Key Points
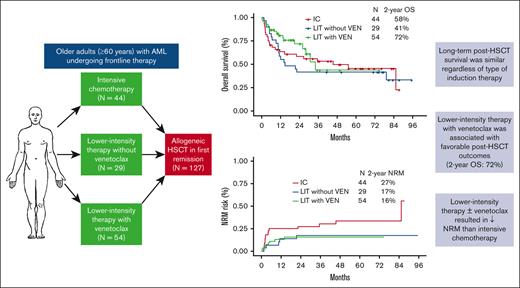
LIT plus venetoclax is associated with favorable posttransplant outcomes in older adults with AML (2-year OS: 72%).
The type of induction did not affect post HSCT survival, although LIT plus venetoclax could result in lower NRM than IC.
Abstract
Although venetoclax-based lower-intensity regimens have greatly improved outcomes for older adults with acute myeloid leukemia (AML) who are unfit for intensive chemotherapy, the optimal induction for older patients with newly diagnosed AML who are suitable candidates for hematopoietic stem cell transplant (HSCT) is controversial. We retrospectively analyzed the post HSCT outcomes of 127 patients ≥60 years of age who received induction therapy at our institution with intensive chemotherapy (IC; n = 44), lower-intensity therapy (LIT) without venetoclax (n = 29), or LIT with venetoclax (n = 54) and who underwent allogeneic HSCT in the first remission. The 2-year relapse-free survival (RFS) was 60% with LIT with venetoclax vs 54% with IC, and 41% with LIT without venetoclax; the 2-year overall survival (OS) was 72% LIT with venetoclax vs 58% with IC, and 41% with LIT without venetoclax. The benefit of LIT with venetoclax induction was greatest in patients with adverse-risk AML (2-year OS: 74%, 46%, and 29%, respectively). Induction with LIT, with or without venetoclax, was associated with the lowest rate of nonrelapse mortality (NRM) (2-year NRM: 17% vs 27% with IC; P = .04). Using multivariate analysis, the type of induction therapy did not significantly affect any of the post HSCT outcomes evaluated; hematopoietic cell transplantation-specific comorbidity index was the only factor that independently predicted RFS and OS. LIT plus venetoclax followed by HSCT is a feasible treatment strategy in older, fit, HSCT-eligible patients with newly diagnosed AML and may be particularly beneficial for those with adverse-risk disease.
Introduction
Acute myeloid leukemia (AML) is a disease of older adults, with a median age at diagnosis of 68 years, and approximately one-third of patients being ≥75 years of age at diagnosis.1 Historically, the outcomes for older adults with AML were dismal.2-4 The poor outcomes of this older AML population are multifactorial and driven by both host-related factors (eg, poorer tolerance of conventional chemotherapy) and disease-related factors (eg, higher incidence of adverse-risk cytogenetic and molecular features).5 Many older patients are not suitable candidates for intensive chemotherapy (IC) and/or allogeneic hematopoietic stem cell transplant (HSCT), thereby limiting their therapeutic options and chance for long-term survival.
The use of venetoclax in combination with hypomethylating agents (HMAs) or low-dose cytarabine has greatly improved the outcomes of older adults with AML and is now standard of care in this population.6-8 In registrational studies, these regimens were evaluated in patients who were considered unsuitable for IC because of advanced age (ie, those ≥75 years of age) or substantial comorbidities.6,8 For older adults with newly diagnosed AML aged between 60 and 74 years and otherwise appear fit for an intensive induction approach and subsequent HSCT, the appropriate choice of induction therapy is controversial.9 IC confers a greater risk for treatment-related morbidity and mortality in this older population than in the younger population.10 However, despite the encouraging results with lower-intensity venetoclax-based approaches in older/unfit patients, the outcomes of older but fit patients receiving these regimens are largely unknown, particularly for those who undergo subsequent HSCT.
Some retrospective studies have suggested that venetoclax-based lower-intensity therapy (LIT) followed by HSCT may be a feasible approach and can result in favorable outcomes.11-17 In 1 study of 21 patients ≥60 years of age who underwent HSCT after frontline therapy with azacitidine plus venetoclax, a 2-year overall survival (OS) of ∼70% to 75% was achieved.16 Although 2 analyses have also suggested similar post HSCT outcomes in patients who received IC or HMA plus venetoclax induction, these studies are limited by their relatively small sample size.11,13
Given the uncertainties about the optimal frontline treatment approach in older, HSCT-eligible patients, we sought to evaluate the impact of the type of induction therapy received upon relapse, treatment-related mortality, and survival outcomes in older adults who underwent HSCT in the first remission. In particular, we aimed to determine whether the use of IC or LIT, with or without venetoclax, affected post HSCT outcomes in these older patients who were fit for HSCT.
Methods
Study design and participants
This is a retrospective study evaluating the clinical impact of the type of induction therapy received among older adults with newly diagnosed AML undergoing allogeneic HSCT during the first remission. Eligible patients were ≥60 years of age, received frontline AML therapy at our institution, achieved complete remission (CR), CR with incomplete hematologic recovery, or morphologic leukemia-free state as the best response, and underwent allogeneic HSCT in the first remission. Patients were stratified based on 3 types of induction regimens: (1) IC (IC), (2) lower-intensity therapy (LIT) without venetoclax, or (3) LIT with venetoclax. Patients who received IC in combination with venetoclax were excluded from this analysis. We also excluded patients with treated secondary AML (ie, those who had received prior HMAs or chemotherapy for an antecedent myeloid malignancy), given the poor prognosis in this population, which is more akin to relapsed/refractory disease than to newly diagnosed disease.18 This study was conducted at a single academic center (The University of Texas MD Anderson Cancer Center). It was approved by the institutional review board of The University of Texas MD Anderson Cancer Center and conducted in accordance with the Declaration of Helsinki.
Response and outcome definitions
Responses were defined based on European LeukemiaNet consensus guidelines.19 Minimal residual disease (MRD) assessment was performed on fresh bone marrow aspiration samples using 8-color multiparameter flow cytometry with a sensitivity of 0.1% or higher, as described previously.20 MRD negativity was defined as the absence of a distinct cluster of at least 20 cells showing altered expression of ≥2 antigens in an adequate sample.21 Relapse-free survival (RFS) was calculated from the time of allogeneic HSCT until relapse or death from any cause, censored if the patient was alive at the last follow-up. OS was calculated from the time of allogenic HSCT until death from any cause and censored if the patient was alive at the last follow-up. Nonrelapse mortality (NRM) was defined as death occurring after HSCT in the absence of documented relapse. Graft-versus-host disease (GVHD) was graded per standard criteria.22,23
Statistical methods
Patient characteristics were summarized using the median (range) for continuous variables and frequencies (percentages) for categorical variables. To compare 2 groups, Fisher exact test was performed for categorical variables, and Wilcoxon rank-sum test was performed for continuous variables. The Kaplan-Meier method was used to estimate the probabilities for RFS and OS, and the differences between groups were evaluated with the log-rank test. Median follow-up was estimated using reverse Kaplan-Meier method.24 The method of Gooley et al was used to estimate the cumulative incidence of NRM, considering relapse as a competing event, and cumulative incidence of relapse (CIR), considering death as a competing event.25 Similarly, the cumulative incidence of GVHD was estimated with relapse or death without GVHD as competing events. CIR, NRM, and cumulative incidence of GVHD between groups were compared using the methods of Fine and Gray.26 Cox proportional hazards models and competing-risk regression models based on the method of Fine and Gray were used to evaluate the risk factors associated with survival outcomes. The multivariate models were obtained by first including the factors with P <.25 upon univariate analysis and then finalizing using backward elimination methods until all remaining factors had a P value < .05. The type of induction therapy was included in the multivariate models throughout all model-building steps, regardless of the significance level. Hazard ratios (HRs) and subdistributional hazard ratios (SHRs) with 95% confidence intervals (CIs) are reported for the time-to-event analyses. Statistical analyses were conducted in Stata version 17 and R version 4.1.0.
Results
Study population
Between November 2012 and July 2021, 127 patients ≥60 years of age received induction therapy at our institution and underwent subsequent allogeneic HSCT in the first remission. Rates of HSCT realization for older adults at our institution over this study period based on the type of induction are shown in supplemental Figure 1. The baseline characteristics of the study population are shown in Table 1. Forty-four patients (35%) had received IC as induction, 29 patients (23%) received LIT without venetoclax, and 54 patients (43%) received LIT with venetoclax. There was more use of LIT with venetoclax in the later part of the study period; among the 44 patients receiving frontline AML therapy after November 2018 (ie, after the US Food and Drug Administration approval of venetoclax for AML), 34 (77%) received LIT with venetoclax. Compared with those who received induction with LIT, with or without venetoclax, patients who received IC were younger (median age of 63 vs 68 years; P < .001), more likely to have an Eastern Cooperative Oncology Group performance status of 0 at the time of AML diagnosis (34% vs 15%; P = .02), and more likely to have de novo AML (80% vs 60%; P = .03), respectively. Patients who received LIT with venetoclax had a higher rate of RUNX1 mutation at diagnosis (22% vs 5% in other groups; P = .007) than those who received LIT without venetoclax. Most patients (≥80% in each group) had intermediate- or adverse-risk AML; only 2 patients had core-binding factor AML, both of whom received IC induction.
Baseline characteristics of study population (N = 127)
| Characteristic . | IC without VEN (N = 44) . | LIT without VEN (N = 29) . | LIT with VEN (N = 54) . | P value . |
|---|---|---|---|---|
| Age (y) | 63 (60-76) | 67 (61-75) | 68 (60-77) | < .0001 |
| ECOG performance status at diagnosis | ||||
| 0 | 15 (34) | 3 (10) | 9 (17) | .06 |
| 1 | 27 (61) | 21 (72) | 39 (72) | |
| 2 | 2 (5) | 5 (17) | 6 (11) | |
| AML type | ||||
| De novo AML | 35 (80) | 20 (69) | 30 (56) | .04 |
| Secondary AML | 9 (20) | 9 (31) | 24 (44) | |
| ELN 2017 risk | ||||
| Favorable∗ | 9 (20) | 4 (14) | 10 (19) | .66 |
| Intermediate | 16 (36) | 11 (38) | 14 (26) | |
| Adverse | 19 (43) | 14 (48) | 30 (56) | |
| Mutations | ||||
| NPM1 | 12 (27) | 7 (24) | 12 (22) | .85 |
| FLT3-ITD | 9 (20) | 7 (24) | 6 (11) | .26 |
| IDH1/IDH2 | 11 (25) | 3 (10) | 14 (26) | .22 |
| KRAS/NRAS | 5 (11) | 3 (10) | 12 (22) | .23 |
| RUNX1 | 3 (7) | 1 (3) | 12 (22) | .02 |
| ASXL1 | 4 (9) | 0 | 9 (17) | .06 |
| TP53 | 8 (18) | 6 (21) | 7 (13) | .62 |
| Characteristic . | IC without VEN (N = 44) . | LIT without VEN (N = 29) . | LIT with VEN (N = 54) . | P value . |
|---|---|---|---|---|
| Age (y) | 63 (60-76) | 67 (61-75) | 68 (60-77) | < .0001 |
| ECOG performance status at diagnosis | ||||
| 0 | 15 (34) | 3 (10) | 9 (17) | .06 |
| 1 | 27 (61) | 21 (72) | 39 (72) | |
| 2 | 2 (5) | 5 (17) | 6 (11) | |
| AML type | ||||
| De novo AML | 35 (80) | 20 (69) | 30 (56) | .04 |
| Secondary AML | 9 (20) | 9 (31) | 24 (44) | |
| ELN 2017 risk | ||||
| Favorable∗ | 9 (20) | 4 (14) | 10 (19) | .66 |
| Intermediate | 16 (36) | 11 (38) | 14 (26) | |
| Adverse | 19 (43) | 14 (48) | 30 (56) | |
| Mutations | ||||
| NPM1 | 12 (27) | 7 (24) | 12 (22) | .85 |
| FLT3-ITD | 9 (20) | 7 (24) | 6 (11) | .26 |
| IDH1/IDH2 | 11 (25) | 3 (10) | 14 (26) | .22 |
| KRAS/NRAS | 5 (11) | 3 (10) | 12 (22) | .23 |
| RUNX1 | 3 (7) | 1 (3) | 12 (22) | .02 |
| ASXL1 | 4 (9) | 0 | 9 (17) | .06 |
| TP53 | 8 (18) | 6 (21) | 7 (13) | .62 |
Continuous variables are listed as median (range) and categorical variables as N (%) or n/N (%).
ECOG, Eastern Cooperative Oncology Group; ELN, European LeukemiaNet; VEN, venetoclax.
Two patients (5%) in the IC group had core-binding factor AML.
The HSCT-related variables for the study population are shown in Table 2. Most patients in all groups achieved CR as the best response before HSCT (84% in the IC group, 83% in the LIT without venetoclax group, and 78% in the LIT with venetoclax group); 2 patients (7%) in the LIT without venetoclax group and 4 patients (7%) in the LIT with venetoclax group achieved morphologic leukemia-free state as the best response. Patients who received induction with LIT plus venetoclax were more likely to receive reduced-intensity conditioning (63% vs 34% with IC and 38% with LIT without venetoclax; P = .01). Patients who received LIT with venetoclax also had numerically lower rates of hematopoietic cell transplantation-specific comorbidity index (HCT-CI) scores of ≥3 (37% vs 59% for IC and 52% for LIT without venetoclax groups). The median number of cycles before HSCT was 3 in LIT with venetoclax and IC groups and 4 in LIT without venetoclax group (P = .01). The rates of post HSCT maintenance therapy were 9% in the IC group, 24% in the LIT without venetoclax group, and 15% in the LIT with venetoclax group.
HSCT-related variables for study population (N = 127)
| Characteristic . | IC without VEN (N = 44) . | LIT without VEN (N = 29) . | LIT with VEN (N = 54) . | P value∗ . |
|---|---|---|---|---|
| Best hematologic response before HSCT | ||||
| CR | 37 (84) | 24 (83) | 42 (78) | .44 |
| CRi | 7 (16) | 3 (10) | 8 (15) | |
| MLFS | 0 | 2 (7) | 4 (7) | |
| MRD status before HSCT | ||||
| MRD– | 23 (52) | 11 (38) | 28 (52) | .03 |
| MRD+ | 17 (39) | 17 (59) | 11 (20) | |
| Insufficient/not done | 4 (9) | 1 (3) | 15 (28) | |
| Number of cycles before HSCT | 3 (1-6) | 4 (1-12) | 3 (1-12) | .01 |
| Months from diagnosis to HSCT | 5.0 (2.0-13.5) | 5.0 (2.8-16.5) | 4.2 (2.1-17.0) | .03 |
| Transplant conditioning intensity† | ||||
| MAC | 28 (64) | 18 (62) | 20 (37) | .01 |
| RIC | 15 (34) | 11 (38) | 34 (63) | |
| Unknown | 1 (2) | 0 | 0 | |
| GVHD prophylaxis | ||||
| MTX + tacrolimus ± MMF | 25 (57) | 22 (76) | 8 (15) | <.0001 |
| Cyclophosphamide + tacrolimus ±MMF | 16 (36) | 7 (24) | 45 (83) | |
| Other | 2 (5) | 0 | 0 | |
| Unknown | 1 (2) | 0 | 1 (2) | |
| HCT-CI score | ||||
| 0 | 5 (11) | 5 (17) | 10 (19) | .27 |
| 1-2 | 13 (30) | 9 (31) | 24 (44) | |
| ≥3 | 26 (59) | 15 (52) | 20 (37) | |
| HLA matching | ||||
| Matched unrelated donor | 22 (50) | 20 (69) | 29 (54) | .45 |
| Matched related donor | 15 (34) | 8 (28) | 16 (30) | |
| Haploidentical donor | 6 (14) | 1 (3) | 8 (15) | |
| Unknown | 1 (2) | 0 | 1 (2) | |
| Received post HSCT maintenance | 4 (9) | 7 (24) | 8 (15) | .37 |
| HMA alone | 3 (7) | 5 (17) | 5 (9) | |
| HMA plus venetoclax | 0 | 2 (7) | 1 (2) | |
| FLT3 inhibitor | 1 (2) | 0 | 2 (4) | |
| None | 40 (91) | 22 (76) | 46 (85) |
| Characteristic . | IC without VEN (N = 44) . | LIT without VEN (N = 29) . | LIT with VEN (N = 54) . | P value∗ . |
|---|---|---|---|---|
| Best hematologic response before HSCT | ||||
| CR | 37 (84) | 24 (83) | 42 (78) | .44 |
| CRi | 7 (16) | 3 (10) | 8 (15) | |
| MLFS | 0 | 2 (7) | 4 (7) | |
| MRD status before HSCT | ||||
| MRD– | 23 (52) | 11 (38) | 28 (52) | .03 |
| MRD+ | 17 (39) | 17 (59) | 11 (20) | |
| Insufficient/not done | 4 (9) | 1 (3) | 15 (28) | |
| Number of cycles before HSCT | 3 (1-6) | 4 (1-12) | 3 (1-12) | .01 |
| Months from diagnosis to HSCT | 5.0 (2.0-13.5) | 5.0 (2.8-16.5) | 4.2 (2.1-17.0) | .03 |
| Transplant conditioning intensity† | ||||
| MAC | 28 (64) | 18 (62) | 20 (37) | .01 |
| RIC | 15 (34) | 11 (38) | 34 (63) | |
| Unknown | 1 (2) | 0 | 0 | |
| GVHD prophylaxis | ||||
| MTX + tacrolimus ± MMF | 25 (57) | 22 (76) | 8 (15) | <.0001 |
| Cyclophosphamide + tacrolimus ±MMF | 16 (36) | 7 (24) | 45 (83) | |
| Other | 2 (5) | 0 | 0 | |
| Unknown | 1 (2) | 0 | 1 (2) | |
| HCT-CI score | ||||
| 0 | 5 (11) | 5 (17) | 10 (19) | .27 |
| 1-2 | 13 (30) | 9 (31) | 24 (44) | |
| ≥3 | 26 (59) | 15 (52) | 20 (37) | |
| HLA matching | ||||
| Matched unrelated donor | 22 (50) | 20 (69) | 29 (54) | .45 |
| Matched related donor | 15 (34) | 8 (28) | 16 (30) | |
| Haploidentical donor | 6 (14) | 1 (3) | 8 (15) | |
| Unknown | 1 (2) | 0 | 1 (2) | |
| Received post HSCT maintenance | 4 (9) | 7 (24) | 8 (15) | .37 |
| HMA alone | 3 (7) | 5 (17) | 5 (9) | |
| HMA plus venetoclax | 0 | 2 (7) | 1 (2) | |
| FLT3 inhibitor | 1 (2) | 0 | 2 (4) | |
| None | 40 (91) | 22 (76) | 46 (85) |
Continuous variables are listed as median (range) and categorical variables as N (%) or n/N (%).
CRi, complete remission with incomplete count recovery; MAC, myeloablative conditioning; MLFS, morphologic leukemia-free state; MMF, mycophenolate mofetil; MTX, methotrexate; RIC, reduced-intensity conditioning; VEN, venetoclax.
Two patients with a haploidentical donor also received 5 mg/kg of thiotepa.
RIC: Fludarabine ranging from 25 to 30 mg/m2 with melphalan 100 mg/m2 or 140 mg/m2.27 Thirteen patients with haploidentical donors also received 200 cGy of total body irradiation.
P value is for comparison of groups with available data only (excluding those with unknown values).
MAC: Fludarabine 40 mg/m2 with pharmacokinetic-guided busulfan dose to reach target total course area under the curve from 16 000 to 24 000 μmol/min.28
CIR and NRM by induction therapy
The median duration of follow-up since HSCT for the 3 groups were 59.8 months (95% CI, 42.3-68.4) for the IC group, 76.8 months (95% CI, 50.7-83.5) for the LIT without venetoclax group, and 20.3 months (95% CI, 15.4-27.7) for the LIT with venetoclax group. The 2-year CIR for patients who received IC and LIT with venetoclax were 19% and 24%, respectively, and were lower than in the LIT without venetoclax group, in which the 2-year CIR was 41% (P = .05 for IC and LIT with venetoclax vs LIT without venetoclax) (Figure 1A). In contrast, the 2-year NRM was lower in those who received LIT with venetoclax (16%) or without venetoclax (17%) than in those who received IC (27%) (Figure 1B). NRM was significantly higher in patients who received IC induction (2-year NRM of 27%) compared with those who received LIT with or without venetoclax (2-year NRM of 17%; P = .04). Induction with IC was associated with a higher rate of neutropenic fever (61% vs 43% with LIT with or without venetoclax; P = .06) but not with rates of serious infections (supplemental Table 1). The type of induction was not significantly associated with the cumulative incidence of acute or chronic GHVD (supplemental Figures 2-3).
Posttransplant CIR and NRM, stratified based on the type of induction therapy. (A) CIR and (B) NRM.
Posttransplant CIR and NRM, stratified based on the type of induction therapy. (A) CIR and (B) NRM.
Among patients who were MRD– before HSCT, a similar pattern was observed with a higher CIR in patients who received LIT without venetoclax than in those who received IC or LIT with venetoclax (2-year CIR: 45% vs 23% and 17%, respectively; supplemental Figure 4). Among patients with favorable/intermediate-risk AML, the 2-year CIR rates for IC, LIT without venetoclax, and LIT with venetoclax were 16%, 27%, and 25%, respectively (supplemental Figure 5). In contrast, the type of induction therapy received had the greatest impact among patients with adverse-risk AML, in which the 2-year CIR for patients who received LIT without venetoclax was 57%, compared with 22% for IC and 23% for LIT with venetoclax (supplemental Figure 6).
RFS and OS using induction therapy
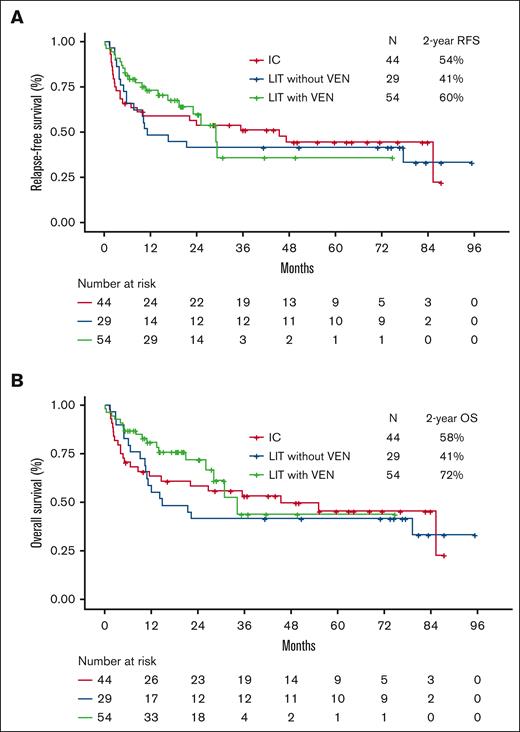
Post HSCT RFS and OS by type of induction therapy received are shown in Figure 2. Both the 2-year RFS and OS were higher in patients who had received induction with LIT with venetoclax than in those who received IC or LIT without venetoclax. The 2-year RFS was 60% for patients who received LIT with venetoclax vs 54% for those who received IC and 41% for those who received LIT without venetoclax (Figure 2A). Similarly, the 2-year OS was 72% in patients who received LIT with venetoclax induction vs 58% in the IC group and 41% in the LIT without venetoclax group (Figure 2B). These differences were driven largely by the higher rates of NRM in the IC group and the higher rates of relapse in the LIT without venetoclax group as compared with the NRM and relapse rates in the LIT with venetoclax group. The 2-year RFS and OS for patients who received LIT with venetoclax induction and achieved MRD negativity were 68% and 78%, respectively (supplemental Figure 7). Post HSCT RFS and OS in patients with favorable/intermediate-risk AML were similar among all 3 treatment groups (supplemental Figure 8). In contrast, post HSCT survival outcomes were superior for patients with adverse-risk AML who received LIT with venetoclax induction than for those who received other induction regimens (supplemental Figure 9). The 2-year RFS was 55% for patients with adverse-risk disease who received LIT with venetoclax, compared with 41% with IC and 29% with LIT without venetoclax. Similarly, the 2-year OS was 74% for patients with adverse-risk disease who received LIT with venetoclax, compared with 46% with IC and 29% with LIT without venetoclax (P = .09 for LIT with venetoclax vs IC; P = .11 for LIT with venetoclax vs LIT without venetoclax). In patients with TP53-mutated AML, LIT with venetoclax was associated with superior RFS and OS compared with that in those who received IC or LIT without venetoclax induction (P = .05 for RFS; P = .02 for OS), although the number of patients for this analysis is limited (supplemental Figure 10). In these patients, LIT without venetoclax was associated with a particularly high 1-year CIR of 67%, and IC was associated with a particularly high 1-year NRM of 63%.
Posttransplant RFS and OS, stratified based on the type of induction therapy. (A) RFS and (B) OS.
Posttransplant RFS and OS, stratified based on the type of induction therapy. (A) RFS and (B) OS.
Univariate and multivariate analyses for post HSCT outcomes
A univariate analysis was performed to evaluate predictors of CIR, NRM, RFS, and OS. Variables assessed in the univariate analysis included age, induction therapy, AML risk group and type (ie, de novo vs secondary), HSCT conditioning intensity, type of GVHD prophylaxis, HLA matching of HSCT, HCT-CI score, year of transplantation, CR or lesser response before HSCT, and pre HSCT MRD. The full univariate analysis is shown in supplemental Table 2. Only HCT-CI ≥3 (SHR, 2.21 [95% CI, 1.07-4.57]; P = .03) was associated with a significantly increased risk of relapse, although there was also a trend toward higher rates of relapse for patients with adverse-risk disease (SHR, 1.97 [95% CI, 0.94-4.13]; P = .07) and using LIT without venetoclax (SHR, 2.16 [95% CI, 0.88-5.28]; P = .09). No statistically significant predictors for NRM were observed, although there was a trend toward a higher risk of NRM with a haploidentical donor (SHR, 2.79 [95% CI, 0.97-8.03]; P = .06). Factors associated with inferior RFS included adverse-risk disease (HR, 1.70 [95% CI, 0.99-2.91]; P = .05), MRD positivity before HSCT (HR, 1.79 [95% CI, 1.05-3.05]; P = .03), and HCT-CI ≥3 (HR, 2.54 [95% CI, 1.46-4.41]; P = .001). Factors associated with inferior OS included MRD positivity before HSCT (HR, 1.78 [95% CI, 1.02-3.09]; P = .04) and HCT-CI ≥3 (HR, 2.62 [95% CI, 1.47-4.68]; P = .001).
The multivariate analysis is shown in Table 3. No statistically significant predictors for post HSCT relapse or NRM were observed. HCT-CI ≥3 was the only factor that independently predicted the RFS (HR, 1.90 [95% CI, 1.11-3.25]; P = .02) and OS (HR, 2.09 [95% CI, 1.20-3.65]; P = .01). Notably, the type of induction therapy received was not statistically significant for any of the post HSCT outcomes evaluated using multivariate analysis.
Multivariate analysis of post HSCT outcomes
| Variable . | OS . | RFS . | Relapse . | NRM . | ||||
|---|---|---|---|---|---|---|---|---|
| HR (95% CI) . | P . | HR (95% CI) . | P . | SHR (95% CI) . | P . | SHR (95% CI) . | P . | |
| Treatment | ||||||||
| IC without VEN | Reference | Reference | Reference | Reference | ||||
| LIT without VEN | 1.10 (0.58-2.11) | .76 | 1.04 (0.55-1.98) | .90 | 1.97 (0.80-4.83) | .14 | 0.50 (0.18-1.38) | .18 |
| LIT with VEN | 0.71 (0.35-1.43) | .34 | 0.81 (0.42-1.56) | .53 | 1.21 (0.49-3.00) | .68 | 0.54 (0.20-1.43) | .21 |
| HCT-CI Score | ||||||||
| Low-/intermediate-risk (0-2) | Reference | Reference | — | — | ||||
| High-risk (≥3) | 2.09 (1.20-3.65) | .01 | 1.90 (1.11-3.25) | .02 | — | — | ||
| Variable . | OS . | RFS . | Relapse . | NRM . | ||||
|---|---|---|---|---|---|---|---|---|
| HR (95% CI) . | P . | HR (95% CI) . | P . | SHR (95% CI) . | P . | SHR (95% CI) . | P . | |
| Treatment | ||||||||
| IC without VEN | Reference | Reference | Reference | Reference | ||||
| LIT without VEN | 1.10 (0.58-2.11) | .76 | 1.04 (0.55-1.98) | .90 | 1.97 (0.80-4.83) | .14 | 0.50 (0.18-1.38) | .18 |
| LIT with VEN | 0.71 (0.35-1.43) | .34 | 0.81 (0.42-1.56) | .53 | 1.21 (0.49-3.00) | .68 | 0.54 (0.20-1.43) | .21 |
| HCT-CI Score | ||||||||
| Low-/intermediate-risk (0-2) | Reference | Reference | — | — | ||||
| High-risk (≥3) | 2.09 (1.20-3.65) | .01 | 1.90 (1.11-3.25) | .02 | — | — | ||
Discussion
In this study, older adults with newly diagnosed AML who received frontline LIT with venetoclax followed by HSCT had relatively favorable outcomes, with a 2-year RFS of 60% and a 2-year OS of 72%. Overall, these data support LIT with venetoclax as a feasible approach for older, HSCT-eligible patients with newly diagnosed AML.
The optimal frontline approach for older but fit patients with newly diagnosed AML is controversial. Although IC induction was once considered the only potentially curative strategy for these patients, the development of safe and effective lower-intensity venetoclax-based regimens has raised questions about the need for intensive approaches in this population,29 even in those who are candidates for HSCT. In our analysis, the relatively favorable outcomes observed in patients who received frontline LIT with venetoclax provide further rationale for consideration of this regimen in older, HSCT-eligible patients with AML. Although we did not observe a difference in relapse rates between these 2 approaches, the encouraging survival in the LIT with venetoclax cohort might have been, in part, driven by a lower rate of NRM compared with those who had received IC (2-year NRM: 16% vs 27%, respectively). One possible reason for the difference in NRM between these 2 groups could be that patients treated with LIT with venetoclax experienced less treatment-related morbidity than those treated more intensively, because some prior analyses have shown higher rates of early morbidity and death in older patients treated with IC as compared with such rates among those treated with LIT plus venetoclax.30 However, given the higher rate of reduced-intensity conditioning in this group (63% vs 34%-38%) and the more frequent use of post HSCT cyclophosphamide (83% vs 24%-36%), perhaps driven by changing the HSCT practice within the study period, we cannot rule out that differences in conditioning intensity or GVHD prophylaxis were also a major driver of these differences. Although these variables have been shown, in other analyses, to affect post HSCT outcomes (particularly NRM), it is worth noting that they were statistically significant in neither our univariate nor multivariate analyses.
In a multivariate analysis, the induction regimen was not significantly associated with any post HSCT outcomes. The only factor that was significantly associated with RFS and OS was HCT-CI. Interestingly, neither pre HSCT MRD status nor cytomolecular risk were associated with these survival end points, contrary to several other studies.31,32 This lack of statistical significance may be partly due to the sample size of our cohort (127 total patients), but it also highlights the paramount importance of comorbidities and frailty assessments in predicting post HSCT outcomes in older adults with AML undergoing HSCT.33-36
Our findings are consistent with those of other small reports that have suggested that frontline LIT with venetoclax followed by HSCT in the first remission yields relatively favorable outcomes in patients with AML compared with IC followed by HSCT.11-17 In an analysis of 24 patients who received frontline azacitidine plus venetoclax followed by HSCT in the first remission (median age, 72 years), the estimated 1-year RFS and OS were 58% and 63%, respectively.11 These outcomes were similar to those of a younger (median age, 58 years) historical cohort of patients who had received IC and HSCT.11 In another analysis, the outcomes of 29 patients who received frontline azacitidine plus venetoclax and HSCT were compared with those of 140 patients who received IC and HSCT.13 Despite the generally more favorable characteristics of the intensively treated population, that is, younger median age (56 vs 65 years) and lower rates of adverse cyto-molecular features (41% vs 66%), RFS and OS were similar in the 2 groups.
Previous studies have largely focused on post HSCT outcomes in patients treated with IC and LIT plus venetoclax. In contrast, our study also evaluated patients who received LIT without venetoclax. Although it is established that the addition of venetoclax to HMA improves OS in older patients unfit for IC,6 whether these approaches result in differential post HSCT outcomes has not been systemically explored. Among the 3 frontline treatment approaches evaluated, LIT without venetoclax resulted in the worst RFS and OS. Although various factors could have contributed to these different outcomes, it is notable that half of the patients treated with LIT without venetoclax were MRD+ before HSCT. Thus, lesser depths of response with this induction approach might have accounted for the relatively inferior outcomes among these patients.
To our knowledge, our analysis represents the largest published cohort of patients treated with LIT with venetoclax followed by HSCT and is also the largest one comparing IC and LIT with venetoclax in an older, HSCT-eligible population. We were therefore able to assess the outcomes of these approaches in different subgroups. A particularly important finding from our study is the better survival outcomes in patients with adverse-risk disease who received LIT with venetoclax as compared with those who received IC. Although broadly treating all patients ≥60 years of age is controversial, there is a growing consensus for treating older adults with adverse-risk AML with LIT with venetoclax rather than IC. This is supported by the most recent National Comprehensive Cancer Network guidelines.37 Our findings support frontline LIT with venetoclax followed by HSCT in older adults with newly diagnosed AML as a reasonable approach that may be superior to using IC induction in these patients.
This study has several limitations. Retrospective analyses comparing intensive and nonintensive treatment approaches in AML are often confounded because younger and fitter patients are generally selected for intensive therapy and older and less fit patients receive less-intensive approaches. Although we observed this same pattern in our data set, with patients treated with IC being younger than those treated with LIT with or without venetoclax, our analysis benefits from being limited to patients ≥60 years of age, in contrast with other reports in which patients of all ages were included in comparisons of IC and LIT approaches.11,13 Furthermore, in the past few years, our institution’s preferred frontline induction for patients ≥60 years of age has been LIT venetoclax-based therapy, regardless of perceived fitness; in fact, 77% of patients in this analysis who were treated after the US Food and Drug Administration approval of venetoclax for AML received LIT plus venetoclax. Although there was minimal bias in treatment selection within this period due to patient fitness, we acknowledge that patients who received IC and LIT with venetoclax were treated in different period (generally after 2018). Thus, advances in treatments for relapsed/refractory AML, better supportive care, and changes in HSCT-related procedures and practices could have affected the outcomes of patients in the IC and LIT plus venetoclax cohorts. Finally, our study only evaluated patients who proceeded to allogeneic HSCT in first remission. Data on the proportion of HSCT-eligible patients treated with IC or LIT with venetoclax who are ultimately able to proceed with HSCT in first remission would provide estimates of the relative efficacy of these approaches in bridging patients to HSCT, an important consideration when evaluating their feasibility in HSCT-eligible patients. An ongoing randomized, phase 2 study comparing frontline IC and HMA plus venetoclax in patients with newly diagnosed AML (irrespective of age) might help to answer this important question (NCT04801797).
In conclusion, frontline LIT plus venetoclax followed by HSCT is feasible in older adults with newly diagnosed AML. This approach resulted in a 2-year OS of 72%. These results compare favorably to those achieved with IC induction followed by HSCT in this population, particularly in patients with adverse-risk disease. This study provided further evidence supporting frontline LIT plus venetoclax in older, fit, and HSCT-eligible patients with newly diagnosed AML.
Acknowledgment
This research was funded in part by the National Institutes of Health/National Cancer Institute Cancer Center (support grant P30) (CA016672).
Authorship
Contribution: N.J.S. designed the study, collected and analyzed the data, treated patients, and wrote the manuscript; F.O. designed the study, collected and analyzed the data, performed statistical analyses, and wrote the manuscript; F.R., T.M.K., N.D., C.D.D., M.K., G.B., E.J.S., B.O., G.A-A., R.M., E.J.J., M.Y., G.C.I., A.M., R.E.C., and U.P. treated patients; G.N.-G. performed statistical analyses; and all authors critically reviewed and approved the manuscript.
Conflict-of-interest disclosure: F.R., C.D.D., and M.K. received honoraria and research funding from AbbVie. G.B. received honoraria from AbbVie. U.P. received research funding from AbbVie. The remaining authors declare no competing financial interests.
Correspondence: Nicholas J. Short, Department of Leukemia, Unit 428, The University of Texas MD Anderson Cancer Center, 1515 Holcombe Blvd, Houston, TX 77030; e-mail: nshort@mdanderson.org.
References
Author notes
∗N.J.S. and F.O. contributed equally to this study.
Data are available upon request from the corresponding author, Nicholas J. Short (nshort@mdanderson.org).
The full-text version of this article contains a data supplement.