TO THE EDITOR:
CD19-directed chimeric antigen receptor (CAR) T cells have greatly improved the prognosis of relapsed/refractory large B-cell lymphoma (LBCL), yet treatment failure occurs in > 50% of patients, usually in the first 3 months after CAR T-cell infusion.1-3 Apart from patient status and tumor burden,4 early factors capable of predicting a durable response would be crucial in better selecting patients at the time of decision to treat (ie, before the manufacture of CAR T cells) to avoid a costly and potentially life-threatening treatment when it has little chance of clinical benefits.
The quality of apheresed T cells, which represent the starting material for CAR T-cell manufacture, has emerged as a strong determinant of antitumor response in patients with B-cell malignancies. Early lineage phenotypes are essential to T-cell fitness but are altered by cumulative chemotherapy cycles.5,6 Naïve and central memory enriched T cells generated CAR products that retained replicative potential and were associated with complete and sustained remission in various clinical trials.7-11 Notably, in patients with LBCL treated with axicabtagene ciloleucel (axi-cel; CD19-CD28 CAR T), a higher proportion of naïve/stem cell memory T cells in the apheresis product was associated with rapid expansion during CAR T-cell manufacturing and a durable response.12 However, data are missing in the real-life setting, from which patients are less often selected. Here, we investigated whether intrinsic characteristics of apheresed T lymphocytes influence the clinical response to tisagenlecleucel (tisa-cel; CD19-4.1BB CAR T) in real-life patients with LBCL.
We studied 30 consecutive patients with aggressive diffuse LBCL or transformed follicular lymphoma who received commercial tisa-cel between April 2019 and March 2020 in our center. All patients were alive beyond 28 days and evaluable for response. Response assessment was performed at 1, 3, 6, and 12 months per institutional practice and based on Lugano criteria.13 Parameters of immune monitoring were routinely determined as a standard of care. The number of total and CAR T cells in the final product was obtained from the manufacturer’s release certificate. The study was performed under institutional review board–approved protocols, and all patients signed informed consent.
Cryopreserved mononuclear cells from the apheresis product were used retrospectively for this study. Cells were stained with combination of antibodies, allowing differentiation of naïve/stem cell memory (TN/SCM; CD45RO− CD27+), central memory (TCM; CD45RO+CD27+), effector memory (TEM; CD45RO+CD27−), and effector (TEFF; CD45RO−CD27−) populations among CD4 and CD8 subsets (supplemental Table 1). We previously verified in 9 patients the perfect correlation between T-cell phenotypes in the cryopreserved apheresis product and fresh peripheral blood collected on the same day (supplemental Figure 1). For proliferation assays, carboxyfluorescein succinimidyl ester (CFSE) –labeled mononuclear cells were cultured with Dynabeads human T-activator CD3/CD28 (1:1 ratio) for 5 days, after which T-cell proliferation was quantified according to CFSE dilution. Samples were acquired using Attune NxT flow cytometer (Thermo Fisher) and analyzed in FlowJo. T-cell phenotypes were validated using unsupervised analysis with the FlowSOM clustering and visualization algorithm (supplemental Figure 2).
Statistics were performed using Prism v9 (GraphPad). Statistical tests used are indicated in figure legends. P values < 0.05 were considered significant.
Characteristics of the patients are shown in supplemental Table 2. The distribution of naïve, memory, and effector CD4 and CD8 subsets among apheresed T lymphocytes was highly heterogeneous among patients (Figure 1A-B). The phenotype of apheresed T cells had a strong impact on their functional fitness, as shown by the correlation between the proportion of naïve/stem cell memory cells and the ability of CD4 and CD8 T cells to proliferate in vitro (Figure 1C). It also impacted the relative expansion of CD4 and CD8 T cells during the CAR T-cell manufacturing process. Thus, the CD4/CD8 ratio among total T cells (or CAR+ T cells) in the final product was significantly related to the ratio between CD4 TN/SCM and CD8 TN/SCM in the apheresis (R = 0.50; P < .01) and proliferative potential of CD4 relative to CD8 T cells (R = 0.65; P < .01) (Figure 1D; supplemental Figure 3). These data indicate that the proportions of CD4 and CD8 TN/SCM cells in the starting material are critical for the quality of the CAR T-cell product.
Intrinsic characteristics of apheresed T cells and quality of the CAR T-cell product. (A) Representative staining of naïve/stem cell memory (TN/SCM; CD45RO− CD27+), central memory (TCM; CD45RO+CD27+), effector memory (TEM; CD45RO+CD27−), and effector (TEFF; CD45RO−CD27−) CD8 and CD4 subsets in apheresed T lymphocytes. (B) Distribution of TN/SCM, TCM, TEM, and TEFF CD8 and CD4 T cells in apheresed T lymphocytes from 30 patients treated with tisa-cel. Bars represent the median. (C) Correlation between the naïve/stem cell memory phenotype and in vitro proliferative capacity (proportion of CFSElow cells) of apheresed CD8 (left panel) and CD4 (right panel) T cells. (D) The CD4/CD8 T-cell ratio in the final product correlates with the ratio of CD4 TN/SCM to CD8 TN/SCM cells in the apheresis (left) and the ratio of CFSElow CD4 to CD8 T cells (right). Pearson correlation test. ρ coefficient (R) and P values are indicated.
Intrinsic characteristics of apheresed T cells and quality of the CAR T-cell product. (A) Representative staining of naïve/stem cell memory (TN/SCM; CD45RO− CD27+), central memory (TCM; CD45RO+CD27+), effector memory (TEM; CD45RO+CD27−), and effector (TEFF; CD45RO−CD27−) CD8 and CD4 subsets in apheresed T lymphocytes. (B) Distribution of TN/SCM, TCM, TEM, and TEFF CD8 and CD4 T cells in apheresed T lymphocytes from 30 patients treated with tisa-cel. Bars represent the median. (C) Correlation between the naïve/stem cell memory phenotype and in vitro proliferative capacity (proportion of CFSElow cells) of apheresed CD8 (left panel) and CD4 (right panel) T cells. (D) The CD4/CD8 T-cell ratio in the final product correlates with the ratio of CD4 TN/SCM to CD8 TN/SCM cells in the apheresis (left) and the ratio of CFSElow CD4 to CD8 T cells (right). Pearson correlation test. ρ coefficient (R) and P values are indicated.
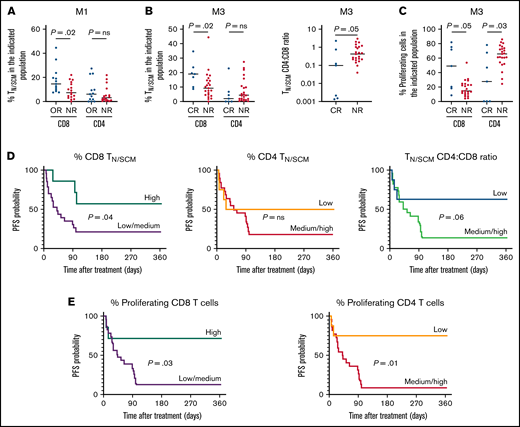
We then determined whether apheresed T-cell characteristics correlated with clinical outcome. Objective responses (partial or complete) around day 30 were observed in 13 out of 30 patients (43.3%). CD4/CD8 ratio in starting material had no impact on clinical response (not shown). However, early responders had a higher proportion of CD8 TN/SCM than nonresponders (mean 17.8% vs 9.0%; P = .02) while CD4 TN/SCM were not different (Figure 2A). At 3 months, 7 patients showed a complete response, which was associated with a higher proportion of CD8 TN/SCM (mean 19.0% vs 11.0% in nonresponders; P = .02) and a lower CD4 TN/SCM/CD8 TN/SCM ratio (mean 0.47 vs 0.73; P = .05) (Figure 2B). Moreover, complete responders had CD8 T cells with higher in vitro proliferative potential than nonresponders (mean 46.4% vs 18.4%; P = .05), while the opposite was observed for CD4 T cells (mean 31.7% vs 63.0%; P = .03) (Figure 2C). These data indicate that stemness and replicative potential of CD8 T cells at the time of apheresis are beneficial for the early response to CAR T cells, while those of CD4 T cells appear to be deleterious.
Characteristics of apheresed T cells and clinical responses to tisa-cel. (A) Higher frequency of CD8 TN/SCM, but not of CD4 TN/SCM, in the starting material from 13 patients with early objective response (OR; ie, partial or complete response) compared with 17 nonresponders (NR; stable or progressive disease) at 1 month (M1). Comparisons between groups based on the Mann-Whitney test. Two-sided P values are indicated. ns, not significant. (B) Higher proportion of CD8 TN/SCM (left panel) and lower CD4N/SCM/CD8N/SCM ratio (right panel) in the starting material from 7 complete responders (CR) compared with 23 NR at 3 months (M3). (C) In vitro proliferative capacity of apheresed CD8 T cells is higher and that of CD4 T cells is lower in CR than in NR at M3. Bars represent the median. (D) Effect of apheresed CD8 TN/SCM (left panel), CD4 TN/SCM (middle panel) cells, and CD4 TN/SCM/CD8 TN/SCM ratio (right panel) in the starting material on 1-year PFS. P values calculated using the log-rank test. “High” and “low” proportions refer to the 4th and 1st quartiles, respectively. (E) PFS is significantly increased in patients with “high” proportions of proliferating CD8 cells (left panel) and “low” proportions of proliferating CD4 T cells (right panel) in the starting material.
Characteristics of apheresed T cells and clinical responses to tisa-cel. (A) Higher frequency of CD8 TN/SCM, but not of CD4 TN/SCM, in the starting material from 13 patients with early objective response (OR; ie, partial or complete response) compared with 17 nonresponders (NR; stable or progressive disease) at 1 month (M1). Comparisons between groups based on the Mann-Whitney test. Two-sided P values are indicated. ns, not significant. (B) Higher proportion of CD8 TN/SCM (left panel) and lower CD4N/SCM/CD8N/SCM ratio (right panel) in the starting material from 7 complete responders (CR) compared with 23 NR at 3 months (M3). (C) In vitro proliferative capacity of apheresed CD8 T cells is higher and that of CD4 T cells is lower in CR than in NR at M3. Bars represent the median. (D) Effect of apheresed CD8 TN/SCM (left panel), CD4 TN/SCM (middle panel) cells, and CD4 TN/SCM/CD8 TN/SCM ratio (right panel) in the starting material on 1-year PFS. P values calculated using the log-rank test. “High” and “low” proportions refer to the 4th and 1st quartiles, respectively. (E) PFS is significantly increased in patients with “high” proportions of proliferating CD8 cells (left panel) and “low” proportions of proliferating CD4 T cells (right panel) in the starting material.
One-year estimates of progression-free survival (PFS) and overall survival (OS) were 26.7% and 53.3%, respectively, for the entire cohort (supplemental Figure 4A). Notably, the PFS was 57.1% in patients with a high proportion of CD8 TN/SCM (>18.6%; ie, the 4th quartile) but only 21.7% for the others (P = .04), while an opposite trend was observed for CD4 TN/SCM. Thus, a low ratio of CD4 TN/SCM/CD8 TN/SCM at apheresis was associated with higher PFS (Figure 2D). PFS was also associated positively with the in vitro proliferative potential of apheresed CD8 T cells but negatively with that of CD4 T cells (Figure 2E). Similar tendencies were observed in OS (supplemental Figure 4B-C). Finally, PFS tended to be higher in patients with a low CD4/CD8 ratio in the final product (among total T cells or CAR+ T cells) (supplemental Figure 4D).
We identified additional patient characteristics associated with early response (supplemental Table 3). Because our intent was to provide an early predictive marker to better aid in the decision whether to proceed with CAR T-cell manufacturing, we performed multivariate analysis, including only the parameters measured at the time of decision to treat. Higher International Prognostic Index and lower CD8 TN/SCM proportion were independently associated with lack of complete response at M3 (P = .009 and .05, respectively).
Altogether, these preliminary results on a small patient cohort show that the TN/SCM phenotype at the time of apheresis has a strong impact on the early and sustained response to tisa-cel in real life. Whether comparable results are observed in patients treated with axi-cel is under investigation. Such information, easily obtained using a simple flow cytometry analysis on a peripheral blood sample, could help prescreen patients to decide whether to proceed with CAR T-cell manufacturing in persons with expected benefits or propose an alternative treatment when there is very little chance of efficacy.
Contribution: A.C., S.C.-Z., and M.M. conceived the work and wrote the manuscript; M.M., J.L., and S.C.-Z. supervised the work; A.C., V.A., L.F., F.M., L.V., M.F.C., A.D., R.d.J., N.P., and C.T. performed the experiments and/or analyzed the data; and all authors read and approved the final manuscript.
Conflict-of-interest disclosure: C.T. received honoraria from Roche, Amgen, Janssen, Celgene, Gilead Science/Kite, and Beigene; reported a consulting/advisory role from Roche, Gilead Sciences, Janssen, Celgene, Novartis, and Beigene; and received research funding and travel and accommodations expenses from Roche and Novartis. J.L. received honoraria from Novartis, Kite/Gilead, BMS/Celgene, and Janssen. S.C.-Z. received honoraria from Novartis and Kite/Gilead. M.M. received honoraria from Novartis, Kite/Gilead, and Janssen. A.C.B. received support from Kite/Gilead for attending a meeting. R.d.J. received honoraria from Novartis and Kite/Gilead. The remaining authors declare no competing financial interests.
Correspondence: Sophie Caillat-Zucman, Laboratoire d’Immunologie, Hôpital Saint-Louis, Assistance Publique-Hôpitaux de Paris, Paris, France; e-mail: sophie.caillat-zucman@aphp.fr.
References
Author notes
Contact the corresponding author for data sharing: sophie.caillat-zucman@aphp.fr.
The full-text version of this article contains a data supplement.