Efficient antigen presentation and T-cell priming are essential components of effective antitumor immunity. Dendritic cells are critical to both of these functions but to date no method has been devised that both targets antigen to these cells and activates them, in situ, in a manner that induces systemic immunity. In this study we combined a dendritic cell growth factor, Flt3 ligand, with a dendritic cell activator, immunostimulatory DNA, and a tumor antigen to activate and load dendritic cells in vivo. Initial studies showed that immunostimulatory DNA not only activates dendritic cells but also prolongs their survival in vivo and in vitro. Following treatment of mice with Flt3 ligand, coadministration of immunostimulatory DNA and antigen induced potent antitumor immunity, resulting in both tumor prevention and regression of existing tumors. CD8 cytotoxic T lymphocytes but not CD4 T cells were required for tumor protection. Natural killer cells also contributed to tumor protection. These results show that dendritic cells can be loaded with antigen and activated, in situ, and provide the basis for dendritic cell- targeted clinical strategies.
Introduction
Dendritic cells (DCs) represent the most potent antigen-presenting cells (APCs) capable of inducing immunity to newly introduced antigen (Ag).1,2 DCs reside as immature cells in peripheral tissues where they continuously take up and process Ag. When activated, Ag-loaded DCs migrate to the draining lymph nodes where they can prime Ag-specific CD4 and CD8 T cells. Multiple factors contribute to the failure of DCs to prime effective antitumor response in tumor-bearing hosts: the low number of DCs available in the tumor site, poor access of DCs to tumor Ag, the limited capacity of tumor cells to activate intratumoral DCs,3-5 and secretion by the tumor cells of factors that inhibit DC maturation.6-8Interestingly, in these studies T cells isolated from tumor-bearing hosts were able to respond normally to various Ags in vitro if stimulated with mature, functional DCs.7,9 Moreover, professional APCs such as DCs or tumor cells10 engineered to stimulate immunity can generate an effective antitumor immune response in vivo,11 12 suggesting again that efficient Ag presentation is critical in tumor immunotherapy.
Dendritic cells can be used as therapeutic cancer vaccines.13 A variety of preparations of DCs can stimulate antitumor immunity, including DCs loaded with proteins, DCs fused with tumor cells, and DCs transduced with tumor-derived RNA or viral vectors. At present, all of these approaches rely on ex vivo manipulation of isolated DCs to produce the vaccine. Recently, administration of a bone marrow growth factor, Flt3 ligand (FL), has been shown to expand in vivo the numbers of DCs in lymph nodes, spleen, and other tissues.14 However, a significant proportion of the FL-mobilized DCs are immature and not efficient in inducing Ag-specific T-cell responses. We therefore focused on developing an approach to activate these mobilized DCs in situ, with the goal of determining whether such an approach can induce effective Ag-specific immunity.
Although various DC maturation factors have been described (eg, tumor necrosis factor-α [TNF-α], lipopolysaccharide [LPS]), the majority of these are associated with toxicity that precludes their use in vivo. However, certain CpG-containing DNA sequences (ISS), which have recently been shown to activate DCs by binding to a specific Toll-like receptor,15 are well tolerated when administered in vivo. In the studies described in this report we demonstrate that ISS can activate and prolong the survival of DCs in vitro and in vivo. Moreover, in mice pretreated with FL, the simultaneous delivery of a tumor Ag and ISS generates Ag-loaded DCs in situ that can induce potent antitumor immunity.
Materials and methods
Animals
Five- to 7-week-old female C57BL/6 and Balb/c mice were purchased from the Jackson Laboratory (Bar Harbor, ME). The 1100Mjb mice express an H-2Kb restricted T-cell receptor (TCR) specific for the octamer peptide ovalbumin257-264.16 All mice were housed in our animal facility.
Cytokines and media
Recombinant human FL, murine granulocyte-macrophage colony-stimulating factor (GM-CSF) and interleukin-4 (IL-4) were purchased from Peprotech (Rocky Hill, NJ). Cytokines were reconstituted in phosphate-buffered saline (PBS). Cells were cultured in vitro in the presence of complete medium (CM), which included RPMI supplemented with 10% fetal calf serum (FCS), penicillin G (100 U/mL), and streptomycin (100 μg/mL). Cell staining and sorting were performed in PBS with 3% FCS and 0.1% azide.
Ag and adjuvants
Ovalbumin protein (OVA) was purchased from Sigma (St Louis, MO). Endotoxin contamination of OVA was removed by a phase separation technique using the detergent Triton X-114 (kindly provided by Dr J. Timmerman, Stanford University, Stanford, CA) such that endotoxin levels were undetectable by limulus amebocyte lysate assay. Complete Freund adjuvant (CFA) was purchased from DIFCO (Detroit, MI). Phosphorothioate-stabilized oligonucleotides were synthesized by Oligos Etc. (Wilsonville, OR). The oligonucleotide sequence bearing ISS was TCCATGACGTTCCTGACGTT; the sequence of the non-ISS control oligonucleotide (C-ODN) was TCCAGGACTTTCCTCAGGTT. All oligonucleotides were reconstituted in sterile pyrogen-free water.
Antibodies and flow cytometry
Cell preparations were preincubated with anti-CD32/16 to minimize nonspecific binding. Three-color analyses were performed on a FACSCalibur (Becton Dickinson, Mountain View, CA). Mouse monoclonal antibodies (MoAbs) to IA-b (Af6-120.1; IgG2a), B-220 (RA3-6B2; IgG2a), CD3ε (145-2C11; IgG1), CD4 (GK1.5; IgG2b), CD8 (Ly-2; IgG2a), CD11c (HL3; IgG1), CD16/CD32 (2.4G2; IgG2b), CD28 (37.51; IgG2), CD54 (3E2, IgG1), CD80 (16-10A1, IgG2), CD86 (GL1; IgG2a), IL-2 (JES6-5H4, IgG), interferon-γ (IFN-γ; XMG1.2, IgG), leukocyte function-associated antigen 1 (LFA-1) (2D7, IgG2a), natural killer 1.1 (NK1.1) (PK136, IgG2a), T-cell receptorβ (TCRβ) chain (H57-597; IgG2), isotype controls, and the second-step antibodies (APC-conjugated streptavidin, phycoerythrin [PE]-conjugated goat anti–rabbit IgG) were purchased from Pharmingen (San Diego, CA).
Tumor cell lines
B16-OVA is an OVA-transfected clone derived from the murine melanoma cell line B16.17 The tumor cell line was cultured in vitro in CM in the presence of geniticin (2 mg/mL) and hygromicin B (60 μg/mL). The lymphoma tumor cell line EL4 was purchased from the American Type Tissue Collection (ATCC, Rockville, MD). EG7 is an OVA-transfected subclone of EL-4,18 kindly provided by Dr M. Bevan (University of Washington, Seattle, WA), and was cultured in CM in the presence of geniticin (2 mg/mL).
Immunization protocols
FL (10 μg in 200 μL PBS) was administered intraperitoneally (IP) daily for 9 days. Control mice were injected in the same manner with PBS. In addition to the last FL injection, mice received where indicated, a single subcutaneous (SC) injection of OVA (300 μg), ISS (30 μg), ISS or C-ODN mixed with OVA (300 μg), or CFA mixed with OVA (300 μg).
Isolation of secondary lymphoid organs
Spleen and draining popliteal and inguinal lymph nodes were harvested and injected with collagenase D (1 mg/mL; Boehringer-Mannheim, Mannheim, Germany) in RPMI and 10% FCS for 20 minutes at 37°C. Digested lymph nodes or spleen were filtered through a stainless-steel sieve, and the cell suspension washed twice in PBS and 5% FCS. Where indicated DCs were isolated from the total lymph nodes or spleen cells using anti-CD11c-microbeads (Miltenyi Biotech, Auburn, CA). The purity of the DC population after sorting was more than 98% based on their coexpression of class II and CD11c antigens. Where indicated CD11c+ cells were cultured overnight in CM in addition to GM-CSF (10 ng/mL) in the presence or absence of ISS (10 μg/mL) or C-ODN (10 μg/mL).
Cytokine profile of ISS-activated DCs in vitro
Dendritic cells were isolated from the spleen of FL-treated mice using anti-CD11c magnetic beads as described above and cultured overnight in CM in addition to GM-CSF (10 ng/mL) in the presence or absence of ISS (10 μg/mL) or C-ODN (10 μg/mL). At the end of the culture, cytokine (IL-12, IFN-γ, and TNF-α) secretion in the supernatant was analyzed by enzyme-linked immunosorbent assay (ELISA).
Allogeneic T-cell stimulation
Graded numbers of DCs isolated from the lymph nodes of immunized or nonimmunized C57BL/6 mice were irradiated (3000 rads), and added to allogeneic (Balb/c) spleen cells (2 × 105 /well) in a final volume of 0.2 mL in 96-well flat-bottom plates (Costar, Corning, NY). IFN-γ in the supernatant was measured by ELISA after 4 days of coculture. Cell proliferation was measured by adding 1 μCi (0.037 MBq) [3H]-thymidine after 3 days. The cells were harvested 16 to 18 hours later and counted in a Microbeta counter (Wallac, Gaithersburg, MD). Results are presented as the mean of triplicate cultures ± SEM.
Natural killer cell activation
NK cells were isolated from the spleen using magnetic beads. Briefly, spleen cells were stained with biotin-conjugated MoAb to NK1.1 and labeled cells were then selected with streptavidin microbeads. Two rounds of positive selection were applied to ensure more than 98% NK cell purity. Purified FL mobilized DCs were isolated from spleen and cultured overnight in the presence or absence of ISS (10 μg/mL) or C-ODN (10 μg/mL). The DCs were then cultured in the presence of NK1.1+ cells at a ratio of 1:1 for 24 hours. In parallel, NK1.1+ cells were cultured in the presence of ISS (10 μg/mL), C-ODN (10 μg/mL), or medium alone for 24 hours. The cultured NK cells were incubated at different ratios with51Cr-labeled YAC-1 cells for 4 hours before measuring their cytolytic activity based on a 51Cr release assay.
DC viability assay
A modification of the protocol of Inaba et al19 was used to generate bone marrow-derived DCs (BM-DCs). Briefly, BM cells were incubated at 1 × 105 cells/mL in CM in the presence of GM-CSF (5 ng/mL), and IL-4 (5 ng/mL). The supernatant was removed at day 2 and replaced with fresh medium and cytokines. At day 4 the cells were centrifuged and resuspended in fresh medium and cytokines for 2 to 3 more days. At the end of the culture CD11c+ DCs were isolated using anti-CD11c-microbeads, washed, and cultured in CM without any additional cytokines in the presence or absence of 10 μg/mL ISS or C-ODN. One, 4, and 7 days later, cell proliferation was measured by adding 1 μCi (0.037 MBq) [3H]-thymidine as described above. CD11c+ DCs and BM cells cultured for 4 days in GM-CSF and IL-4 were used as controls for cell proliferation assays. Cell numbers were counted daily using trypan blue exclusion
Lymph node cell activation after Ag restimulation in vitro
C57BL/6 mice were immunized with FL (9 daily injections) in addition to OVA (300 μg) alone or in association with ISS or C-ODN (30 μg). Control groups were immunized with OVA (300 μg) alone. At 7, 12, and 18 days after immunization, cells isolated from the draining lymph nodes were restimulated in the presence or absence of ovalbumin257-264 (10 μg/mL) and anti-CD28 (1 μg/mL) for 8 hours in addition to brefeldin (10 μg/mL; Sigma) and analyzed for intracellular cytokine secretion by flow cytometry. Simultaneously, lymph node cells were cultured in the presence of different concentrations of OVA and cell proliferation was measured by adding at day 3, 1 μCi (0.037 MBq) [3H]-thymidine. At 16 to 18 hours later, cells were harvested and the incorporated thymidine counted in a Microbeta counter.
Cytokine measurements
Cytokines present in the supernatant were quantified by ELISA as described elsewhere.20 For intracellular cytokine detection, cells were stained for surface membrane Ag as described above, washed extensively, and then permeabilized using Cytofix/Cytoperm (Pharmingen) for 20 minutes on ice. Permeabilized cells were resuspended in Perm Wash buffer (Pharmingen) and stained with anti–IFN-γ or anti–IL-2 antibodies.
Tumor experiments
Tumor cells (1 × 104) in 0.1 mL PBS were injected SC into mice. In the tumor protection experiments, mice received a tumor challenge 1 week after the completion of the therapy. To explore whether the treatment had induced a memory response against melanoma cells, mice that were still alive 6 weeks following the tumor challenge were rechallenged SC with the same dose of tumor cells without any further treatment. In the tumor therapy experiments, mice were first challenged with tumor cells and received the first FL injection 3 days later followed by immunization with ISS or C-ODN (30 μg) and OVA (300 μg). Mice were examined twice a week for the presence of tumors. Tumor size represents the product of 2 perpendicular diameters. As per protocol, mice were killed when tumors reached 20 mm in their largest dimension or when ulceration or bleeding or both developed.
Tumor experiments in lymphocyte-depleted mice
Following the same treatment schedule described in the tumor protection experiment, groups of 8 mice were injected IP with 0.5 mg anti-CD4 (GK1.5), anti-CD8 (2.43), or anti-NK cell (PK136) antibody 3 times at 2-day intervals and then weekly for the duration of the experiment. Where indicated anti-CD4 antibody injection was started 6 days before the first FL injection, or 24 hours after immunization with ISS plus Ag. Anti-CD8 antibody and NK antibody treatments started 24 hours after immunization. Lymphocyte depletion was confirmed in each depletion experiment by FACS analysis of peripheral blood 10 and 30 days following antibody treatment and showed less than 0.2% CD4+ cells, less than 0.02% CD8+ cells, and less than 0.3% NK+ cells, respectively. In mice treated with anti-CD8 there was no change in the number of IA-b+, CD11c+, DEC205hi, and CD11blowcells in the spleen and lymph nodes, suggesting that CD8+DCs were not affected by the anti-CD8 antibody.
Statistical analyses
The Kaplan-Meier plot for tumor survival was assessed for significance with the Mantel-Cox test (StatView, SAS Institute, Cary, NC).
Results
ISS can activate FL-mobilized DCs in situ
Although FL administration can expand DCs in vivo, most of these DCs are not fully mature. We evaluated the capacity of ISS to activate FL-mobilized DCs (FL-DCs) in vitro. After 9 days of FL administration to mice, CD11c+ DCs were harvested from spleen and cultured for 18 hours with or without ISS. ISS-treated CD11c+ DCs had an increase in their surface expression of major histocompatibility complex (MHC) class II molecules, as well as the costimulatory molecules CD80 and CD86 and the adhesion molecule CD54 (Figure1A), and secreted higher levels of IL-12 and IFN-γ compared to C-ODN–treated DCs and unstimulated DCs (Figure1B). ISS administration also increased the immunostimulatory capacity of DCs as shown by their ability to induce allogeneic T-cell proliferation and IFN-γ secretion compared to DCs obtained from untreated animals or animals treated with FL either alone or in association with C-ODN (Figure 1C-D). Because ISS was initially described as a strong NK cell activator, we explored the effect of ISS as well as ISS-treated FL-DCs on a highly purified population of NK cells. As shown in Figure 1E, ISS did not activate NK cells directly but dramatically enhanced the capacity of FL-DCs to activate these cells.
ISS induces activation of FL-DCs.
(A,B) CD11c+ cells isolated from the spleens of FL-treated mice were incubated overnight in CM and GM-CSF. (A) Histograms show the coexpression of IA-b, CD80, CD86, and CD54 on CD11c+ gated cells cultured in the presence (dotted line) or absence (black line) of ISS (10 μg/mL). Results shown are representative of 6 different experiments. (B) Cytokine profile of DCs treated with ISS (10 μg/mL) and C-ODN (10 μg/mL) compared to untreated DCs. These results represent the mean of 3 different experiments. (C,D) CD11c+DCs isolated from the popliteal and inguinal lymph nodes of FL-ISS (●), FL–C-ODN (○), FL alone (▴), or untreated animals (▵) were irradiated (3000 rads) and cultured with 3 × 105allogeneic spleen cells. ⋄ indicates DC alone; ♦, T cell alone. Cell proliferation (C) and IFN-γ secretion in the supernatant (D) of the DC/spleen cell coculture were measured after 4 days of culture. These results are the mean of 3 different experiments. (E) Purified spleen-derived NK cells were cultured in the presence of ISS (▴), C-ODN (▵), or IL-12 (●), or in the presence of ISS-treated FL-DCs (▪), C-ODN–treated FL-DCs (■) or untreated FL-DCs (♦). After 24 hours of culture, viable lymphocytes were tested against the YAC-1 cell line in an immunocytotoxicity assay. These data represent the mean of 3 different experiments.
ISS induces activation of FL-DCs.
(A,B) CD11c+ cells isolated from the spleens of FL-treated mice were incubated overnight in CM and GM-CSF. (A) Histograms show the coexpression of IA-b, CD80, CD86, and CD54 on CD11c+ gated cells cultured in the presence (dotted line) or absence (black line) of ISS (10 μg/mL). Results shown are representative of 6 different experiments. (B) Cytokine profile of DCs treated with ISS (10 μg/mL) and C-ODN (10 μg/mL) compared to untreated DCs. These results represent the mean of 3 different experiments. (C,D) CD11c+DCs isolated from the popliteal and inguinal lymph nodes of FL-ISS (●), FL–C-ODN (○), FL alone (▴), or untreated animals (▵) were irradiated (3000 rads) and cultured with 3 × 105allogeneic spleen cells. ⋄ indicates DC alone; ♦, T cell alone. Cell proliferation (C) and IFN-γ secretion in the supernatant (D) of the DC/spleen cell coculture were measured after 4 days of culture. These results are the mean of 3 different experiments. (E) Purified spleen-derived NK cells were cultured in the presence of ISS (▴), C-ODN (▵), or IL-12 (●), or in the presence of ISS-treated FL-DCs (▪), C-ODN–treated FL-DCs (■) or untreated FL-DCs (♦). After 24 hours of culture, viable lymphocytes were tested against the YAC-1 cell line in an immunocytotoxicity assay. These data represent the mean of 3 different experiments.
To test the ability of ISS to activate FL-DCs in vivo, mice were injected with FL for 9 consecutive days and, on the final day, were also given a single SC injection of either ISS, a similar oligonucleotide (C-ODN) that lacks the CpG motif, or PBS. Forty-eight hours later, DCs within the draining lymph nodes were assessed phenotypically. The percentage of mature DCs expressing high levels of MHC class II molecules and costimulatory molecules such as CD86 was higher in mice treated with both FL and ISS than mice treated with FL alone (Figure 2A). The effect of ISS on FL-DC maturation was localized to the draining lymph nodes because the phenotype of DCs in the contralateral lymph node was unaffected (data not shown).
ISS increases the number of mature DCs in FL-treated animals and prolongs the survival of mature DC in vitro.
(A,B) Groups of 3 mice were treated with 9 daily injections of FL followed by one SC injection of ISS (30 μg), C-ODN (30μg), or PBS (FL alone). Forty-eight hours after ISS injection, popliteal and inguinal lymph node cells were isolated and analyzed by flow cytometry. In panel A the contour plots represent IA-b versus CD86 profiles of CD11c+ gated cells. The percentages of CD11c+cells that are IA-b−CD86− and IA-b+CD86+ are provided in each quadrant. In panel B the number of cells coexpressing IA-b and CD11c was determined on days 1, 7, 12, and 18 after treatment. ■ indicates PBS; ▪, FL-ISS; ▴ FL–C-ODN; ▵, FL alone. (C) The number of IA-b+ CD11c+ cells coexpressing CD86, CD54, or LFA-1 was determined on day 7 after treatment. These results are the mean of 4 different experiments. ■ indicates PBS; ▪, FL-ISS; ▨, FL–C-ODN; ░, FL alone. (D) BM-DCs were cultured for 11 days in the presence or absence of 10 μg/mL ISS (●), C-ODN (▴), or medium alone (♦). Viable DCs were counted using trypan blue exclusion. Results shown are the mean of 3 separate experiments.
ISS increases the number of mature DCs in FL-treated animals and prolongs the survival of mature DC in vitro.
(A,B) Groups of 3 mice were treated with 9 daily injections of FL followed by one SC injection of ISS (30 μg), C-ODN (30μg), or PBS (FL alone). Forty-eight hours after ISS injection, popliteal and inguinal lymph node cells were isolated and analyzed by flow cytometry. In panel A the contour plots represent IA-b versus CD86 profiles of CD11c+ gated cells. The percentages of CD11c+cells that are IA-b−CD86− and IA-b+CD86+ are provided in each quadrant. In panel B the number of cells coexpressing IA-b and CD11c was determined on days 1, 7, 12, and 18 after treatment. ■ indicates PBS; ▪, FL-ISS; ▴ FL–C-ODN; ▵, FL alone. (C) The number of IA-b+ CD11c+ cells coexpressing CD86, CD54, or LFA-1 was determined on day 7 after treatment. These results are the mean of 4 different experiments. ■ indicates PBS; ▪, FL-ISS; ▨, FL–C-ODN; ░, FL alone. (D) BM-DCs were cultured for 11 days in the presence or absence of 10 μg/mL ISS (●), C-ODN (▴), or medium alone (♦). Viable DCs were counted using trypan blue exclusion. Results shown are the mean of 3 separate experiments.
To determine the duration of the ISS effect, DCs within the draining lymph nodes were enumerated and analyzed by flow cytometry at 1, 7, 12, and 18 days after treatment. At day 1, the number of DCs was similar in all FL-treated groups (Figure 2B). In contrast, at days 7, 12, and 18, the number of DCs was 39, 14, and 7 times greater in mice treated with FL and ISS, respectively, compared to PBS-treated mice. In animals treated with FL alone the number of DCs was only 6 times above baseline at day 7 and returned to baseline by day 12. Furthermore, the number of DCs that expressed high levels of MHC class II, CD86, CD54, and LFA-1 was significantly greater at day 7 in mice treated with FL and ISS compared to mice treated with FL alone (Figure 2C). This effect persisted for more than 18 days after treatment (data not shown).
Additional studies suggested that the persistence of DCs in ISS-treated animals may be due to a direct effect of ISS on DC viability (Figure 2D). Thus, when purified DCs were cultured in the absence of ISS, the vast majority died by day 5. In contrast, more than 50% of ISS-activated DCs were still alive on day 11. This effect was not due to an increase of cellular proliferation induced by ISS, because proliferation analyzed at days 1, 4, and 7 was similar in ISS, C-ODN, and untreated cells (data not shown).
ISS increases CD4 and CD8 T-cell responses to Ag in vivo in FL-treated mice
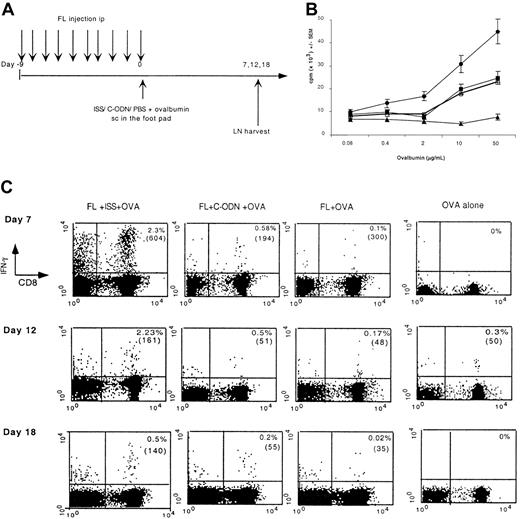
Because ISS activates DCs in vitro and in vivo, we sought to determine whether ISS could increase the capacity of FL-DCs to prime T-cell immunity by injecting a mixture of Ag (OVA) and ISS into FL-treated mice (Figure 3A). The combined treatment was well tolerated and no sign of clinical toxicity or autoimmunity was observed. As shown in Figure 3, panels B and C, the combination of DC mobilization with DC activation at the site of Ag administration dramatically enhanced the induction of both CD4 and CD8 T-cell immunity, based on measurement of lymph node cell proliferation in response to soluble OVA and IFN-γ secretion by CD8 T cells. OVA-specific CD8 T cells could be detected in draining lymph nodes more than 3 weeks after the immunization.
ISS increases T-cell priming in FL-treated mice.
(A) Experimental design. Groups of 3 mice received daily injections of FL or PBS from day −9 to day 0. OVA in association with ISS, C-ODN, or PBS was injected SC on day 0. Seven, 12, and 18 days after immunization the draining popliteal and inguinal lymph nodes were harvested and assayed. (B) Seven days after immunization, lymph node cells were cultured for 4 days in the presence of graded doses of OVA and proliferation was measured. Results shown represent the mean of 4 separate experiments. ● indicates FL-ISS-OVA; ▪, FL–C-ODN–OVA; ■, FL-OVA; ▴, OVA alone. (C) Lymph node cells isolated 7, 12, and 18 days after immunization were restimulated in the presence of OVA257-264 and anti-CD28 for 8 hours and then assayed for IFN-γ secretion by flow cytometry. The percentage of lymph node cells that are CD8+ IA-b− IFN-γ+ is provided in the appropriate quadrant and the mean IFN-γ fluorescence intensity is in brackets. These results represent the mean of 3 different experiments.
ISS increases T-cell priming in FL-treated mice.
(A) Experimental design. Groups of 3 mice received daily injections of FL or PBS from day −9 to day 0. OVA in association with ISS, C-ODN, or PBS was injected SC on day 0. Seven, 12, and 18 days after immunization the draining popliteal and inguinal lymph nodes were harvested and assayed. (B) Seven days after immunization, lymph node cells were cultured for 4 days in the presence of graded doses of OVA and proliferation was measured. Results shown represent the mean of 4 separate experiments. ● indicates FL-ISS-OVA; ▪, FL–C-ODN–OVA; ■, FL-OVA; ▴, OVA alone. (C) Lymph node cells isolated 7, 12, and 18 days after immunization were restimulated in the presence of OVA257-264 and anti-CD28 for 8 hours and then assayed for IFN-γ secretion by flow cytometry. The percentage of lymph node cells that are CD8+ IA-b− IFN-γ+ is provided in the appropriate quadrant and the mean IFN-γ fluorescence intensity is in brackets. These results represent the mean of 3 different experiments.
Ag loading and ISS activation of FL-mobilized DCs induce tumor-specific immunity
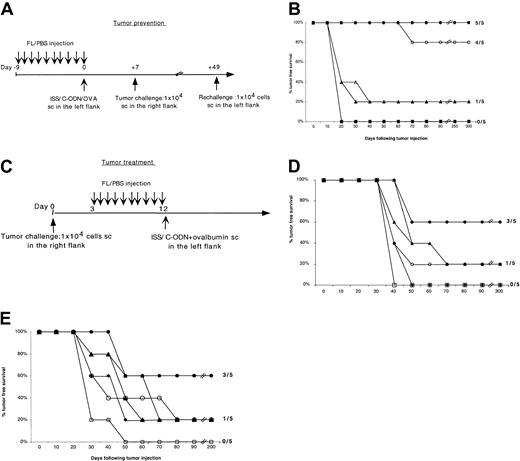
Next, we explored whether the enhanced immunity induced with this combined treatment could protect mice against a lethal tumor challenge (Figure 4A). As shown in Figure 4B, 100% of the mice treated with FL plus ISS plus OVA survived without developing any tumors compared to only 20% of mice treated with FL plus OVA alone or FL plus OVA plus C-ODN (Figure 4B). None of the mice treated with OVA plus ISS without FL, with OVA plus CFA, or with OVA alone survived the tumor challenge (Figure 4B and data not shown). Also, 80% of the mice that were initially treated with FL, ISS, and OVA survived a second tumor challenge without any additional immunization indicating that a memory antitumor immune response had been induced by the first treatment (Figure 4B).
Ag loading and ISS activation of FL-mobilized DCs induce tumor protection and regression of established tumors.
(A,C) Experimental design. Groups of 5 mice were immunized with 9 daily injections of FL followed by 1 SC injection of OVA mixed with ISS, C-ODN, or PBS on day 0. The mice were challenged with one SC injection of 1 × 104 B16-OVA melanoma tumor cells in the opposite flank 1 and 7 weeks following immunization (tumor prevention) or 3 days prior to the first FL injection (tumor treatment). (B) Kaplan-Meier graphs showing the tumor-free survival of mice treated as described in panel A. Results shown are representative of 3 separate experiments. ● indicates FL-ISS-OVA (1 tumor challenge); ○, FL-ISS-OVA (rechallenge); ▴, FL–C-ODN–OVA; ▵, FL-OVA; ♦, ISS-OVA; ■, OVA alone. (D,E) Kaplan-Meier graphs showing the tumor-free survival of mice treated as described in panel C. These graphs are the results of 2 separate experiments. Statistically significant differences in tumor-free survival were found between mice treated with FL plus ISS plus OVA compared to control groups in both the tumor prevention (P = .0002) and the 2 tumor treatment experiments (P = .004). There was no statistical difference in tumor-free survival between FL plus OVA or FL plus OVA plus C-ODN and the control groups (P > .1) in any of the experiments. In panels D-E, ● indicates FL-ISS-OVA; ○, FL-ISS; ▴, FL–C-ODN–OVA; ▵, FL-OVA; ♦, ISS-OVA; ■, OVA alone.
Ag loading and ISS activation of FL-mobilized DCs induce tumor protection and regression of established tumors.
(A,C) Experimental design. Groups of 5 mice were immunized with 9 daily injections of FL followed by 1 SC injection of OVA mixed with ISS, C-ODN, or PBS on day 0. The mice were challenged with one SC injection of 1 × 104 B16-OVA melanoma tumor cells in the opposite flank 1 and 7 weeks following immunization (tumor prevention) or 3 days prior to the first FL injection (tumor treatment). (B) Kaplan-Meier graphs showing the tumor-free survival of mice treated as described in panel A. Results shown are representative of 3 separate experiments. ● indicates FL-ISS-OVA (1 tumor challenge); ○, FL-ISS-OVA (rechallenge); ▴, FL–C-ODN–OVA; ▵, FL-OVA; ♦, ISS-OVA; ■, OVA alone. (D,E) Kaplan-Meier graphs showing the tumor-free survival of mice treated as described in panel C. These graphs are the results of 2 separate experiments. Statistically significant differences in tumor-free survival were found between mice treated with FL plus ISS plus OVA compared to control groups in both the tumor prevention (P = .0002) and the 2 tumor treatment experiments (P = .004). There was no statistical difference in tumor-free survival between FL plus OVA or FL plus OVA plus C-ODN and the control groups (P > .1) in any of the experiments. In panels D-E, ● indicates FL-ISS-OVA; ○, FL-ISS; ▴, FL–C-ODN–OVA; ▵, FL-OVA; ♦, ISS-OVA; ■, OVA alone.
To determine if the combination of FL, ISS, and Ag can induce regression of existing tumors, we established melanoma tumors in mice for 3 days before starting the daily FL injections (Figure 4C). Twelve days following injection of tumor, mice were immunized with OVA with or without ISS. Sixty percent of the mice treated with both FL mobilization and OVA plus ISS survived without developing detectable tumors (Figure 4D-E). In contrast, less than 20% of the mice treated with OVA plus ISS without FL, or FL plus OVA without ISS, and none of the mice treated with OVA plus CFA or OVA alone, were free of tumor (Figure 4D-E and data not shown). Administration of OVA in association with ISS and FL was always required to induce tumor regression (Figure 4D-E).
CD8 T cells and NK cells mediate the antitumor effect of FL, Ag, and ISS
To identify the effector cells responsible for the observed antitumor effect, mice were depleted of T-cell subsets or NK cells before or after immunization (Figure 5A). CD8 T cells were required for tumor protection as depletion of these cells before immunization abrogated this effect (Figure 5B). By contrast, ISS-activated DCs did not require CD4 T cells either as effectors or helpers to induce Ag-specific CD8 T cells because mice depleted of CD4 lymphocytes before treatment and immunization could still reject the tumor challenge. NK cells also appear to play an important role in the tumor protection induced by ISS and Ag following FL mobilization, because 50% of mice depleted of NK cells succumbed to tumor challenge (Figure 5B).
CD8 T cells and NK cells mediate the antitumor effect of FL plus Ag plus ISS.
(A) Experimental design. Groups of 8 mice were immunized with PBS alone or with FL followed by a SC injection of ISS mixed with OVA. Cohorts of mice were injected with either anti-CD4 antibody 6 days prior to immunization or with anti-CD4, anti-CD8, or anti-NK cell antibody 1 day after immunization. Mice were then challenged 1 week following immunization with 1 × 104 melanoma tumor cells. (B) Kaplan-Meier graphs showing the tumor-free survival of mice inoculated as described in panel A. Statistically significant differences in tumor-free survival were found between nondepleted mice versus CD8-depleted mice (P < .001) and between nondepleted versus NK cell-depleted mice (P = .003). ● indicates FL-ISS-OVA; ⋄, FL-ISS-OVA (CD4 depletion day +10); ▴, FL-ISS-OVA (CD4 depletion day −6); ○, FL-ISS-OVA (NK depletion); ▪, FL-ISS-OVA (CD8 depletion); ■, OVA alone.
CD8 T cells and NK cells mediate the antitumor effect of FL plus Ag plus ISS.
(A) Experimental design. Groups of 8 mice were immunized with PBS alone or with FL followed by a SC injection of ISS mixed with OVA. Cohorts of mice were injected with either anti-CD4 antibody 6 days prior to immunization or with anti-CD4, anti-CD8, or anti-NK cell antibody 1 day after immunization. Mice were then challenged 1 week following immunization with 1 × 104 melanoma tumor cells. (B) Kaplan-Meier graphs showing the tumor-free survival of mice inoculated as described in panel A. Statistically significant differences in tumor-free survival were found between nondepleted mice versus CD8-depleted mice (P < .001) and between nondepleted versus NK cell-depleted mice (P = .003). ● indicates FL-ISS-OVA; ⋄, FL-ISS-OVA (CD4 depletion day +10); ▴, FL-ISS-OVA (CD4 depletion day −6); ○, FL-ISS-OVA (NK depletion); ▪, FL-ISS-OVA (CD8 depletion); ■, OVA alone.
Discussion
The capacity of DCs to prime Ag-specific immunity relies on both their access to relevant Ag and their state of activation.1,21 22 In this study we demonstrated that DCs can be manipulated in vivo such that both parameters are enhanced. However, to induce effective antitumor immunity, increasing DC availability through in vivo expansion and activation of these DCs at sites where Ag is present are both required.
In these studies FL provided a critical growth stimulus for DCs, directing sufficient numbers of these cells to peripheral tissues to enable local delivery of Ag and ISS. The failure of FL alone to induce an antitumor response contrasts with a prior report.23However, the latter study used slow-growing tumors rather than aggressive and poorly immunogenic tumors such as the B-16 melanoma used here. Our results are also in contrast to the results reported by Pulendran and coworkers demonstrating that FL can be used as a vaccine adjuvant.24 This study, however, was performed with TCR transgenic mice in which the predominant population of T-cell precursors is specific for the target Ag. Our study was performed in nontransgenic animals with diverse T-cell repertoires. In this setting, FL-expanded DCs may not efficiently prime the low frequency (< 0.01%) of relevant T-cell precursors, a setting typical of clinical situations.
ISS strongly activated FL-DCs in vitro and in vivo. Interestingly, we found that ISS dramatically increases the persistence of mature DCs in FL-treated animals. One potential explanation for this persistence is that DCs simply survive longer after their exposure to ISS, which was confirmed in the in vitro experiments. Although DCs deprived of cytokines and cultured without ISS died in 4 days, a significant proportion of DCs cultured with ISS survived beyond 11 days. The possibility exists that continuous migration of DCs from the periphery to draining lymph nodes25 contributed to the high numbers of DCs in the lymph nodes of mice treated with FL and ISS. Nevertheless, these DCs continue to stimulate Ag-specific T cells within the lymph nodes even 3 weeks after treatment with ISS and Ag.
The combination of DC mobilization with DC activation at the site of Ag administration dramatically enhanced the induction of systemic T-cell immunity. Thus, the number of Ag-specific CD4 and CD8 T cells as evidenced by T-cell proliferation and IFN-γ secretion, respectively, was significantly increased in the draining lymph nodes. This effect was likely not due to a direct effect of FL or ISS on T cells, because FL alone does not induce T-cell proliferation,14 and ISS failed to costimulate a purified population of T cells activated with anti-CD3 or anti-TCR but yet enhanced the capacity of APC to activate T cells.26,27 Consistent with the latter study, we found that ISS enhances the capacity of FL-mobilized DCs to prime Ag-specific T cells (Figures 1C-D and 3). This effect may be due to the up-regulation of MHC class II and costimulatory molecules (Figures 1A and 2A) on ISS-activated DCs, as well as to the induction of secretion of cytokines such as IL-12 and IFN-γ by ISS-activated DCs (Figure1B). Ag-specific CD8 T cells could be detected in draining lymph nodes more than 3 weeks after the immunization. Because activated T cells leave the lymph node within a few days, these results suggest that Ag-loaded DCs continue to prime newly arrived naı̈ve T cells. Alternatively, memory T cells that recirculate back to the lymph nodes might be restimulated by persistent Ag-loaded DCs.28
To determine whether the potent, persistent Th1 response induced by FL followed by ISS and Ag could induce effective antitumor immunity, we used an aggressive B16 melanoma tumor line that had been engineered to express OVA as the target tumor Ag. The combination of FL mobilization followed by ISS activation of DC in the presence of OVA induced a protective antitumor response and also led to regression of established tumors. CD8 T cells were required for tumor protection because depletion of these cells before immunization abrogated this effect. By contrast, ISS-activated DCs did not require CD4 T cells either as effectors or helpers to induce Ag-specific CD8 T cells because mice depleted of CD4 lymphocytes before treatment and immunization could still reject tumor challenge. These results are consistent with recent findings showing that vaccination with ISS and Ag was able to induce a similar level of Ag-specific CD8 cytotoxic T lymphocyte-mediated response29 and to confer a similar level of tumor protection in CD4 wild-type and knock-out mice.30 These results suggest that similar to viruses, double-stranded RNA, and CD40L, ISS is able to “license” DCs to directly prime CD8 T cells.30-32 Although CD4 T cells were not required for CD8 priming, CD4 help clearly enhanced priming because 40% of the CD4-depleted mice still succumbed to tumor challenge.
The NK cells also played an important role in the tumor protection provided by FL, ISS, and Ag, because 50% of mice depleted of NK cells succumbed to tumor challenge. FL treatment can induce modest NK cell proliferation, but does not induce NK activation.33 In addition, FL-deficient mice have a striking defect in NK cell function,34 suggesting a major role for FL in NK cell development and maturation. ISS failed to activate NK directly. However, ISS increased the capacity of FL-DCs to activate NK cells. This effect does not rely on cell-cell contact because similar results were obtained when the cells were cocultured on opposite sides of a porous membrane (data not shown). As shown in Figure 1B, ISS-stimulated DCs secreted large amounts of IL-12 and IFN-γ, which are known to activate NK cells. These findings are consistent with previous results showing that ISS does not activate human NK cells directly but augments NK activity by inducing the secretion of IL-12, IFN-α, IFN-β and TNF-α.35 The combination of these results suggests that FL and ISS work together through DCs to accentuate NK activation.
Although ISS can strongly activate B cells,36 FL neither activates B cells nor induces mobilization of mature B cells into the periphery.14 Because, in our studies, FL was absolutely required for any antitumor efficacy, it seems unlikely that B cells played a significant role in this effect.
It is important to note that in the absence of exogenously administered tumor Ag, FL and ISS did not lead to tumor rejection indicating that the mechanism responsible for the antitumor effect is Ag specific. This also indicates that FL-mobilized DCs either do not reach a critical number in the tumor sites or are not activated in such sites. Specifically recruiting DCs to tumor sites and then activating them with ISS would be a potential approach to overcome the need for Ag immunization.
In summary, we described an effective method of inducing DCs to prime an Ag- specific response in vivo without the requirement of DC manipulation ex vivo. Our study demonstrates the immunotherapeutic potential of combining FL with ISS and Ag treatment for induction of a strong cell-mediated immune response against tumor Ag. These findings provide the basis for human trials to test the efficacy of combining DC mobilization with DC activation and Ag delivery in situ for clinical immunotherapy.
We thank Claudia Benike for critical review of the manuscript and Donna Jones for formatting the manuscript.
Supported, in part, by grants from the National Institutes of Health (CA71725, HL57443, and CA72103), and from the Tobacco Related Disease Research Program of the University of California (9RT-0229) L.F. was supported by a physician-scientist award from the National Cancer Institute (K23 CA82584-01).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Edgar G. Engelman, Stanford Blood Center, 800 Welch Rd, Palo Alto, CA 94304; e-mail: edengleman@stanford.edu.




This feature is available to Subscribers Only
Sign In or Create an Account Close Modal