Abstract
Histo-blood group A and B antigens are oligosaccharide antigens important in transfusion and transplantation medicine. The final steps in the synthesis of these antigens are catalyzed by glycosyltransferases encoded by the functional alleles at the ABO locus. Humans have 3 major alleles (A, B, and O), whereas pigs are known to have only A and O alleles. This paper reports the molecular genetic basis of the porcine AO system. The porcine A gene is homologous to the ABO genes in humans and other species. It encodes an α1 → 3N-acetyl-D-galactosaminyltransferase that synthesizes A antigens. Southern hybridization experiments using a porcine A gene coding-sequence probe failed to identify a corresponding homologous sequence in genomic DNA from group O pigs, thus suggesting a major deletion in the O gene. Therefore, inadvertent activation of a silent O gene seems unlikely in porcine organs xenotransplanted into humans.
Introduction
The immunodominant structures of A and B antigens are defined as N-acetyl-D-galactosamine (GalNAc) α1 → 3 (Fuc α1→2) Gal- and Gal α1 → 3 (Fuc α1 → 2) Gal-, respectively. The blood group A gene encodes A transferase, which transfers GalNAc to the galactose residue of the acceptor H substrates (Fuc α1 → 2 Gal-), whereas the B gene encodes B transferase, which transfers galactose to the same substrates. Group O genes are nonfunctional, and H substrates in group O individuals remain without additional modifications.
For the past decade, we have been studying the genes that specify the histo-blood group ABO polymorphism.1 On the basis of the partial amino acid sequence of the soluble form of A transferase,2 we cloned human A transferase complementary DNAs (cDNAs).3 Subsequently, we cloned B transferase cDNAs and nonfunctional O allelic cDNAs and elucidated the molecular genetic basis of the human ABO locus.4 Four amino acid substitutions were identified between A transferase and B transferase. Most O alleles were found to contain a single base deletion near the N-terminus of the coding sequence. In rare cases in which O alleles were missing the single base deletion, an amino acid substitution was found at a residue crucial for nucleotide-sugar recognition and binding.5 In addition to the 3 major alleles, we identified mutations in subgroup alleles and in cisAB and B(A) alleles that specify the synthesis of both A and B antigens.1 The updated list of ABO alleles is available at the Blood Group Antigen Gene Mutation Database Web site (http://www.bioc.aecom.yu.edu/bgmut/index.htm).
ABH antigens are not unique to humans; rather, they are widely present in nature.6 Pigs, for example, have A and H.7,8 Using a human A gene probe, we observed strong hybridization signals in genomic DNAs from various mammals.9 We also determined partial nucleotide sequences of ABO genes from several primate species and observed that conservation of amino acid residues critical for nucleotide-sugar substrate recognition and binding in monkeys was dependent on their ABO genotypes. We have now extended our ABO studies to other mammals. Because pigs are primary candidates for organ donors for humans, characterization of porcine O gene deficiency has both clinical and scientific importance. We here report the molecular genetic basis of the porcine AO system.
Study design
This study used polymerase chain reaction (PCR) amplification of porcine AO gene fragments. The AO phenotypes of 33 porcine submaxillary glands were immunologically determined by examining A antigen expression with use of murine monoclonal antibody mixtures (Ortho, Raritan, NJ), avidin-biotin complex reagents, and 3, 3′-diaminobenzidine substrates (Vector, Burlingame, CA). A transferase activity of submaxillary gland extracts was measured by transfer of carbon 14 (14C) from 14C–uridine diphosphate–GalNAc to the acceptor substrate 2′-fucosyllactose as described previously.10 Genomic DNA was prepared from representative group A and O pig glands. DNA fragments were amplified by using FY-520 (5′-CCGGAATTCAACACTTCATGGTGGGACAC) and FY-521 (CCGGAATTCTAGCTCTCATCATGCCACAC), which are 2 primers that correspond to sequences conserved between human ABO genes (AF134412), and recently cloned murine genes (AB041039; M.Y. et al, unpublished data, 2001). Amplified DNA fragments were digested with EcoRI and cloned into pT7T3α18 vector. The nucleotide sequences were then determined.
Results and discussion
Of the 33 porcine submaxillary glands, 31 were group A and 2 were group O. A transferase activity was detected only in the glands positive for A antigen. Nucleotide sequencing revealed that 2 sequences were amplified from the group A genomic DNA. One sequence (AB041040) showed high homology to human and murine ABO genes. A search of GenBank with the Basic Local Alignment Search Tool database identified the entry AF050177, which appears to contain the complete coding sequence of the putative A transferase. We cloned the sequence by reverse transcriptase–PCR using poly A+ RNA from a group A submaxillary gland and constructed pPigA expression constructs in sense and antisense orientations. DNA from these constructs was then used for DNA transfection assays. Both A antigen expression and A transferase activity were observed with the sense construct (Table1). Therefore, we concluded that the sequence encoded A transferase.
Cell-surface A and B antigen expression and A and B transferase activity in the extracts of HeLa cells transfected with eukaryotic expression constructs
| Construct . | A antigen expression . | A transferase activity . | B antigen expression . | B transferase activity . | Summary . |
|---|---|---|---|---|---|
| No DNA | − | − | − | − | − |
| pA(arg) | − | − | − | − | − |
| pAAAA | +++ | +++++ | − | − | A |
| pBBBB | − | − | +++ | +++ | B |
| pPigA (s) | +++ | +++ | − | − | A |
| pPigA (as) | − | − | − | − | − |
| Construct . | A antigen expression . | A transferase activity . | B antigen expression . | B transferase activity . | Summary . |
|---|---|---|---|---|---|
| No DNA | − | − | − | − | − |
| pA(arg) | − | − | − | − | − |
| pAAAA | +++ | +++++ | − | − | A |
| pBBBB | − | − | +++ | +++ | B |
| pPigA (s) | +++ | +++ | − | − | A |
| pPigA (as) | − | − | − | − | − |
Poly A+ RNA was prepared from group A porcine submaxillary glands and used for RT-PCR of a putative porcine A transferase complementary DNA using 2 primers: FY-530 (5′-CTCCAGGCACCTGGCTTG) and FY-531 (5′-CCCTCCTCCTGTTCGTCG). The sequences in these primers corresponded to the sequences in the 5′ UTR and the 3′ UTR regions of the complementary DNA. The PCR product was cloned into pT-Adv vector by using the T-A cloning method and subsequently transferred into the pSG-5 eukaryotic expression vector. DNA from the pPigA expression constructs was used for the Lipofectamine (Gibco, Grand Island, NY)-mediated DNA transfection assays. The human uterus–derived adenocarcinoma cell line HeLa was the recipient, as described previously.10 11 Human A and B transferase expression constructs pAAAA and pBBBB were used as positive controls, and nonfunctional pA(arg) construct was used as a negative control. Two days after transfection, some floating cells were plated into a 96-well plate. The next day, cells were fixed and immunostained with anti-A and anti-B murine monoclonal antibody mixtures, avidin-biotin complex reagents, and 3,3′-diaminobenzidine substrates. The cells from the original plates were harvested and lysed, and A and B transferase activity was determined by measuring the transfer of 14C from 14C-UDP-N-acetyl-D-galactosamine and 14C-UDP-galactose, respectively, to the acceptor substrate 2′-fucosyllactose. The reactions without 2′-fucosyllactose were used as negative controls. A summary of the activity of the constructs is shown in the last column.
RT-PCR indicates reverse transcriptase–polymerase chain reaction; UTR, untranslated region; UDP, uridine diphosphate.
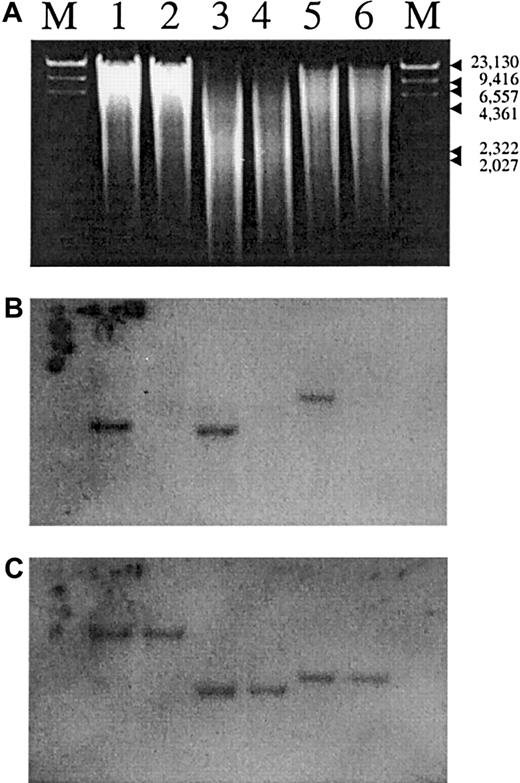
The other amplified sequence was also homologous to the ABO genes but to a lesser degree. This sequence had greater homology to the human hgt4 pseudogene sequence12 (data not shown). When genomic DNA from a group O pig was used as a PCR template, however, only the pseudogene fragment was amplified. Because amplification failure may have resulted from mismatch of primer sequences, we did additional evaluations using Southern hybridization. As shown in Figure1B, no corresponding bands were hybridized in the group O pig DNA with the porcine A gene coding-sequence probe. We analyzed 2 group O pigs and obtained the same results (data not shown). Therefore, the results showed that most of the coding region was missing in the porcine O gene.
Southern hybridization experiments.
Genomic DNA from group A (lanes 1, 3, and 5) and group O (lanes 2, 4, and 6) porcine submaxillary glands was digested with EcoRI (lanes 1 and 2) alone or BamHI (lanes 5 and 6) alone or with both EcoRI and BamHI (lanes 3 and 4) and electrophoresed through a 1% agarose gel. DNA was then transferred by using the Southern method to a nylon membrane and hybridized in Ultrahyb hybridization buffer (Ambion, Austin, TX) with a cloned porcine A gene fragment probe, which was radiolabeled by using the random-hexamer method. Hybridization was done at 42°C overnight. Subsequently, the filter was washed twice with 2 × standard saline citrate (SSC) and 0.1% sodium dodecyl sulfate (SDS) at 42°C for 10 minutes. This was followed by another washing with 0.1 × SSC and 0.1% SDS at 42°C for 15 minutes. The filter was then exposed to x-ray film. The same filter was later hybridized with a radiolabeled porcine α 1-3 galactosyltransferase gene sequence probe.13 Figure 1A shows the results of electrophoresis, and Figures 1B and 1C show the results of hybridization experiments with the porcine A gene probe (B) and the porcine α 1 → 3 galactosyltransferase gene probe (C). The very weak bands common to the A and O pig DNA may have resulted from cross-hybridization of the A gene probe with the sequence homologous to the human hgt4 pseudogene.11
Southern hybridization experiments.
Genomic DNA from group A (lanes 1, 3, and 5) and group O (lanes 2, 4, and 6) porcine submaxillary glands was digested with EcoRI (lanes 1 and 2) alone or BamHI (lanes 5 and 6) alone or with both EcoRI and BamHI (lanes 3 and 4) and electrophoresed through a 1% agarose gel. DNA was then transferred by using the Southern method to a nylon membrane and hybridized in Ultrahyb hybridization buffer (Ambion, Austin, TX) with a cloned porcine A gene fragment probe, which was radiolabeled by using the random-hexamer method. Hybridization was done at 42°C overnight. Subsequently, the filter was washed twice with 2 × standard saline citrate (SSC) and 0.1% sodium dodecyl sulfate (SDS) at 42°C for 10 minutes. This was followed by another washing with 0.1 × SSC and 0.1% SDS at 42°C for 15 minutes. The filter was then exposed to x-ray film. The same filter was later hybridized with a radiolabeled porcine α 1-3 galactosyltransferase gene sequence probe.13 Figure 1A shows the results of electrophoresis, and Figures 1B and 1C show the results of hybridization experiments with the porcine A gene probe (B) and the porcine α 1 → 3 galactosyltransferase gene probe (C). The very weak bands common to the A and O pig DNA may have resulted from cross-hybridization of the A gene probe with the sequence homologous to the human hgt4 pseudogene.11
We then hybridized the membrane with the α 1-3 galactosyltransferase gene coding-sequence probe. The α 1-3 galactosyltransferase specifies the α1 → 3 Gal epitope that is expressed on various porcine cells, including erythrocytes and aortic endothelial cells. Human immune systems launch an immediate hyperacute rejection of cells expressing this epitope.14 The gene that encodes the enzyme is a member of the ABO gene family,11 and the porcine α 1-3 galactosyltransferase gene is mapped on chromosome 1q2.10 to 2.11, which is homologous to the human chromosome 9q34, where the ABO locus resides.13 The presence of the α 1-3 galactosyltransferase gene in the group O pig DNA (Figure 1C) indicated that the deletion does not extend to the α 1-3 galactosyltransferase gene locus.
In spite of concern that porcine viruses could enter human cells and cause disease or recombine with other viral sequences to create a new virus,15 xenotransplantation of porcine organs into patients with chronic organ failure has been considered an option for overcoming the shortage of human organs for transplantation. Successful pig cloning16,17 may help to reduce the risk of pig xenotransplantation. With the aim of developing pigs suitable for organ supply, attempts have been made to modify α1 → 3 Gal epitope synthesis. These efforts include knockout of the α 1-3 galactosyltransferase gene in mice (but not yet in pigs)18and production of transgenic pigs with the introduced human α 1-2 fucosyltransferase gene.19 Because the biosynthetic pathways of carbohydrate structures of glycolipids and glycoproteins are complex and interrelated, manipulation of α 1-3 galactosyltransferase alone may not be sufficient. Pigs are negative for the Forssman antigen, and the inactivating mechanism of the Forssman glycolipid synthetase gene is still unknown.
In a phenomenon called incompatible A expression,20,21expression of A antigen is observed in tumors in individuals with blood group B or O, who do not normally produce A antigens. The molecular mechanism underlining this aberrant expression of A antigen is unknown; however, the appearance of not only A antigens but also A transferase activity22 suggests that the messages encoding functional A transferase were produced by premature splicing or some other mechanism. In this study, we found evidence of a major deletion in the porcine O gene. In contrast to what may occur with human O genes, it is unlikely that the dormant O gene would be fortuitously activated in porcine organs xenotransplanted in humans, since most of the coding sequence is missing in the porcine O gene.
We thank Dr Sandra S. Matsumoto for critically reading the manuscript and Ami Yamamoto for editing the manuscript.
Supported in part by funds from the Burnham Institute and the Department of Defense Breast Cancer Research Program (DAMD 17-98-1-8168).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Fumiichiro Yamamoto, Burnham Institute, La Jolla Cancer Research Center, 10901 N Torrey Pines Rd, La Jolla, CA 92037; e-mail: fyamamoto@burnham.org.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal