Key Points
Loss of mDia1 induces neutropenia through increased adhesion of neutrophils to blood vessels and transendothelial migration.
mDia1-deficient neutrophils show upregulation of CD11b that is caused by attenuated CD11b endocytosis.
Abstract
Formin protein mDia1 is involved in actin polymerization and plays important roles in the migration and adhesion of hematopoietic cells. The mDia1 encoding gene is located on chromosome 5q, which is commonly deleted in patients with del(5q) myelodysplastic syndromes (MDSs). We previously reported that mice with mDia1 deficiency developed neutropenia that closely mimics MDS. However, the mechanism of neutropenia in these mice and patients with del(5q) MDS remains incompletely defined. Here, we reveal that mDia1 knockout mice show cell-autonomously increased CD11b expression on neutrophils in the peripheral blood and bone marrow. The level of CD11b was also higher in patients with del(5q) MDS compared with normal individuals. Mechanistically, loss of mDia1 significantly attenuated the endocytosis of CD11b on neutrophils, which led to an increased number of neutrophils adhering to the blood vessels in mDia1 knockout mice. Administration of CD11b antibody to mDia1 knockout mice reduced the adhesion of neutrophils to the vessels and rescued neutropenia. Our study reveals the role of mDia1 deficiency in the upregulation of CD11b on neutrophils, which leads to their increased binding to the blood vessels. These results may provide important clues for the pathogenesis of neutropenia in patients with del(5q) MDS.
Introduction
Myelodysplastic syndromes (MDSs) are clonal disorders of hematopoietic stem cells characterized by ineffective hematopoiesis with an increased risk of progression to acute myeloid leukemia.1 Patients with MDS have an increased risk of infection because of neutropenia, the main predisposing factor.2 The degree of neutropenia has a significant prognostic impact in low-risk MDS.3 The etiology of neutropenia in MDS is unclear, which is partially a result of the heterogeneous nature of the pathogenesis of MDS. Deletion of chromosome 5q is one of the most common cytogenetic abnormalities in MDS. We previously reported that mice with loss of mDia1, which is encoded by one of the chromosome 5q genes, developed significant neutropenia.4 However, the mechanism of neutropenia induced by the loss of mDia1 is still unknown.
The formin protein mDia1 is involved in linear actin filament polymerization.5 It has important functions in cell migration, adhesion, and cell-cell interactions.6 A previous report demonstrated that mDia1 is critical for the migration of neutrophils during chemotaxis.7 However, this does not explain steady-state neutropenia in mDia1-deficient mice. How mDia1 is involved in the interaction of neutrophils with their microenvironment is also unclear.
β2 integrin Mac-1 (CD11b/CD18) mediates the stable adhesion of neutrophils to endothelial cells through interactions with intercellular adhesion molecules (ICAM-1 or ICAM-2) and extracellular matrix proteins such as fibronectin, laminin, and collagen.8 CD11b interacts and co-localizes with Ly6G; a low dose of anti-Ly6G antibody blocks neutrophil adhesion in experimental arthritis and thioglycollate-stimulated peritonitis through a CD11b-dependent mechanism.9 In this study, we found that neutrophil surface expression of CD11b is significantly increased through an mDia1 deficiency–induced endocytosis defect. The upregulated CD11b causes increased adhesion of circulating neutrophils to the endothelium and transendothelial migration.
Methods
Materials
Iscove modified Dulbecco medium (IMDM) was purchased from Gibco. The biotin mouse lineage panel, Cyanine 5 (Cy5)–labeled Annexin V, phycoerythrin (PE)-conjugated rat anti-mouse CD11b (M1/70), and allophycocyanin (APC)-conjugated rat anti-mouse CD18 (C71/16) were purchased from BD Pharmingen. PE-Cy7 rat anti-mouse Ly6G (RB6-8C5), PE rat anti-mouse Ly6G (1A8), APC rat anti-mouse CD11b (M1/70), and APC rat anti-mouse CD184 (CXCR4) (2B11) were purchased from eBioscience. Pacific blue rat anti-mouse Gr1 (RB6-8C5) and APC rat anti-mouse CD11a (M17/4) were from BioLegend. 4′,6-diamidino-2-phenylindole and propidium iodide were purchased from Invitrogen/Molecular Probes.
Mice
mDia1 knockout mice harboring green fluorescent protein (GFP) were as previously described.4 All the experiments were approved by the Institutional Animal Care and Use Committees at Northwestern University.
Patient data
MDS patient data were obtained after informed consent under institutional review board-approved protocols at H. Lee Moffitt Cancer Center and Research Institute.
Bone marrow transplantation
Bone marrow transplantation was performed as previously described.4,10,11 In brief, 2 × 106 donor bone marrow cells from 6- to 8-week-old mice were injected retro-orbitally into a lethally irradiated (10 Gy) recipient mouse. The competitive bone marrow transplantation was carried out under the same conditions, except that 1 × 106 mDia1 knockout cells harboring GFP were premixed with 1 × 106 wild-type cells from their littermate control mice. At least 5 mice were used for each genotype group. The recipient mice were then supplied with water with antibiotics (sulfamethoxazole and trimethoprim oral suspension from Hi-Tech Pharmacal) for 3 weeks. The mice were analyzed for complete blood count (CBC) and flow cytometric assay of neutrophils from both peripheral blood and bone marrow.
RNA extraction and quantitative reverse transcription polymerase chain reaction
To purify neutrophils from the peripheral blood, the blood was treated with red blood cell (RBC) lysis buffer for 5 minutes on ice, and after extensive wash with phosphate-buffered saline (PBS), Gr1/Mac1+ neutrophils were purified with a magnetic bead–based separation method that used a biotin mouse lineage panel, following manufacturer’s protocol (BD Pharmingen). RNA isolation, complementary DNA synthesis, and quantitative polymerase chain reaction were performed as previously described.4 Primer sequences for CD11b were forward 5′-CTGAACATCCCATGACCTTCC-3′ and reverse 5′-GCCCAAGGACATATTCACAGC-3′. For 18S ribosomal RNA, the primer sequences were forward 5′-GCAATTATTCCCCATGAACG-3′ and reverse 5′-GGCCTCACTAAACCATCCAA-3′.
Flow cytometric analysis and endocytosis assay
Peripheral blood obtained from either retro-orbital or tail vein bleeding was lysed with 1 mL RBC lysis buffer (eBioscience) for 5 minutes on ice. Cells were recovered by adding 4 mL of cold PBS followed by centrifuge at 1500 rpm for 10 minutes at 4°C. For the endocytosis assay, after the last wash with PBS, the cells were aliquoted into 5 mL polystyrene round-bottom tubes in 20 μL volume. Then, 500 μL of prewarmed serum-free IMDM was added to each tube, and the tubes were then transferred to a 37°C water bath. After incubation to the desired time point (see figures), the tubes were placed on ice to stop the endocytosis, and 4 mL of cold PBS was added immediately. All the cells were then centrifuged at 4°C and stained with fluorescence-labeled anti-CD11b antibodies on ice for 30 minutes, after a wash with cold PBS. All the cells were kept on ice prior to flow cytometry acquisition. Propidium iodide was added to exclude the dead cells.
Adhesion assay
Peripheral blood granulocytes were negatively purified with a magnetic bead–based separation method that used a biotin mouse lineage panel. In brief, after RBC lysis and extensive wash with PBS, cells were incubated with anti-CD3ε, anti-Ter119, and anti-B220 antibodies to exclude erythrocytes and lymphocytes and were then subjected to magnetic bead–based separation, according to the manufacturer’s instructions. For the adhesion assay, equal numbers of cells were re-suspended in 75 μL of serum-free IMDM and incubated on cover slips coated with poly-L-lysine from Corning for 10 minutes at 37°C. The adherent cells were fixed with 4% formaldehyde solution after being washed with PBS 3 times. The number of cells per 20× field was recorded.
Transendothelial migration assay
Transendothelial migration (TEM) assays were performed as previously described.12 Briefly, the confluent human umbilical vein endothelial cell monolayers grown on hydrated type-I collagen gels were pretreated with 20 ng/mL human recombinant tumor necrosis factor (R&D Systems) for 4 hours. After being washed twice with medium, 100 µL of negatively selected neutrophils from peripheral blood (0.6 × 106 cells per mL) were added to tumor necrosis factor–activated human umbilical vein endothelial cells and allowed to migrate for 45 minutes at 37°C in 5% CO2. Monolayers were washed twice with 1 mM EDTA in PBS (no Ca2+ or Mg2+) and then twice with PBS (with Ca2+ and Mg2+) before being fixed in 1% glutaraldehyde in 0.1 M sodium cacodylate buffer (Electron Microscopy Sciences). Fixed cells were stained with modified Wright-Giemsa stain (Protocol Hema3; Thermo Fisher Scientific) and mounted on glass slides. At least 300 leukocytes per monolayer were scored from a total of 6 random fields by using an Ultraphot microscope (Carl Zeiss) with Nomarski optics and a SPOT Insight Color charge-coupled device (CCD) (Diagnostic Instruments Inc.). Leukocytes were scored on the basis of their position relative to the endothelial nuclei (above or below). The relative percentage of the TEM was quantified as transendothelial migrated cells vs total adherent cells.
Intravital imaging
Mice were anesthetized by intraperitoneal injection of 100 mg/kg ketamine and 5 mg/kg xylazine solution. The mixture of anti-PECAM-1 (clone 390) and anti-Ly6G (clone RB6-8C) monoclonal antibodies conjugated to Alexa 555 and Alexa 660 (2 μg/mL), respectively, were injected via the tail vain to outline the intestinal blood vessels and neutrophils. After the injection, a piece of the small intestine was gently exteriorized and placed on a custom-designed platform superfused with warmed physiological saline solution. Core body temperature was maintained by placing the mouse on a warming pad set to 37°C. Imaging experiments were performed by using an Ultraview Vox imaging system equipped with 488, 561, and 640 nm laser lines, a Nipkow disk confocal head (CSU 10, Yokogawa Electric, Tokyo, Japan) attached to an intensified CCD camera and a 20× water-immersion objective (numerical aperture, 1.00). Laser power and camera gain settings were unchanged throughout all the experiments. Images were digitally acquired at a resolution of 512 × 512 pixels using Volocity software (Perkin Elmer, Naperville, IL) and analyzed by using ImageJ software.
CD11b antibody blocking in vivo
After a CBC assay with Hemavet 950 (Drew Scientific), mDia1 wild-type and knockout mice were retro-orbitally injected with 50 μg of low-grade endotoxin anti-CD11b (BioLegend) antibody or isotype control immunoglobulin G (BioLegend) in 100 μL of sterile PBS for 2 consecutive days. At 48 or 90 hours (after the last injection), peripheral blood was retro-orbitally collected with tubes containing EDTA for CBC assay.
Statistical analysis
Results are expressed as mean ± standard error of the mean unless otherwise indicated. The statistical analysis was performed with Student t test using GraphPad Prism version 5.0 software. P < .05 was considered statistically significant.
Results
Consistent with our previous report,4 mDia1 knockout mice showed a nearly 50% decrease in the absolute number of circulating neutrophils, confirming their neutropenia. However, the absolute number of lymphocytes, monocytes, RBCs, and platelets remained unchanged (Figure 1A; supplemental Figure 1). The presence of neutropenia was evident throughout the life of the mDia1 knockout mice (data not shown). In addition, neutropenia induced by the loss of mDia1 was cell-autonomously revealed by a bone marrow transplantation assay (Figure 1B). This was further demonstrated by a reverse transplantation assay in which normal CD45.1+ congenic bone marrow cells were transplanted into the lethally irradiated mDia1 knockout mice and their wild-type controls. In this case, mDia1 knockout mice showed levels of neutrophils comparable to those of wild-type mice (Figure 1C).
mDia1 knockout (KO) mice develop neutropenia with increased expression levels of CD11b on neutrophils. (A) Absolute neutrophil count of 20-week old mDia1 KO (n = 13) and their littermate wild-type (WT) control mice (n = 13). (B) mDia1 WT (n = 8) and KO (n = 8) mouse bone marrow cells (CD45.2+) were retro-orbitally injected into lethally irradiated CD45.1+ congenic female mice. Absolute neutrophil count was analyzed at 3 months posttransplantation. (C) Bone marrow cells from CD45.1+ wild-type mice were transplanted into lethally irradiated CD45.2+ mDia1 WT (n = 5) or KO (n = 7) mice. Absolute neutrophil count was analyzed 3 months after transplantation. (D) Relative percentage and MFI of CD11b expression levels on the peripheral blood (PB) neutrophils and bone marrow (BM) granulocytes in mDia1 WT (n = 8) and KO (n = 6) mice. Data were obtained from 4 independent experiments. (E) CD11b expression levels on premature (CD11blow) and mature (CD11bhigh) bone marrow granulocytes. Data were obtained from 4 independent experiments (WT mice, n = 8; KO mice, n = 6). (F) Correlation analysis of neutrophil counts and CD11b expression levels (WT mice, n = 12; KO mice, n = 11). (G-H) CD11b expression levels on bone marrow granulocytes and peripheral blood neutrophils of the receipt mice, as described in panels B and C, were analyzed by flow cytometric analysis and presented in panels G and H, respectively. All data are shown as mean ± standard error of the mean. (I) Schematic illustration of the competitive bone marrow transplantation assay (left) and the levels of CD11b in the recipient mice (n = 3) from wild-type and knockout donor cells. (J) The absolute cell numbers for total bone marrow cells and granulocytes were quantified by flow cytometer (n = 3 in each group). (K) The percentage of apoptotic granulocytes in peripheral blood were assayed by annexin V (AnV) staining (WT mice, n = 9; KO mice, n = 11). (L) Relative mRNA levels of CD11b in the granulocytes from bone marrow and peripheral blood were assayed by quantitative polymerase chain reaction (WT mice, n = 7; KO mice, n = 5). (M). In vitro endocytosis assay of CD11b with peripheral blood neutrophils from mDia1 wild-type and knockout mice. Data were obtained from 3 independent experiments. (N) Representative flow cytometry results of the relative CD11b expression levels in a normal patient and a patient with del(5q) MDS. The cells were analyzed from the gated granulocytic population. (O) Quantitative analysis of data from panel N (del(5q) MDS patients, n = 8; normal patients, n = 14). *P < .05; **P < .01. FACS, fluorescence-activated cell sorting; NE, neutrophils; rRNA, ribosomal RNA; SSC-A, side scatter pulse area.
mDia1 knockout (KO) mice develop neutropenia with increased expression levels of CD11b on neutrophils. (A) Absolute neutrophil count of 20-week old mDia1 KO (n = 13) and their littermate wild-type (WT) control mice (n = 13). (B) mDia1 WT (n = 8) and KO (n = 8) mouse bone marrow cells (CD45.2+) were retro-orbitally injected into lethally irradiated CD45.1+ congenic female mice. Absolute neutrophil count was analyzed at 3 months posttransplantation. (C) Bone marrow cells from CD45.1+ wild-type mice were transplanted into lethally irradiated CD45.2+ mDia1 WT (n = 5) or KO (n = 7) mice. Absolute neutrophil count was analyzed 3 months after transplantation. (D) Relative percentage and MFI of CD11b expression levels on the peripheral blood (PB) neutrophils and bone marrow (BM) granulocytes in mDia1 WT (n = 8) and KO (n = 6) mice. Data were obtained from 4 independent experiments. (E) CD11b expression levels on premature (CD11blow) and mature (CD11bhigh) bone marrow granulocytes. Data were obtained from 4 independent experiments (WT mice, n = 8; KO mice, n = 6). (F) Correlation analysis of neutrophil counts and CD11b expression levels (WT mice, n = 12; KO mice, n = 11). (G-H) CD11b expression levels on bone marrow granulocytes and peripheral blood neutrophils of the receipt mice, as described in panels B and C, were analyzed by flow cytometric analysis and presented in panels G and H, respectively. All data are shown as mean ± standard error of the mean. (I) Schematic illustration of the competitive bone marrow transplantation assay (left) and the levels of CD11b in the recipient mice (n = 3) from wild-type and knockout donor cells. (J) The absolute cell numbers for total bone marrow cells and granulocytes were quantified by flow cytometer (n = 3 in each group). (K) The percentage of apoptotic granulocytes in peripheral blood were assayed by annexin V (AnV) staining (WT mice, n = 9; KO mice, n = 11). (L) Relative mRNA levels of CD11b in the granulocytes from bone marrow and peripheral blood were assayed by quantitative polymerase chain reaction (WT mice, n = 7; KO mice, n = 5). (M). In vitro endocytosis assay of CD11b with peripheral blood neutrophils from mDia1 wild-type and knockout mice. Data were obtained from 3 independent experiments. (N) Representative flow cytometry results of the relative CD11b expression levels in a normal patient and a patient with del(5q) MDS. The cells were analyzed from the gated granulocytic population. (O) Quantitative analysis of data from panel N (del(5q) MDS patients, n = 8; normal patients, n = 14). *P < .05; **P < .01. FACS, fluorescence-activated cell sorting; NE, neutrophils; rRNA, ribosomal RNA; SSC-A, side scatter pulse area.
By using a flow cytometric assay, we found that the percentages of Gr1+ and CD11b+ neutrophils (especially those of CD11b) and their mean fluorescence intensity (MFI) increased significantly in the peripheral blood of mDia1 knockout mice (Figure 1D and data not shown). The increase of CD11b is specific because there was no increased level detected for CD18 and CD11a (supplemental Figure 2). When we analyzed the more mature subpopulation of bone marrow CD11bhigh granulocytes, this population was significantly increased in mDia1 knockout mice (Figure 1E), indicating a higher level of CD11b accumulation on mDia1 knockout neutrophils during granulocytic differentiation. We also found that the increase in CD11b on mDia1 knockout neutrophils was strongly associated with a decrease in the absolute neutrophil count (Figure 1F).
Similarly, mice transplanted with bone marrow cells from mDia1 knockout mice showed increased CD11b expression on neutrophils from the peripheral blood and increased CD11b MFI in the bone marrow (Figure 1G). In contrast, wild-type bone marrow cells transplanted into mDia1 knockout mice showed few differences in CD11b levels compared with those transplanted into wild-type mice (Figure 1H). To further confirm the cell-autonomous effect of CD11b upregulation in mDia1 knockout neutrophils, we performed a competitive transplantation assay in which wild-type CD45.2+ and GFP– bone marrow cells were mixed with an equal amount of mDia1 knockout CD45.2+ and GFP+ bone marrow cells. These cells were transplanted into lethally irradiated mice; after transplantation, CD11b analysis was performed by using flow cytometry in the bone marrow, peripheral blood, and spleen. As expected, GFP+ mDia1 knockout cells showed significant upregulation of CD11b compared with GFP– wild-type controls (Figure 1I).
We confirmed that this decrease in circulating neutrophil count in mDia1 knockout mice was not a result of the accumulation or sequestration of neutrophils in the bone marrow (Figure 1J). In addition, loss of mDia1 did not affect the apoptosis of neutrophils in these mice under steady state (Figure 1K). Taken together, these results demonstrate that neutropenia in mDia1 knockout mice is not caused by the inhibition of granulocytic differentiation or proliferation or induction of apoptosis, but it could be related to the increased surface expression of CD11b.
To determine how CD11b is upregulated in mDia1 null neutrophils, we first analyzed the messenger RNA (mRNA) levels of CD11b because mDia1 is reported to play an important role in transcriptional regulation in addition to its function in actin polymerization.13 However, we did not detect a statistically significant increase of the mRNA level of CD11b in the neutrophils from either bone marrow or peripheral blood in mDia1 knockout mice (Figure 1L). These results indicate that the expression of surface CD11b may be deregulated at the posttranscriptional level in mDia1 knockout mice.
Efficient cell migration and adhesion necessitate constant endocytosis of integrins, which are either recycled or degraded after internalization.14 Integrins in the plasma membrane pool are recycled through the endosomal system.15 mDia1, as a regulator of actin dynamics, is also required for the formation of the actin coat around endosomes and plays a unique role in endosome transport.16 We next tested whether mDia1 deficiency affects the endocytosis of CD11b. By using an in vitro endocytosis assay, we detected a significant delay of endocytotic dynamics of CD11b on the membrane of mDia1 knockout neutrophils compared with their wild-type controls (Figure 1M).
To determine whether the increase of CD11b surface expression could also be identified in patients with del(5q) MDS, we analyzed the MFI of CD11b on the bone marrow granulocytes in a group of del(5q) MDS patients. Compared with normal patients, these patients showed significant increase of CD11b expression on bone marrow granulocytes (Figure 1N-O). Many of these patients presented with varying degree of neutropenia (data not shown) and different kinds of infections (supplemental Table 1).
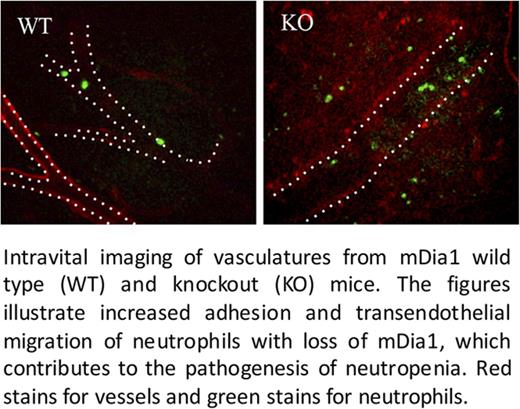
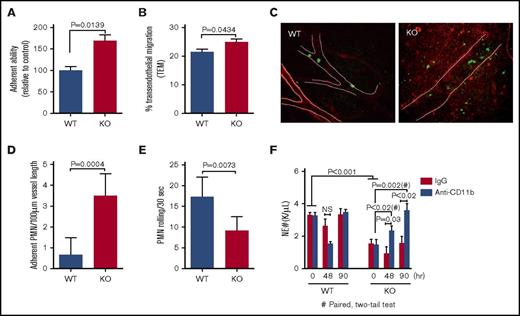
CD11b is the predominant β2 integrin on granulocytes and plays critical roles in the adhesion and transendothelial migration of leukocytes. Through an in vitro adhesion assay, we found that mDia1 knockout neutrophils from the peripheral blood exhibited increased adhesion abilities compared with wild-type cells (Figure 2A). In addition, the transendothelial migration capacity of mDia1 knockout neutrophils was also increased in a statistically significant way compared with the wild-type cells (Figure 2B). Importantly, we identified through a live in vivo imaging assay that there was a markedly increased number of mDia1 knockout neutrophils adhered to the blood vessels (Figure 2C-D). Furthermore, the neutrophil rolling velocity along the blood vessels is significantly decreased (Figure 2E; supplemental Movie 1), which is consistent with previous reports that CD11b is a major negative regulator of neutrophil rolling.8,17,18 We also observed significantly increased infiltrated neutrophils in the peritoneum (Figure 2C; supplemental Movie 1), suggesting a higher level of neutrophil transendothelial migration in mDia1 knockout mice.
Increased adhesion to blood vessels and TEM of mDia1-deficient neutrophils. (A) In vitro adhesion assay was performed with negatively selected peripheral blood neutrophils on coverslips coated with poly-L-lysine. The relative percentage of adherent mDia1-deficient neutrophils relative to wild-type cells is presented (n = 3 in each group). (B) Negatively selected neutrophils from peripheral blood were seeded on a monolayer of human endothelial cells pretreated with recombinant human tumor necrosis factor (20 ng/mL) for 4 hours. After incubation at 37°C for 45 minutes, the cells were fixed, stained, and counted for 6 random fields (>50 cells in each field). The relative percentage of the transendothelial migrated cells was quantified. (C) Intravital imaging was performed on vasculature of the mDia1 wild-type and knockout mice to evaluate the rolling speed and adhesion of neutrophils in blood vessels. Blood vessels and neutrophils were immunofluorescently stained for PECAM-1 (red, outlined by white dashed lines) and Gr1 (green), respectively. Original magnification ×20. Data were quantified from 6 random fields per group (n = 2 in each group). A representative field is presented. (D-E) Quantitative analysis of the adherent neutrophils and their rolling speed in panel C. (F) mDia1 wild-type and knockout mice were injected twice with anti-CD11b or control antibodies at 50 μg per mouse. The absolute neutrophil count was determined at the indicated time after the second injection (n = 3 mice in each group). IgG, immunoglobulin G; PMN, polymorphonuclear leukocytes.
Increased adhesion to blood vessels and TEM of mDia1-deficient neutrophils. (A) In vitro adhesion assay was performed with negatively selected peripheral blood neutrophils on coverslips coated with poly-L-lysine. The relative percentage of adherent mDia1-deficient neutrophils relative to wild-type cells is presented (n = 3 in each group). (B) Negatively selected neutrophils from peripheral blood were seeded on a monolayer of human endothelial cells pretreated with recombinant human tumor necrosis factor (20 ng/mL) for 4 hours. After incubation at 37°C for 45 minutes, the cells were fixed, stained, and counted for 6 random fields (>50 cells in each field). The relative percentage of the transendothelial migrated cells was quantified. (C) Intravital imaging was performed on vasculature of the mDia1 wild-type and knockout mice to evaluate the rolling speed and adhesion of neutrophils in blood vessels. Blood vessels and neutrophils were immunofluorescently stained for PECAM-1 (red, outlined by white dashed lines) and Gr1 (green), respectively. Original magnification ×20. Data were quantified from 6 random fields per group (n = 2 in each group). A representative field is presented. (D-E) Quantitative analysis of the adherent neutrophils and their rolling speed in panel C. (F) mDia1 wild-type and knockout mice were injected twice with anti-CD11b or control antibodies at 50 μg per mouse. The absolute neutrophil count was determined at the indicated time after the second injection (n = 3 mice in each group). IgG, immunoglobulin G; PMN, polymorphonuclear leukocytes.
Loss of CD11b expression or functional blocking of CD11b reduces the sequestration and adhesion of neutrophils.19-21 Injection of antibodies against CD11b in mice was shown to be able to inhibit lipopolysaccharide-induced leukopenia.19 In addition, inactivation of CD11b by a transgenic overexpression of CD11b antagonist neutrophil inhibitory factor results in an increase in circulating neutrophil count.8 These reports indicate that blocking CD11b may ameliorate neutropenia in mDia1 knockout mice. To verify this, we injected mDia1 knockout or wild-type mice with anti-CD11b antibodies and performed an absolute neutrophil count. Anti-CD11b injection led to a dramatic and time-related increase of neutrophil count in mDia1 knockout mice almost to the same level as in wild-type control mice (Figure 2F). These data further confirm the role of the increased surface CD11b expression in the pathogenesis of neutropenia in mDia1 knockout mice.
Discussion
Taken together, our study reveals the role of mDia1 deficiency in the pathogenesis of neutropenia in patients with del(5q) MDS. Loss of mDia1 leads to the upregulation of CD11b because of its reduced endocytosis, which in turn causes increased adhesion and transendothelial migration of neutrophils. We previously reported that loss of mDia1 also induces increased level of CD14.4 Unlike CD14, which is upregulated through the transcriptional level, the mRNA level in CD11b is not affected by the loss of mDia1. The posttranslational regulation of CD11b by mDia1 is specific because CD11a and CD18 are not increased. As in the mDia1 knockout mouse, MDS patients with del(5q) exhibit increased CD11b on their bone marrow neutrophils. Notably, there is a variation of CD11b levels among the MDS patients. Ideally, levels of CD11b would be correlated with absolute neutrophil counts, but confounding factors, including complex genetic abnormalities and treatment history, made it impractical to establish any correlations. Indeed, most of these patients presented with neutropenia and various kinds of infections.
Our study indicates that in addition to the ineffective hematopoiesis that causes reduced neutrophil production,2 neutrophil surface protein expression changes, which leads to its functional abnormalities that are also important in contributing to the pathogenesis of neutropenia in patients with del(5q) MDS.
The full-text version of this article contains a data supplement.
Acknowledgments
The authors thank Xinyan Lu for help with the cytogenetic analyses of the MDS patients.
This work was supported by grant R01 DK102718 from the National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, and by a Department of Defense Career Development Award (CA140119) (P.J.).
Authorship
Contribution: Y.M. designed and performed the research, analyzed and interpreted the data, and wrote the manuscript; R.S. performed in vivo imaging and data interpretation; G.F., B.Z., J.Z., L.C., N.R., and J.Y. performed the experiments; L.Z., J.G., and Y.-H.C. interpreted the clinical data; W.A.M. interpreted the data; and P.J. designed the study, analyzed and interpreted the data, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Peng Ji, Department of Pathology, Feinberg School of Medicine, Northwestern University, 303 East Chicago Ave, Ward 3-245, Chicago, IL 60611; e-mail: peng-ji@fsm.northwestern.edu.