Abstract
Protein Z deficiencies have recently been described in women with unexplained early fetal loss. Using a new, specifically elaborated, commercially available enzyme-linked immunosorbent assay (ELISA), we performed a case-control study on anti–protein Z immunoglobulin G (IgG) and IgM antibodies in 191 nonthrombotic, nonthrombophilic women with consecutive pathologic pregnancies. Levels of anti–protein Z antibodies were categorized in 3 strata (percentiles 1 through 74, 75 through 97, 98 through 100 among controls). The 2 upper levels of IgG and IgM anti–protein Z antibodies were associated with the risk of unexplained recurrent embryo loss or fetal death independently from habitual antiphospholipid/anticofactor antibodies, and a dose-effect relationship between antibody levels and the clinical risks was evidenced. In women, enhanced immune-complex formation with protein Z may play a role in unexplained embryo losses and, from the 10th week of gestation, may favor hypercoagulability in the maternal placenta side.
Introduction
Protein Z is a vitamin K–dependent plasma protein that serves as a cofactor for the control of the coagulation factor Xa by the protein Z–dependent protease inhibitor.1,2
We have recently described the incidence of protein Z deficiency in women with unexplained pregnancy loss, showing that concentrations lower than 1 mg/L mainly increase the risk of a first early fetal demise between the beginning of the 10th and the end of the 15th weeks of gestation,3 but not the risk of recurrent embryo loss before the 8th week.
Here we describe the incidence of anti–protein Z antibodies in women with abnormal pregnancies. The affinity of protein Z for negatively charged phospholipids also led us to examine the relationship between anti–protein Z antibodies and conventional antiphospholipid/anticofactor antibodies.
Study design
Patients
All the women we studied had given their informed consent for participation in our Abnormal Pregnancy Study Program, which is based on the recruitment of a vast cohort of patients from the south of France with at least one miscarriage/fetal loss, or with pre-eclampsia or placental abruption, and on the recruitment of healthy parity- and age-matched controls (Nîmes Obstetricians and Haematologists Study [NOHA]).4-6 Approval was obtained from the Nîmes University Hospital institutional review board for these studies. Informed consent was provided according to the Declaration of Helsinki.
In this program, all participants were investigated for classical prothrombotic factors as described (essential thrombocythemia; dysfibrinogenemia; deficiency in antithrombin, protein C, or protein S; factor V Leiden mutation; 20210A allele in the prothrombin gene; high plasminogen activator inhibitor–1 [PAI-1] plasma levels; and hyperhomocysteinemia).
In this work, we first considered 216 consecutive patients investigated between January 1, 2001, and April 1, 2001. We included the 171 women with negative thrombotic antecedents and prothrombotic factors. They were divided into 3 groups: 112 patients with unexplained primary recurrent embryo losses before the 8th week of gestation (at least 3 episodes; median age, 25.4 years); 41 patients with 1 unexplained episode of fetal death from the 10th week (last pregnancy; median age, 23.8 years); and 18 patients with 1 episode of severe pre-eclampsia that had necessitated admission to our obstetric intensive care unit (last pregnancy; median age, 24.3 years). We also studied a group of 20 patients known to be carriers of an association of unexplained primary recurrent embryo losses with a protein Z deficiency (less than 1 mg/L) (median age, 24.7 years), leading to the final inclusion of 191 patients.
A control group of 191 healthy mothers, matched for age, number of pregnancies, and time elapsed since the end of the last pregnancy, with negative thrombotic antecedents and prothrombotic factors, was concomitantly recruited during the same period of time as patients, among consecutive women attending the outpatient department of gynecology for a systematic medical exam.
Methods
Blood samples were collected, processed, and stored at –80°C, according to standard procedures, at least 6 months after the last obstetric accident, in nontreated patients.
Protein Z plasma concentrations were measured by means of a commercially available enzyme-linked immunosorbent assay (Asserachrom Protein Z; Diagnostica Stago and Serbio, Asnières, France).
Autoantibodies. Testing for antiphospholipid/anticofactor antibodies included testing for plasma lupus anticoagulant activity, anticardiolipin immunoglobulin G (IgG) and IgM antibodies, and anti–β2-glycoprotein I IgG or IgM antibodies, according to Gris et al.6
Anti–protein Z IgG and IgM antibodies were assayed by means of a research enzyme-linked immunosorbent assay (ELISA) kit (Zymutest Anti–Protein Z) developed by Hyphen BioMed (Andrésy, France). Highly purified human protein Z was extracted from citrated human plasma following barium citrate absorption-elution, ammonium sulfate salt precipitation, and double ion exchange chromatography on diethylaminoethane [DEAE]–Sepharose and on DEAE-Sephacel; this protein Z preparation was characterized by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and with the use of monoclonal antibodies specific for protein Z. The highly purified human protein Z was coated onto micro-ELISA plates (Nunc type I, Maxisorb, Roskilde, Denmark), at 10 μg/mL in a 0.05 M sodium phosphate buffer at pH 7.50. After being washed with physiologic saline containing 0.1% Tween 20, the plates were saturated by incubation for 1 hour at room temperature with a 0.05 M sodium phosphate and 0.15 M NaCl buffer at pH 7.50 (phosphate-buffered saline [PBS]) containing 10% goat serum. Plasma samples were tested at a dilution of 1:100 in PBS containing 10% goat serum. We prepared the calibration curves and the positive controls from chimeras obtained by coupling an affinity-purified rabbit polyclonal antibody specific for protein Z with purified human IgG or IgM. We initially tested a panel of 50 healthy individuals together with dilutions of the corresponding chimeras: the mean absorbance value in healthy individuals plus 2 standard deviations was said to correspond, by definition, to 10 arbitrary units (AUs) of anti–protein Z IgG or IgM antibodies. The between- and within-assay coefficients of variation were evaluated with the use of 3 plasmas (8, 15, and 50 AU) tested 5 times on 5 different occasions and were lower than 2.2%.
Statistical analysis. Results are described by using the Stat View 5 software (Abacus Concepts, Berkeley, CA) and nonparametric statistics (median, lower-upper quartiles values, range). Associations between continuous biomarkers were examined by means of Spearman correlation coefficients (p). Association between categorical variables was tested after cross-tabulation by the Pearson chi square and the Fisher exact tests. A women negative for the 5 antiphospholipid/anticofactor antibodies was categorized as having a normal status for antiphospholipid antibodies but an abnormal one in the case of any positivity. All comparisons of cases and controls were conducted by means of nonparametric methods: Mann-Whitney rank-sum test and Kruskal-Wallis analysis of variance (ANOVA) by ranks test for continuous variables. The relationships between the global risk of pathologic pregnancies and the levels of anti–protein Z antibodies were assessed by means of logistic regression analysis.
Anti–protein Z IgM and anti–protein Z IgG antibodies were finally categorized into 3 levels, corresponding to percentiles 1 through 74, 75 through 97 (IgG values between 7.1 and 14 AU; IgM values between 5.3 and 11.9 AU), and 98 through 100 among control women, and multiparametric logistic regression analysis for odds ratios corresponding to the 2 upper levels and an abnormal status for antiphospholipid antibodies was performed, the control group of healthy mothers being the reference. P < .05 was considered statistically significant.
Results and discussion
Measurements of autoantibody levels in patients were not substantially correlated with corresponding measurements in controls (all Spearman correlation coefficients [p] between –0.14 and +0.18). In both patients and controls, only anticardiolipin IgG and anti–β2-glycoprotein 1 IgG (p = 0.27), or anticardiolipin IgM and anti–β2-glycoprotein 1 IgM (p = 0.31), were moderately correlated. Anti–protein Z antibodies were not correlated with other autoantibodies. The rates of abnormal status for antiphospholipid/anticofactor antibodies were globally different in the 5 groups of women (P < .0001; control women, 1 of 191; women with unexplained primary recurrent embryo losses, 36 of 112; women with an unexplained episode of fetal death, 9 of 41; women with severe pre-eclampsia, 3 of 18; women with an association of unexplained primary recurrent embryo losses with a protein Z deficiency, 4 of 20).
Anti–protein Z IgG and IgM antibody levels were not correlated with age (P = .08 and .46, respectively) or with the time elapsed since the end of the last pregnancy (P = .13 and P = .19, respectively). Only slightly higher levels of anti–protein Z IgG and IgM were detected in controls with at least 4 pregnancies (n = 114; P = .02), showing a discrete pregnancy effect on antibody levels, but consistently higher levels of antibodies were evidenced in patients (n = 114 cases; P < .001). This apparent discrepancy is due to the fact that most of the patients with at least 4 pregnancies were members of the subgroup with early recurrent embryo losses (74 of the 112 women with no protein Z deficiency plus the 20 with protein Z deficiency) in which antibody levels are high. The remaining patients with at least 4 pregnancies were 12 women with a fetal death and 8 with severe pre-eclampsia. It may be surprising that a number of patients had successful pregnancies. By definition, their pathologic pregnancies were always the last ones; these pregnancies were always consecutive in case of recurrent ones; and autoantibodies are acquired biologic markers, not constitutional ones.
Anti–protein Z IgG and IgM antibody levels were not correlated with plasma protein Z concentrations (controls and patients). We found, however, a good inverse correlation between anti–protein Z IgM antibody levels and protein Z concentrations in a subgroup of patients: those with recurrent embryo losses and protein Z deficiency (p =–0.43). We thus looked at these previously studied samples of women with unexplained pregnancy loss3 : Levels of anti–protein Z IgG and IgM antibodies were not statistically different in protein Z–deficient and nondeficient individuals (P = .78 and P = .32, respectively).
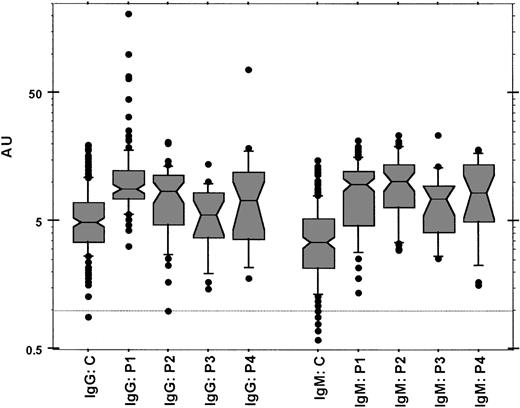
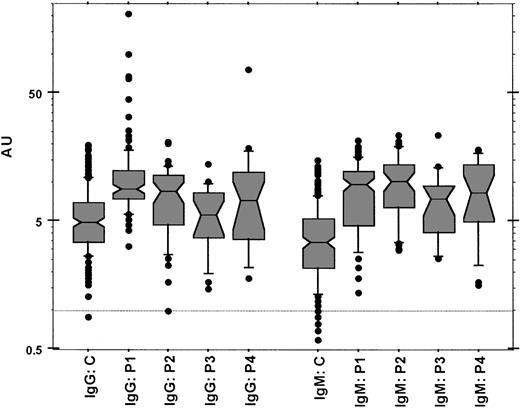
Figure 1 shows the distributions of anti–protein Z IgG and IgM levels in the 5 groups of women. Higher levels of anti–protein Z IgG and IgM antibodies were detected in patients than in controls (P < .0001). The global risk of pathologic pregnancies increased with increasing levels of anti–protein Z antibodies. (Mean variation of risk for an increase of 1 AU was as follows: IgG, odds ratio 1.2, 95% confidence interval 1.13-1.27, P < 10–4; IgM, mean odds ratio 1.42, 95% confidence interval 1.32-1.52, P < 10–4.) IgG levels were higher in women with recurrent embryo losses and in women with fetal death than in controls (P < .0001 and P = .0007, respectively). IgM levels were higher in women with recurrent embryo losses, fetal death, severe pre-eclampsia, or recurrent embryo losses and protein Z deficiency than in controls (P < .0001, P < .0001, P = .0002, and P < .0001, respectively).
Box-and-whiskers plot showing the distribution of anti–protein Z IgG and IgM antibody levels in the 5 groups of women. Data are given in arbitrary units (AUs) as described. C indicates control healthy women; P1, patients with at least 3 unexplained primary recurrent embryo losses before the 8th week of gestation; P2, patients with one unexplained episode of fetal death from the 10th week; P3, patients with one episode of severe pre-eclampsia; P4, patients known to be carriers of an association of unexplained primary recurrent embryo losses with a protein Z deficiency. The horizontal line represents the 1-AU level.
Box-and-whiskers plot showing the distribution of anti–protein Z IgG and IgM antibody levels in the 5 groups of women. Data are given in arbitrary units (AUs) as described. C indicates control healthy women; P1, patients with at least 3 unexplained primary recurrent embryo losses before the 8th week of gestation; P2, patients with one unexplained episode of fetal death from the 10th week; P3, patients with one episode of severe pre-eclampsia; P4, patients known to be carriers of an association of unexplained primary recurrent embryo losses with a protein Z deficiency. The horizontal line represents the 1-AU level.
Table 1 gives the results of the multiparametric logistic regression analysis. High anti–protein Z IgG and IgM antibody levels were found to be highly associated with unexplained primary early fetal losses and early fetal death, and IgM antibodies are also associated with severe eclampsia and primary recurrent embryo losses in women with protein Z deficiency. A dose effect was evidenced between anti–protein Z antibody levels and the risk of pathologic pregnancy. The clinical risk depicted by anti–protein Z antibodies was independent from the risk associated with abnormal classical antiphospholipid antibodies.
In 10 patients, 5 with initial anti–protein Z antibody levels in the p75-p97 range of values and 5 in the p98-p100 range of values, antibodies were checked on 3 additional samples obtained over a 9-month period. Variations of antibody levels were low, always lower, for IgG and IgM, than 1 AU, and all the patients had results in the same range of risk-related values.
In conclusion, our data indicate that new autoantibodies related to hemostasis, namely anti–protein Z antibodies IgG and IgM, can help to define the clinical risk of pathologic pregnancies. A dose-effect relationship between antibody levels and the clinical risk is evidenced. Anti–protein Z antibodies are independent from classical antiphospholipid antibodies (lupus anticoagulant, anticardiolipin, and anti–β2-glycoprotein I antibodies).
This case-control study has one major limitation: It is an observational study; we cannot distinguish primary from secondary effects and can document associations only. One question, however, is how these antibodies act. Protein Z deficiency is basically not associated with recurrent embryo losses,3 but we show that, in case of protein Z deficiency found by accident in such patients, anti–protein Z IgM levels are good indicators of the clinical risk and are inversely related to protein Z concentrations. The underlying mechanism is thus unlikely to depend on the direct hemostatic consequences of protein Z inhibition by antibodies. It is rather induced by the consequences of enhanced immune-complex formation, like, for instance, complement or cellular activation, as already shown for anti–β2-glycoprotein I IgM antibodies.7,8 This hypothesis may be the prevalent one in unexplained recurrent embryo losses. In case of unexplained fetal death or severe pre-eclampsia, 2 pathologies that are linked with hypercoagulability in mothers, anti–protein Z antibodies may exert their pathogenic effect through protein Z inhibition. Future studies using a protein Z functional assay or studying the effect of anti–protein Z antibodies on white blood, endothelial, and trophoblastic cells are warranted.
Prepublished online as Blood First Edition Paper, March 6, 2003; DOI 10.1182/blood-2002-12-3802.
One of the authors (J.A.) has declared a financial interest in a company (Hyphen BioMed) whose product was studied in the present work.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank the numerous current and past colleagues—obstetricians and gynecologists—who agreed to participate in our Mediterranean Abnormal Pregnancy Study Program and, of course, all patients and controls who agreed to join us in this long-distance-running adventure. We thank Mrs Christine Thirion for logistic support. We thank Mrs Margaret Manson for editorial assistance.