Abstract
The effect of the organization and delivery of health care at medical centers, referred to as “center effects,” with clinical outcomes after hematopoietic stem cell transplantation (HSCT) is not clear. We examined the association between center and treatment provider factors and mortality after HSCT. We surveyed 163 (87% response rate) United States transplantation centers that performed HLA-identical sibling HSCT for leukemia or autologous HSCT for lymphoma between 1998 and 2000 among patients at least 18 years old. One hundred thirteen (69%) centers performed HLA-identical sibling transplantations, whereas 162 (99%) performed autologous transplantations. Factors associated with decreased 100-day mortality in the allogeneic setting include a higher patient-per-physician ratio (P = .003) and centers where physicians answer calls after office hours (P = .03). Medical school affiliation was not associated with increased 100-day mortality except in centers where students/residents are present without fellows (P = .02). Center effects were weaker in autologous HSCT at 1 year. Differences in 100-day mortality in patients receiving transplants in centers with favorable versus unfavorable factors were greater in allogeneic than autologous HSCT. Greater physician involvement in patient care is important in producing favorable outcomes after HSCT. To more clearly establish the role of the factors we identified, further studies are recommended.
Introduction
High-dose chemotherapy with or without radiotherapy followed by hematopoietic stem cell support is widely used to treat diverse malignant and nonmalignant diseases.1-4 Hematopoietic stem cell transplantation (HSCT) carries high risks of early morbidity and mortality. Treatment-related mortality (TRM) ranges from 3% to over 50%, a considerably higher risk than other complex medical procedures.4,5 For example, after autologous HSCT, 1-year TRM ranges from 5% to 15%, compared with 20% to 50% in allogeneic HSCT. Most treatment-related deaths occur in the first year after transplantation.
Although predictors of mortality are traditionally evaluated using clinical parameters related to the patient, the disease, or the treatment procedure, biologic paradigms do not completely explain outcome variations between patients and across treatment centers. Searching for factors related to the organization and delivery of health6-8 when outcomes vary among patients with similar disease biology and treatment provides opportunities to improve treatment results. These center-dependent factors are referred to as “center effects.”
Most studies on center effects in the medical and surgical literature, including HSCT, have focused on the association between procedure volume and survival.9-18 Too great a focus on procedure volume, without exploration of other health care factors involved in the delivery of care, may lead to erroneous conclusions about how to improve quality of care and patient outcomes. We therefore collected transplantation center and treatment provider characteristics in the United States and examined their association with survival outcome after HSCT for hematologic malignancies.
Patients and methods
Data source
Data on transplantation centers were obtained from the International Bone Marrow Transplant Registry (IBMTR) and the Autologous Blood and Marrow Transplant Registry (ABMTR) Center Characteristics Survey for the year 2001. The IBMTR is a voluntary working group of more than 400 transplantation centers worldwide and the ABMTR, of more than 250 transplantation centers in North and South America. Transplantation centers contribute data on consecutive transplantations with yearly follow-up to a Statistical Center at the Health Policy Institute of the Medical College of Wisconsin (Milwaukee, WI). Compliance is monitored with on-site audits and currently has an accuracy rate of 98%.
We surveyed the 187 IBMTR/ABMTR transplant centers that performed HLA-identical sibling HSCT for acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), or chronic myeloid leukemia (CML) or autologous HSCT for Hodgkin lymphoma (HL) or non-Hodgkin lymphoma (NHL) from 1998 to 2000. The survey was limited to centers in the United States because health care delivery systems differ markedly among countries. Center characteristics (Table 1) for eligible transplant centers were obtained using a survey questionnaire, which was completed by the center's medical director or designee. The Medical College of Wisconsin Institutional Review Committee approved the study protocol and survey questionnaire.
Patient selection for outcome studies
Centers completing the survey registered a total of 20 446 patients, older than 18 years and having transplants between 1998 and 2000 (14 817 [72%] autologous HSCT; 3579 [18%] HLA-identical sibling HSCT; 2050 [10%] alternative donor HSCT). From these, 2 study cohorts were created focusing on the most common indications for HSCT. The allogeneic transplant cohort consisted of 1426 patients with AML, ALL, or CML receiving HLA-identical sibling transplants. The autologous cohort consisted of 2859 patients with HL or NHL. All patients had at least 1 year of potential follow-up. An additional 3 eligible allograft recipients and 22 autograft recipients were lost to follow-up and excluded from the study.
Statistical analysis
Step 1: derivation of the patient clinical severity index. Prior to analysis of potential center effects, we derived a clinical severity index (CSI) for each patient to adjust for the preexisting risk of death based on demographic, disease, and treatment factors. The CSI is a numerically derived quantification of how `sick' a patient is at the time of transplantation and can theoretically range from 0.01 to 1.0, with 1.0 signifying the highest likelihood of death. Because of intrinsic differences in the risk of mortality between allogeneic and autologous transplants, the CSI was derived separately for the 2 procedures and at the 2 outcome time points (100 days and 1 year after transplantation) using forward stepwise multivariate logistic regression using patient-related (age, sex, race), disease-related (disease type, disease stage at the time of transplantation, disease duration, chemosensitivity for lymphoma), and transplant-related (type of graft, use of total body irradiation, graft-versus-host disease [GVHD] prophylaxis for allogeneic transplants, and year of transplant) factors as independent variables (risk factors). Note that the absolute values of the derived CSI for the autologous and allogeneic transplantation populations cannot be compared because they result from 2 separate models. Similarly, it should be noted that this score is calculated for the patients for this study and cannot be generalized for use as a calculatable prognostic scoring system for all patients. A normally distributed random “center effect” term was included in the model.19-21 Independent variables with a P less than or equal to .05 were included in the final model used to calculate each patient's CSI. Variables in the final model were age, disease type, disease stage, and sensitivity of the disease to chemotherapy (for the autologous cohort). Because the relationship between a unit increment in the derived CSI and survival probability was nonlinear, we created a binary variable for the CSI based on the population median. The median cutoff was chosen after division of the CSI values into quartiles did not show significant differences in survival between the first and second quartiles or between the third and fourth quartiles. This also allowed ease in interpreting the results when center characteristics were tested for interaction in step 2 of the analysis. Patients with a CSI greater than the median were considered high-risk patients, whereas patients with a CSI less than or equal to the median CSI were considered low-risk patients.
Step 2: determining center factors associated with mortality. The second part of the analysis fitted separate forward stepwise multivariate logistic regression models to the 1426 allograft recipients and 2859 autograft recipients, using death probability within 100 days or within 1 year as the outcome of interest and considering center characteristics as potential predictor variables. The derived CSI (high risk versus low risk) was forced in the model building.
The transplant center characteristics tested can be categorized into 3 groups (Table 1): (1) physician and health care provider characteristics, (2) transplant unit activities and resources, and (3) medical center characteristics. Because of the exploratory nature of our study and the lack of previous studies including most of the center characteristics we examined, we decided to retain factors with a P less than or equal to .05. Interactions between factors in the final model were tested. To further verify if the variation in outcome can be explained by other unmeasured treatment center factors aside from what we already examined and found significant in the final model, we tested for a “fixed center effect term.” Empirical estimates of the survival curves in the first 100 days were computed from the logistic model and plotted. Because all patients included had the potential of 1-year follow-up, survival at 100 days and at 1 year were compared between any 2 groups (eg, low-risk patients with favorable center factors versus low-risk patients with unfavorable center factors) using the χ2 test. All analyses were performed using SAS version 8.2 for Unix (SAS, Cary, NC).
Results
Center characteristics
Of 187 centers, 163 (87%) responded, 6 (3%) refused to participate, and 18 (10%) were excluded because they no longer participate in the IBMTR/ABMTR. Among those that completed the survey, 113 (69%) performed HLA-identical sibling HSCT and 162 (99%) performed autologous HSCT. However, only 88 of the 113 responding allograft centers and 142 of the 162 responding autograft centers had patients fulfilling the disease and age eligibility requirements for the study.
Tables 2 and 3 show the characteristics of the transplant centers in the United States that perform HLA-identical sibling HSCT for leukemia or autologous HSCT for lymphoma that were included in this study. The proportion of high- and low-risk patients (based on CSI) was not associated with procedure volume.
Center and treatment provider factors in the allogeneic cohort
Table 4 shows results of the multivariate analyses of survival in the allogeneic cohort. The median CSI was 0.21 (range, 0.07-0.54) and 0.22 (range, 0.06-0.56) at 100 days and 1 year, respectively. As expected, high-risk (CSI > 0.21) patients were 3 times more likely to die than low-risk (CSI ≤ 0.21) patients within 100 days after transplantation. Factors associated with lower mortality within 100 days after transplantation were (1) higher patient-per-physician ratio, that is, patients undergoing transplantation in centers where physicians cared for more than 20 patients per year were 33% less likely to die than those with lower physician case loads, and (2) patients undergoing transplantation in centers where physicians answered after office hours or emergency calls were 28% less likely to die than those in centers where calls were answered by nurses or physician assistants. Affiliation of the transplantation program with a medical school was not significantly associated with 100-day survival except for centers with programs that had rotating students and residents but not hematology-oncology or HSCT fellows on the transplant service; 100-day mortality of patients having transplantation in these centers was about twice higher than in centers without medical school affiliations. At 1 year, only the clinical severity of patients and ratio of patients per physician per year were significantly associated with mortality.
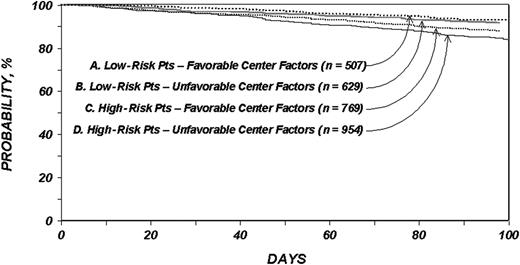
Figure 1 shows overall survival probabilities stratified by factors associated with 100-day mortality. Low-risk patients undergoing transplantation in centers with one or more favorable factors (> 20:1 patient-to-physician ratio, physicians answering after office or emergency calls, and no medical school affiliation or a medical school affiliation with rotating hematology-oncology or HSCT fellows) had a 100-day survival probability of 87% (95% confidence interval [CI], 84%-89%) versus 77% (95% CI, 69%-85%) among low-risk patients receiving transplants in centers with none of the favorable factors, a 10% difference in survival probability. High-risk patients undergoing transplantation in centers with one or more favorable factors had a 100-day survival probability of 68% (95% CI, 63%-72%) versus 53% (95% CI, 42%-64%) among high-risk patients receiving transplants in centers with none of the favorable factors, a 15% difference in survival probability. The distributions of primary causes of death in centers with favorable versus unfavorable center factors were not statistically different.
Overall survival probabilities with allogeneic transplantation. Probability of survival within 100 days among patients with leukemia who underwent HLA-identical sibling transplantation according to clinical severity and center factors.
Overall survival probabilities with allogeneic transplantation. Probability of survival within 100 days among patients with leukemia who underwent HLA-identical sibling transplantation according to clinical severity and center factors.
Center and treatment provider factors in the autologous cohort
Table 5 shows results of multivariate analyses for survival in the autologous cohort. The median CSI was 0.27 (range, 0.13-0.62) and 0.29 (range, 0.12-0.65) at 100 days and 1 year, respectively. High-risk patients (CSI > 0.27) had a 2-fold higher risk of dying compared to low risk patients (CSI ≤ 0.27) both at 100 days and 1 year after autologous transplantation for lymphoma. Two center factors were associated with a decreased risk of dying within 100 days after autologous transplantation. Patients receiving transplants in centers with higher patient-per-physician ratios, that is, where physicians cared for more than 12 patients per year, had a 26% lower risk of mortality than patients receiving transplants in centers with lower physician caseloads. Similar to the allograft setting, medical school affiliation was not significantly associated with 100-day survival outcome except for centers with programs that had rotating students and residents but no hematology-oncology or HSCT fellows; 100-day mortality of patients undergoing transplantation in these centers was 1.8 times higher than in centers without medical school affiliations. At 1 year, only the clinical severity of patients and ratio of patients per physician per year were significantly associated with probability of mortality.
Figure 2 shows overall survival according to factors associated with 100-day mortality in the autologous setting. Low-risk patients undergoing transplantation in centers with one or more of the favorable factors (> 12:1 patient-to-physician ratio and no medical school affiliation or with medical school affiliation with rotating hematology-oncology or HSCT fellows) had a 100-day survival probability of 93% (95%, CI 90%-95%) versus 92% (95% CI, 89%-94%) among those receiving transplants in centers with unfavorable factors. The 100-day probability of survival in high-risk patients receiving transplants in centers with one or more favorable factors was 88% (95% CI, 85%-90%) versus 84% (95% CI, 81%-86) among those treated in centers with unfavorable factors, a 4% difference in survival probability.
Overall survival probabilities with autologous transplantation. Probability of survival within 100 days among patients with lymphoma who underwent autologous transplantation according to clinical severity and center factors.
Overall survival probabilities with autologous transplantation. Probability of survival within 100 days among patients with lymphoma who underwent autologous transplantation according to clinical severity and center factors.
Further analysis showed that procedure volume was associated with the ratio of patients per physician (r = 0.42 for allogeneic HSCT and r = 0.48 for autologous HSCT), but not with center experience (age of program). However, neither procedure volume or center experience were significantly associated with survival even after the removal of ratio of patients per physician in the model. Additionally, adding a “fixed center effect term” in both the allograft and autograft models did not indicate that factors other than CSI and the identified center factors were associated with outcomes.
Discussion
Our data show that aside from the clinical severity of the patient's condition, center factors in the allogeneic and autologous HSCT setting are associated with better 100-day mortality. In the allogeneic HSCT setting, a strong association was demonstrated between physician caseload (more patients per physician) and better 100-day mortality. The type of medical school affiliation and presence of physicians answering after hours calls were weakly associated with 100-day mortality. In the autologous HSCT setting, a weak association was seen between caseload and medical school affiliation and 100-day mortality. In both HSCT settings, physician caseloads have a weak association with 1-year mortality. These findings suggest that the most important center characteristic affecting outcomes aside from clinical severity is the activity level and role of transplantation physicians and senior trainees.
Although other reports9-18 in the medical and surgical fields have shown a direct relationship between procedure volume and survival, it is unknown whether procedure volume directly affects outcome (eg, by increasing experience of personnel) or whether this is a surrogate for unmeasured factors that are associated with both improved outcomes and larger volumes. This distinction has policy implications. One can make a strong case for restricting certain complex procedures to large-volume centers if volume per se is the important parameter. However, if volume is a surrogate for other factors that are more common in large versus small centers, but which could be introduced to small centers, the appropriate course of action would be to institute these factors at all centers. Although our findings deserve further study, they suggest that there are center characteristics that may be adopted by small-volume centers that may improve survival outcomes.
Our study evaluated 2 types of HSCT that have different degrees of medical sophistication and risk. The lack of association between procedure volume (total number of transplants per year, allogeneic or autologous transplants) and center experience (number of years centers have been performing HSCT) with survival in our study contrasts with published reports in the general medicine setting. The reason for this is unclear, but one possible explanation could be the relatively small and homogenous subset of stem cell transplantation population we used to examine the relationship between procedure volume/experience and survival.
When we computed the ratio of annual procedures to physicians, representing an index of average caseload or physician experience or both, we found a decreased risk of 100-day and 1-year mortality associated with higher caseloads. This ratio may be identical for a large center with many physicians caring for many patients and a small center with fewer physicians and patients. It may also be similar for a center with one predominant full-time clinician among several clinicians attending “part-time” and a center of similar size where each attending is on service for 1 month. However, this ratio must also be distinguished from the average number of patients cared for by a physician at each center per unit time. Our present analysis focused on centers as the unit of analysis, whereas the latter measure is a complicated summary figure that requires carefully designed prospective data collection at the physician level. Such data should not only be able to evaluate physicians' case load and attentiveness to actual patient care, but also their seniority, specific experience with transplantation issues (as opposed to general oncology care), and their level of clinical versus research commitment. These data would also allow a more refined analysis of the contribution of physician-provided care to patient outcomes. There may exist a U-shaped curve in which neither too few nor too many patients are optimal.
We also found a protective effect of having physicians answer after hours or emergency calls from patients who underwent HLA-identical sibling transplantation, suggesting that posttreatment complications are better recognized and managed by more experienced personnel. Similarly, both nursing and attending physician staff caseload have been found to be associated with mortality after invasive procedures.18,22-26 Although our study was not able to detect a relationship between nurse-to-patient staffing and outcome, it should be noted that most of the centers in this study had 1:3 or higher nurse-to-patient ratios, limiting power to evaluate lesser staffing resources.
A higher 100-day mortality was seen in both allogeneic and autologous settings if transplant procedures were performed in centers affiliated with medical schools with rotating student or residents but not rotating hematology-oncology or HSCT fellows. This finding could be consistent with the belief that large-volume centers, more often affiliated with medical schools, attract sicker patients, and thus have inferior outcomes. However, we failed to detect any significant associations between patient disease severity and center factors, suggesting that the HSCT centers in this study had relatively equal chances of treating sicker patients, albeit among the limited number of diseases and transplant types included in the analysis. Our findings are contrary to those reported by Allison and colleagues in the setting of acute myocardial infarction, where hospitals with medical school affiliations demonstrated superior outcomes compared with minor teaching and nonteaching institutions.27 Possible reasons for the discrepancy include the type of procedure studied (management of a relatively common condition such as myocardial infarction versus a more complex and uncommon situation preceding and following HSCT), differences in the criteria for categorizing institutions, and consideration of the roles of different trainees (students, residents, and fellows). If our findings are confirmed in the HSCT setting, it is possible that (1) the involvement of inexperienced students and residents in patient management can result in deleterious effects if not accompanied by close supervision from more experience physicians; (2) the presence of students and residents decreases the time or focus devoted to actual patient care by the attending physicians as they also try to teach; (3) the absence of trainees may be associated with better outcomes because their responsibilities are assumed by physician extenders who provide more consistent care; or (4) a combination of all of these.
The pronounced modulation in survival outcomes in the allogeneic compared to the autologous setting is an indication that “center effect” is more evident as the complexity of the treatment increases. This mirrors findings in surgical oncology, where volume-outcome relationships are consistently seen in high-risk surgical procedures, but are otherwise conflicting in lower-risk surgery.10 Our study findings also suggest that the “center effects” we evaluated operate as an early rather than long-term influence after HSCT. This may be explained by the fact that centers are often only directly responsible for the early care of patients and thereafter care is transferred back to the referring oncologist.
On the other hand, our study failed to show an association between factors that would be expected to correlate with superior outcomes, such as Foundation for the Accreditation of Cellular Therapy (FACT) accreditation, National Cancer Institute (NCI) Cancer Center designation, or Center for Excellence designation. The current study should encourage further exploration of factors necessary for high-quality care including confirmation of those found in this study. Additional outcomes, such as patient satisfaction and measures of morbidity, should also be addressed.
Our study has several limitations. First, the final sample used in the multivariate analysis of the allograft cohort included only 88 of the 113 centers surveyed. The median procedure volume of the 113 centers was 50 transplants per year; the sample studied had larger annual volumes. However, 39% of the centers included in the study performed fewer than 60 transplants per year. We feel there was ample representation of the small-volume centers. Additionally, prior experience suggests centers not registering to the IBMTR are more likely to be nonacademic centers, implying that they are likely to be doing more autologous transplants, are not involved in research, and do not have trainees. Procedure volume and center experience are probably the same as those registering to the IBMTR/ABMTR. Second, the characteristics of the centers were retrospectively collected. Because the questionnaire was sent in 2001, respondents may not have had an accurate recollection of their center's clinical practice in 1998 to 2000. However, it seems unlikely that the center factors we examined drastically changed within the relatively short interval studied. Third, the outcomes we studied did not include disease recurrence or disease-free survival. We feel these outcomes are more likely determined by disease biology and are not primarily affected by center factors, but to the extent that they influence overall survival, they could be masking center effects. However, we compared the relapse rates across center by procedure volume and found no statistically significant differences. Fourth, our findings may not be applicable in pediatric HSCT where procedure volume is generally lower. Lastly, our derived CSI may not have adjusted completely for the patient, disease, and transplant-related factors known to affect transplant outcome. However, the factors included in the CSI are consistent with other studies reporting outcomes adjusted for patient characteristics.
Despite the exploratory nature of our study and the lack of complete understanding of the processes involved as to how the various center factors we identified contribute to better survival, it appears that the greater involvement of properly trained physicians is associated with better early outcomes, particularly in the allogeneic HSCT and autologous HSCT for high-risk patients, and should be encouraged. We recommend that further studies, preferably prospective in design, be done to establish a more definite role for the factors we identified as affecting mortality after HSCT.
Prepublished online as Blood First Edition Paper, December 14, 2004; DOI 10.1182/blood-2004-10-3863.
Supported by the Agency for Healthcare Research and Quality (AHRQ-5R03 HS 13046-02) and the Medical College of Wisconsin Research Affairs Committee (F.R.L.) and National Institutes of Health (NIH 5K23 [A82350-04]; J.D.R.). CIBMTR is supported by Public Health Service Grant U24-CA76518 from the National Cancer Institute, the National Institute of Allergy and Infectious Diseases, and the National Heart, Lung and Blood Institute; Agency for Healthcare Research and Quality; and grants from Aetna; AIG Medical Excess; Allianz Life/Life Trac; American Red Cross; American Society of Clinical Oncology; Amgen; anonymous donation to the Medical College of Wisconsin; AnorMED; Aventis Pharmaceuticals; Baxter Healthcare; Baxter Oncology; Berlex Laboratories; Biogen IDEC; Blue Cross and Blue Shield Association; The Lynde and Harry Bradley Foundation; BRT Laboratories; Cedarlane Laboratories; Celgene; Cell Pathways; Cell Therapeutics; CelMed Biosciences; Centocor; Cubist Pharmaceuticals; Dynal Biotech ASA; Edwards Lifesciences RMI; Endo Pharmaceuticals; Enzon Pharmaceuticals; ESP Pharma; Excess; Fujisawa Healthcare; Gambro BCT; Genzyme; GlaxoSmithKline; Human Genome Sciences; ICN Pharmaceuticals; ILEX Oncology; Kirin Brewery; Ligand Pharmaceuticals; Eli Lilly; Nada and Herbert P. Mahler Charities; Merck; Millennium Pharmaceuticals; Miller Pharmacal Group; Milliman USA; Miltenyi Biotec; The Irving I. Moskowitz Foundation; National Leukemia Research Association; National Marrow Donor Program; NeoRx; Novartis Pharmaceuticals; Novo Nordisk Pharmaceuticals; Ortho Biotech; Osiris Therapeutics; PacifiCare Health Systems; Pall Medical; Pfizer U.S. Pharmaceuticals; Pharmametrics; Pharmion; Protein Design Labs; QOL Medical; Roche Laboratories; Schering AG; StemCyte; StemCell Technologies; Stemco Biomedical; StemSoft Software; SuperGen; Sysmex; THERAKOS, a Johnson & Johnson company; University of Colorado Cord Blood Bank; Upside Endeavors; ViaCell; ViaCor Biotechnologies; WB Saunders Mosby Churchill; Wellpoint Health Network; and Zymogenetics.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
The authors extend their great appreciation and gratitude to all the medical directors and other medical staff, including the data managers from all the transplantation centers that participated in this study.