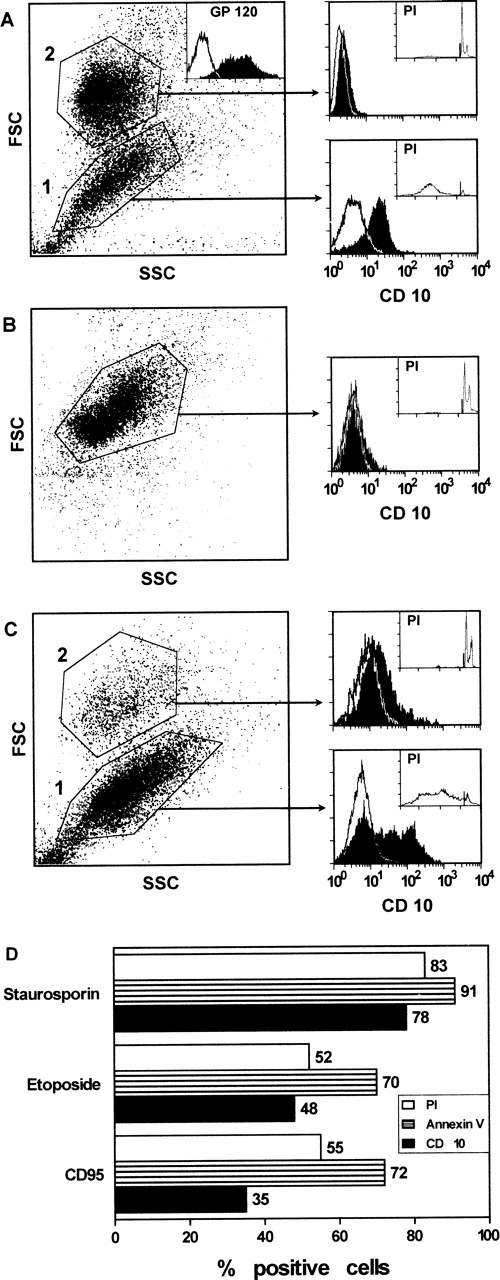
H9 T cells, chronically infected with HIV, were analyzed for CD10 expression. These cultures were composed of a substantial number of CD10+ cells (approximately 50%) and of an equivalent proportion of apoptotic cells. Both CD10+ cells and apoptotic cells were gated within the same cell subset (gate 1, Fig1A) characterized by FSC and side scatter light (SSC) profiles distinct from those of the remaining CD10− and nonapoptotic cells (gate 2, Fig1A). CD10-positive cells as well as apoptotic cells were not observed in the control (non–HIV-infected) cells (Fig 1B). These experiments suggested that cells undergoing apoptosis concomitantly expressed surface CD10. This correlation was investigated further in the experiments where H9 T cells were induced into apoptosis by exposure to CD95 MoAb for 24 hours. Again, 2 cell subsets with different FSC and SSC profiles were observed in the cell suspensions exposed to CD95 MoAb. One of these (gate 1, Fig 1C), which was composed of the majority of the cells, was enriched in both apoptotic and CD10-positive cells. CD10-positive or apoptotic cells were virtually absent in the other cell subset gated in 2 (Fig 1C). No CD10-positive or apoptotic cells were seen in the control cultures exposed to an irrelevant MoAb and the flow cytometry profile of these cells was identical to that of untreated cells (not shown). In another set of experiments, H9 T cells were exposed to staurosporin, etoposide, or CD95 MoAb, and both CD10 expression and apoptosis were measured after 24 hours. Again, after these treatments, there was an accumulation of apoptotic and CD10-positive cells in gate 1 (Fig 1D). Collectively, these experiments show that apoptosing H9 T cells expressed CD10, irrespective of the nature of the apoptotic signal.
CD10 expression by H9 T cells after HIV infection (A), exposure to CD95 MoAb (C), and staurosporin or etoposide (D) for 24 hours. The cells induced into apoptosis by one of these treatments were separated from nonapoptotic cells based on their FSC and SSC. Apoptotic and CD10+ cells were observed in the same gate (gate 1). Apoptosis was measured by PI staining of permeabilized cells (A through D) or Annexin-V staining (D) and flow cytometry. (D) Only the percentage of CD10+ and apoptotic cells gated in 1 is reported. Control cells (B), incubated with medium or with an irrelevant MoAb, were composed of a homogeneous cell population that did not express CD10 and was not apoptotic. The inset in A shows the flow cytometry profile for gp120 staining to document the ongoing HIV infection of the cells in vitro.
CD10 expression by H9 T cells after HIV infection (A), exposure to CD95 MoAb (C), and staurosporin or etoposide (D) for 24 hours. The cells induced into apoptosis by one of these treatments were separated from nonapoptotic cells based on their FSC and SSC. Apoptotic and CD10+ cells were observed in the same gate (gate 1). Apoptosis was measured by PI staining of permeabilized cells (A through D) or Annexin-V staining (D) and flow cytometry. (D) Only the percentage of CD10+ and apoptotic cells gated in 1 is reported. Control cells (B), incubated with medium or with an irrelevant MoAb, were composed of a homogeneous cell population that did not express CD10 and was not apoptotic. The inset in A shows the flow cytometry profile for gp120 staining to document the ongoing HIV infection of the cells in vitro.
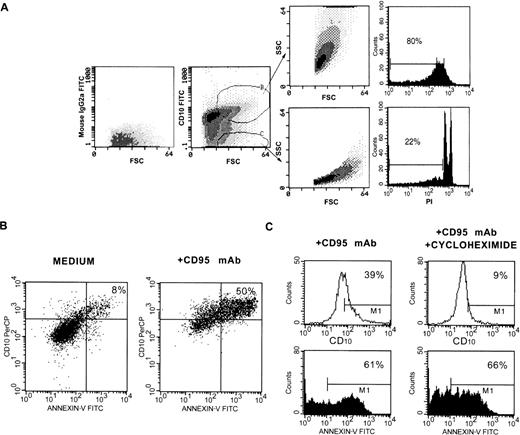
The above observations were checked in a number of control experiments. First, H9 T cells were induced into apoptosis by exposure to CD95 MoAb for 18 hours, stained with CD10 MoAb and analyzed by flow cytometry. Both large CD10+ and large CD10− cells that differed for SSC features (gates B and C in Fig2A) were sorted and reanalyzed for their SSC features and for apoptosis by PI staining. Small cells were excluded from the analyses because they were composed of some cells that become necrotic after an “early” apoptosis. Sorted CD10+ cells were composed of a large number of apoptotic cells. In contrast, in the sorted CD10 cells there were few apoptotic cells. Second, H9 T cells were exposed to medium or CD95 MoAb for 48 hours, obtained and triple stained in suspension with PI, CD10 and Annexin-V-FITC. Because the cells were stained in isotonic medium, PI staining discriminated between viable and nonviable cells with damaged membranes. The viable cells were gated and analyzed for CD10 and Annexin-V-FITC staining. As shown in Fig 2B, 50% of the viable cells present in the cultures exposed to CD95 MoAb double stained for Annexin-V and CD10, whereas only 8% of these cells were double stained in the control cultures. This finding rules out the hypothesis that both the CD10 and Annexin-V staining was because of nonspecific binding of the reagents by damaged (possibly necrotic) cells, which accumulate in culture after exposure to CD95 MoAb for a prolonged period. Finally, H9 T cells were exposed to CD95 MoAb for 24 hours in the presence of cycloheximide. Under these conditions, apoptosis was not blocked, as previously reported34; however, CD10 expression was inhibited, indicating that CD10 was synthesized by the cells induced into undergoing apoptosis.
Control tests to ascertain that CD10 is synthesized and expressed by apoptotic T cells. (A) Cells were exposed to CD95 MoAb for 18 hours, gated as indicated, and sorted. The sorted cells were analyzed for SSC and FSC or apoptosis by PI staining. (B) Cells were exposed to CD95 MoAb or medium for 48 hours and triple stained in isotonic medium with PI, Annexin-V-FITC, and CD10 MoAb-PerCP. The cells that excluded PI were gated and analyzed. (C) Cells were exposed to CD95 MoAb in the presence or absence of cycloheximide (50 μg/mL) and analyzed for CD10 expression and apoptosis by Annexin-V FITC staining.
Control tests to ascertain that CD10 is synthesized and expressed by apoptotic T cells. (A) Cells were exposed to CD95 MoAb for 18 hours, gated as indicated, and sorted. The sorted cells were analyzed for SSC and FSC or apoptosis by PI staining. (B) Cells were exposed to CD95 MoAb or medium for 48 hours and triple stained in isotonic medium with PI, Annexin-V-FITC, and CD10 MoAb-PerCP. The cells that excluded PI were gated and analyzed. (C) Cells were exposed to CD95 MoAb in the presence or absence of cycloheximide (50 μg/mL) and analyzed for CD10 expression and apoptosis by Annexin-V FITC staining.
Treatment of the cells with caspase inhibitors prevents CD10 expression.
The earliest event in apoptosis is the activation of caspases as documented by the finding that the inhibition of these enzymes results in the concomitant inhibition of their downstream events.35Caspase blockage can be achieved with synthetic cell-permeable peptides that are noncleavable analogs of their substrates and function as noncompetitive inhibitors.36 To test whether inhibition of caspases also resulted in downregulation of CD10 expression, we used VAD-FMK.37
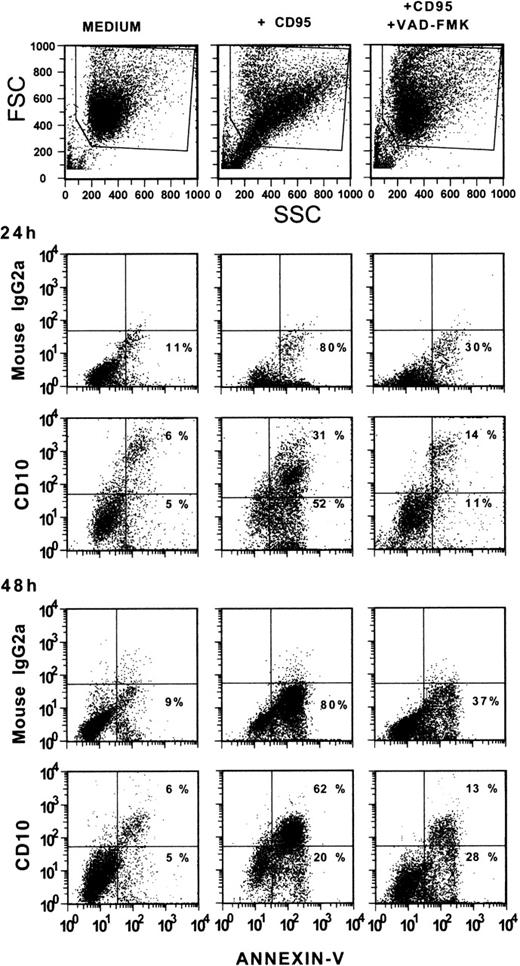
H9 T cells were cultured with an irrelevant MoAb, an anti-CD95 MoAb, or an anti-CD95 MoAb in the presence of VAD-FMK at a final concentration of 10 μmol/L. This concentration was selected based on the results of preliminary tests. The cells were cultured for 24 or 48 hours, obtained and double stained with Annexin-V-FITC and CD10 MoAb. As shown in Fig 3, a substantial proportion of cells had already undergone apoptosis after exposure to CD95 MoAb after 24 hours. At 48 hours, most of the CD95 MoAb-treated cells were apoptotic. Apoptotic cells coexpressed CD10. This coexpression was particularly evident in the 2-color staining tests performed at 48 hours, whereas the proportion of Annexin-V-positive cells somewhat exceeded that of CD10-positive cells at 24 hours, possibly indicating that the expression of phosphatidyl serine residues preceded that of CD10 (see Fig 1D). Indeed, Annexin-V binds to the phosphatidyl serine residues, which are expressed from the early stages of apoptosis.38 39 In the presence of VAD-FMK there were very few CD10-positive and Annexin-V–positive cells after 24 hours of culture and their percentage was virtually identical to that observed in the cultures with medium alone. Interestingly, the intensity of staining for CD10 of the few positive cells present in these 2 preparations was higher than that seen in CD95 MoAb-treated cells. This finding does not have an apparent explanation. At 48 hours, the proportion of cells staining for CD10 and Annexin-V in the culture exposed to CD95 MoAb and VAD-FMK was slightly increased compared with the 24-hour cultures. These values, although substantially lower than those observed in the cultures exposed to the CD95 MoAb alone, may indicate an inferior efficiency of VAD-FMK in preventing apoptosis in cultures extended over relatively long periods. By 48 hours, a large proportion of cells exposed to CD95 MoAb alone expressed both CD10 and Annexin-V binding sites, whereas a small minority of cells bound Annexin-V but were CD10-negative or only weakly positive. This minority, however, acquired the capacity to express CD10 on prolonged culture periods (72 hours, not shown).
Inhibition of CD10 expression by treatment with VAD-FMK. H9 T cells were cultured with CD95 MoAb in the presence or absence of VAD-FMK (10 μmol/L, final concentration) for 24 or 48 hours. Control suspensions were cultured with medium alone. At the end of the culture period, the cells were obtained and stained with CD10 MoAb and Annexin-V-FITC or an irrelevant MoAb and Annexin-V-FITC.
Inhibition of CD10 expression by treatment with VAD-FMK. H9 T cells were cultured with CD95 MoAb in the presence or absence of VAD-FMK (10 μmol/L, final concentration) for 24 or 48 hours. Control suspensions were cultured with medium alone. At the end of the culture period, the cells were obtained and stained with CD10 MoAb and Annexin-V-FITC or an irrelevant MoAb and Annexin-V-FITC.
When H9 T cells were exposed to etoposide for 48 hours in the presence of VAD-FMK, inhibition of both CD10 and apoptosis (Annexin-V-FITC–positive cells) was noticed (data not shown). Because chemically induced apoptosis is initiated primarily via the activation of executioner caspases,40 41 these observations indicate an involvement of such caspases in CD10 expression.
Expression of CD10 by peripheral blood CD4+ T cells after apoptosis induction.
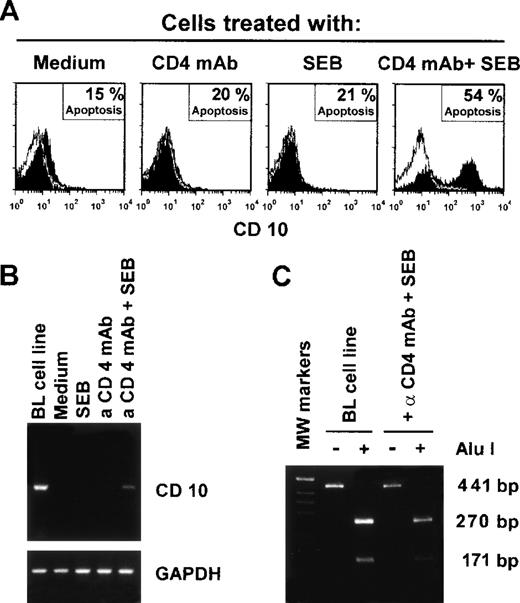
Peripheral blood CD4+ T cells were cultured with SEB in the presence or absence of a CD4 MoAb under cross-linking conditions. As already observed by other investigators,31,42 T cells exposed to SEB or CD4 MoAb alone failed to undergo apoptosis, whereas substantial apoptosis was detected in the cells exposed to the combination of the 2 reagents (Fig 4A). T cells that underwent apoptosis also expressed CD10 (Fig 4A). In these experiments, CD10 mRNA was also measured by RT-PCR in the T cells exposed to the various stimuli. It was only detected in the T cells exposed to both CD4 MoAb and SEB (Fig 4B), indicating that, after the induction of apoptosis, there was de novo CD10 synthesis. The CD10 mRNA band from the T cells was of identical size to that observed in BL cells that express CD10 constitutively,24 32 and also displayed the same restriction pattern after treatment with Alu 1 esonuclease (Fig 4C). Moreover, the nucleotide sequence of CD10 cDNA amplified from both T and BL cells was identical to that obtained from the EMBC databases (not shown).
Expression of CD10 by CD4+ T cells induced into apoptosis in vitro. (A) Purified CD4+ T cells were exposed to SEB and/or CD4 MoAb under cross-linking conditions in different combinations as indicated. The cells were obtained after 24 hours and CD10 expression and apoptosis (PI staining) were measured by flow cytometry. (B) RT-PCR analysis of CD10 mRNA expression in CD4+ T cells exposed to the indicated stimuli for 24 hours in vitro. Burkitt’s lymphoma B cells (LAM cell line) were used as positive control. (C) Alu I esonuclease digestion and analysis of the RT-PCR fragments extracted from the indicated cells. These results represent a typical experiment of the 3 performed.
Expression of CD10 by CD4+ T cells induced into apoptosis in vitro. (A) Purified CD4+ T cells were exposed to SEB and/or CD4 MoAb under cross-linking conditions in different combinations as indicated. The cells were obtained after 24 hours and CD10 expression and apoptosis (PI staining) were measured by flow cytometry. (B) RT-PCR analysis of CD10 mRNA expression in CD4+ T cells exposed to the indicated stimuli for 24 hours in vitro. Burkitt’s lymphoma B cells (LAM cell line) were used as positive control. (C) Alu I esonuclease digestion and analysis of the RT-PCR fragments extracted from the indicated cells. These results represent a typical experiment of the 3 performed.
Expression of CD10 by CD8+ T-cell blasts on induction of apoptosis.
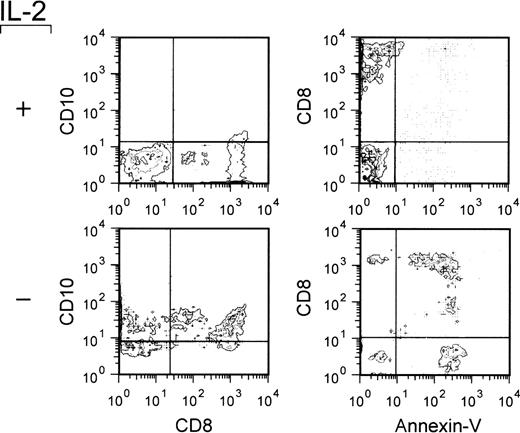
In these experiments, we investigated whether CD8+ T cells also expressed CD10 after the induction of apoptosis. Continuous cell lines of normal peripheral blood T cells stimulated with PHA and maintained in culture with IL-2 were used. These cell lines, which were composed of both CD4+ and CD8+ cells (not shown), were induced into apoptosis by culturing in the absence of IL-2.43 After 48 hours of IL-2 deprivation (which represented the optimal timing for detecting apoptosis), the cells were obtained and double stained with CD8 and CD10 MoAb or with CD8 MoAb and Annexin-V. As shown in Fig 5, which reports the results of a typical experiment out of the 3 performed on different cell lines, virtually all of the CD8+ cells also expressed CD10 in the absence of IL-2. Likewise, the large majority of CD8+ cells were Annexin-V–positive, thus indicating that CD8+ cells were capable of expressing CD10 on induction of apoptosis. As expected, analogous double-staining tests showed that the CD4-positive cells present in the same cell lines were able to express CD10 on induction of apoptosis (not shown).
Expression of CD10 by apoptosing CD8-positive T-cell blasts. T-cell blasts from IL-2–dependent continuous T-cell lines were cultured in the presence or absence of IL-2 (20 U/mL) for 48 hours. The cells were obtained, double stained as indicated, and analyzed by flow cytometry. The quadrants were drawn based on the analysis of negative controls stained with an irrelevant (isotype-matched) MoAb or analyzed in the absence of Annexin-V-FITC staining.
Expression of CD10 by apoptosing CD8-positive T-cell blasts. T-cell blasts from IL-2–dependent continuous T-cell lines were cultured in the presence or absence of IL-2 (20 U/mL) for 48 hours. The cells were obtained, double stained as indicated, and analyzed by flow cytometry. The quadrants were drawn based on the analysis of negative controls stained with an irrelevant (isotype-matched) MoAb or analyzed in the absence of Annexin-V-FITC staining.
Expression of CD10 by T cells that undergo apoptosis in vivo.
In these experiments, we investigated whether T cells apoptosing in vivo had the capacity to express CD10. The depletion of T lymphocytes, which occurs in HIV+ individuals, seems to be related to a large extent to apoptosis.44-47 Hence, tissues and peripheral blood from HIV+ subjects were likely to be composed of a sufficient number of apoptosing T cells to allow an assessment of their CD10 expression.48-51
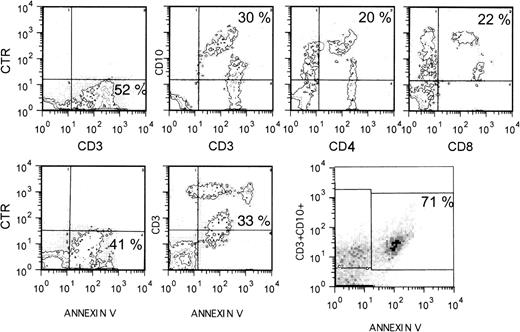
The initial experiment was performed on cell suspensions prepared from a lymph node of an HIV+ patient undergoing a diagnostic biopsy. These cells were double stained for CD3, Annexin-V, CD4, or CD8 in various combinations. As shown in Fig 6, a substantial proportion of CD3+ cells also stained for CD10 or Annexin-V. Triple staining showed that the majority of CD3+ cells that expressed CD10 also bound Annexin-V, thus indicating that CD10 represents a marker for apoptosing T cells in vivo. The apoptosing (CD10+) cells were found among CD4+ and CD8+ cells (Fig 6). From the flow cytometry profiles shown, it is evident that the intensity of staining for CD10 was inversely correlated with that for CD3, CD4, and CD8, consistent with the notion that apoptosing T cells downregulate a number of surface markers, including CD3, CD4, and CD8.52 53
Expression of CD10 by apoptosing T cells in vivo. Lymph node cell suspensions from an HIV-seropositive individual were double or triple stained as indicated and analyzed by flow cytometry. CTR (control) indicates that the cells were exposed to an irrelevant MoAb.
Expression of CD10 by apoptosing T cells in vivo. Lymph node cell suspensions from an HIV-seropositive individual were double or triple stained as indicated and analyzed by flow cytometry. CTR (control) indicates that the cells were exposed to an irrelevant MoAb.
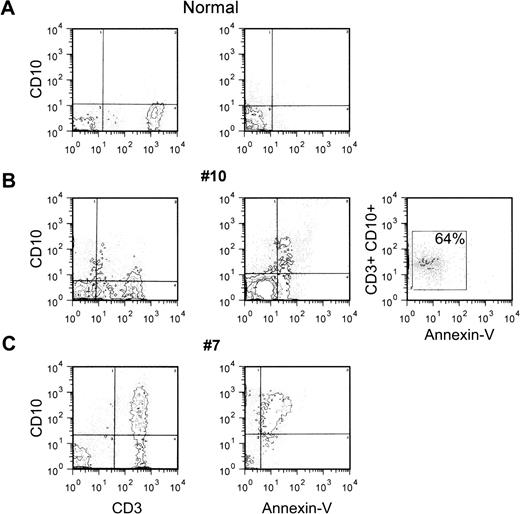
Subsequent experiments were performed on the peripheral blood MNC from 10 HIV-seropositive patients and 10 normal controls. As summarized in Fig 7, in normal controls the proportions of CD3+ cells that also expressed CD10 were 5% or less (3.3 ± 0.95). Values within the normal range were observed in only 3 of the 10 HIV+ individuals, whereas in the others there was a substantial, although variable, proportion of T cells that expressed CD10 (Fig 7). The CD10+ cells belonged to both the CD4+ and CD8+ cell subsets (Fig 7). Staining with Annexin-V showed that CD10-positive cells were apoptosing cells because in normal controls and in the 3 HIV-seropositive patients with low percentages of circulating CD10+ T cells, the proportion of Annexin-V+cells was also 5% or less (not shown). Moreover, double-staining tests confirmed that CD10 was on cells that bound Annexin-V. Figure8 shows 3 different flow cytometry profiles from 1 normal control and 2 HIV-seropositive individuals with different proportions of CD10+ cells. Triple-staining tests on the cells from patient no. 10 showed that the majority of cells that coexpressed CD10 and CD3 also stained with Annexin-V.
Expression of CD10 by T cells from the peripheral blood of HIV-seropositive individuals. Summary of the data obtained on the peripheral blood T cells from 10 HIV-seropositive patients and the average values obtained in 10 control individuals.
Expression of CD10 by T cells from the peripheral blood of HIV-seropositive individuals. Summary of the data obtained on the peripheral blood T cells from 10 HIV-seropositive patients and the average values obtained in 10 control individuals.
Expression of CD10 by apoptosing peripheral blood T cells from HIV-seropositive patients. Peripheral blood MNC from 1 normal control or 2 HIV-seropositive patients were double or triple stained as indicated in Fig 6, and analyzed by flow cytometry. The quadrants were drawn based on the analysis of negative controls stained with an irrelevant (isotype-matched) MoAb or in the absence of Annexin-V-FITC.
Expression of CD10 by apoptosing peripheral blood T cells from HIV-seropositive patients. Peripheral blood MNC from 1 normal control or 2 HIV-seropositive patients were double or triple stained as indicated in Fig 6, and analyzed by flow cytometry. The quadrants were drawn based on the analysis of negative controls stained with an irrelevant (isotype-matched) MoAb or in the absence of Annexin-V-FITC.
DISCUSSION
The present study shows a close correlation between CD10 expression and apoptosis of T cells. Evidence supporting this conclusion was obtained from in vitro tests and from observations on T cells obtained from HIV+ patients. Collectively, the experiments show that apoptosing cells of both the CD4+ and CD8+ cell subsets synthesize and express CD10, irrespective of the nature of the stimuli causing apoptosis.
As alluded to above, CD10 has a neutral endopeptidase activity. Structural studies have determined that the catalytic activity as well as the regulatory, zinc-binding moiety of this type II membrane protein is located in the extracellular segment near the COOH terminus.3,4 Therefore, CD10 hydrolyzes polypeptide molecules in the fluids that surround the apoptosing cells. Given the fact that CD10 is selectively expressed during apoptosis, its function may be that of cleaving proteins with inflammatory or proinflammatory activity released by the dying T cells. Hence, CD10 expression would represent a safety device intended to limit a potential inflammatory reaction triggered by the apoptosing cells. An alternative, and not mutually exclusive, function of CD10 might be that of degrading cytokines that reach the cell when the apoptotic process has already started. Because a variety of cytokines have a protective effect on T-cell apoptosis, CD10 expression may be seen as a means for potentiating the cell’s apoptotic ability by inhibiting the protective signal(s). Notably, CD10 is able to hydrolyze a variety of biologically active peptides, including growth and chemotactic factors.54-59 The latter of the 2 mechanisms described above might also apply to germinal center B cells, which have the propensity to undergo apoptosis and upregulate CD10 expression on apoptotic induction.19,21,22 Conceivably, CD10 expression makes the process of selection in the germinal center more stringent by increasing the threshold of cytokines required to prevent B-cell apoptosis. In a separate study, we found that those thymocytes that undergo apoptosis in vivo also express CD10 (Cutrona et al, manuscript in preparation). Because processes of positive and negative selection also occur within the thymus,60 one could speculate that CD10 augments the stringency of selection in this case, too. The present and previous studies suggest a strong correlation between CD10 expression and apoptosis in both T and B cells. Although an extrapolation of findings on lymphoid cells to other cells is not immediate, it is possible that the correlation also exists in other cell types.
A number of observations can be interpreted, at least in part, in light of the involvement of CD10 in cell apoptosis. These include the finding that inhibition of CD10 enzyme activity results in an increased capacity of CD10+ murine B-cell progenitors to proliferate in vitro and to reconstitute the B-cell compartment of irradiated mice in vivo,61 as well as the observation that the bombesin-dependent growth of the cells from small-cell lung cancers is enhanced after inhibition of CD10 activity.62 The expression of CD10 by the cells of certain malignant T-cell lines growing in culture may also be related to the number of cells undergoing apoptosis in vitro.63 Although the issue of whether inhibition of CD10 enzyme activity also results in an impaired apoptotic capacity of the cells will be the subject of a separate study, preliminary data have shown that inhibition of the neutral endopeptidase activity does not affect the intrinsic capacities of the cells to undergo apoptosis, a finding which is in line with the concept that CD10 expression may regulate cell apoptosis by interfering with the negative or positive signals delivered to the cell from the environment.
The present observations confirm that apoptosing cells have the ability to synthesize and transport into the cell membrane a new set of molecules, some of which may serve to facilitate recognition of the apoptotic cell by macrophages causing its subsequent elimination.64 Whether CD10 also has such recognition function must still be clarified.
T cells from HIV+ individuals were found to express CD10 (see Figs 6, 7, and 8). Two-color flow cytometry disclosed that these CD10+ T cells bound Annexin-V, thus showing that CD10 may represent a marker for T cells apoptosing in vivo. In line with the results of previous studies, we found substantial variations in the number of spontaneously apoptosing T cells in different individuals. These variations may be related to the clinical features of individual patients (ie, clinical stage and/or viral burden).48,49,51Although this aspect was not specifically investigated in this study, it may be worth mentioning that we have found correlations between proportions of CD10-expressing T cells, viral burden, and clinical stage of HIV infection in the course of preliminary tests. The method selected to measure apoptosis may also account for some of the existing discrepancies44-51 on the reported numbers of spontaneously apoptosing T cells in HIV-seropositive patients.65
To be a useful marker for apoptosing T cells, CD10 has to be detected simultaneously with another T-cell marker (eg, CD3) in double staining tests. This may pose some difficulties as apoptosing T cells tend to downregulate the expression of a number of surface molecules. Perhaps for this reason, we found that flow cytometry is the most suitable method for detecting apoptosing CD10+ T cells, possibly owing to its great sensitivity. In contrast, we faced a number of difficulties with immunohistochemistry techniques in tissue sections, probably because of the impossibility of detecting minute amounts of CD3 on apoptosing T cells (V.L.B., unpublished data, 1998).
Although there is general consensus that the in vitro apoptosis of cells from HIV-harboring cell lines is directly related to the infection,66,67 the mechanisms that cause the in vivo apoptosis of T cells in HIV+ individuals are more complex, one particular feature being that the large majority of apoptosing CD4+ or CD8+ T cells are non–HIV-infected cells.44-48 Indeed, in these experiments we found that a substantial fraction of T cells apoptosing in vivo was represented by CD8+ T cells that usually do not harbor HIV (see Figs 6 and7). Several mechanisms have been proposed to explain the apoptosis of uninfected T cells, including surface CD4 cross-linking by gp120, autocrine interaction of surface Fas with its ligand, and IL-2 starvation31,42 68; however, the phenomenon remains poorly understood. CD10 expression provides a new tool for identifying and possibly isolating apoptosing T cells ex vivo and, hence, for exploring the problem further. Moreover, the proportion of CD10+ T cells may possibly represent an additional parameter for evaluating HIV-related disease progression.
ACKNOWLEDGMENT
We thank M. Ulivi for revising the manuscript, Drs F. Fais and S. Zupo for helpful discussion, and T. Tavilla for excellent assistance in the preparation of this manuscript.
Supported by grants from Istituto Superiore di Sanità (ISS) (AIDS Project), Associazione Italiana per la Ricerca sul Cancro (AIRC), and Fondazione Andrea Cesalpino (to V.L.B.); N.L. is a fellow of the Associazione Italiana Leucemie (AIL).
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. section 1734 solely to indicate this fact.
REFERENCES
Author notes
Address reprint requests to Giovanna Cutrona, PhD, Servizio di Immunologia Clinica, Istituto Nazionale per la Ricerca sul cancro, IST, Largo Rosanna Benzi, n. 10, 16132 Genova GE—Italy; e-mail: gcutrona@hp380.ist.unige.it.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal