Abstract
We have recently demonstrated that the proteasome inhibitor, bortezomib, administered immediately following murine allogeneic bone marrow transplantation (BMT) resulted in marked inhibition of acute graft-versus-host disease (GVHD) with retention of graft-versus-tumor effects. We now assessed the effects of delayed bortezomib administration (5 or more days after BMT) on GVHD. Recipient C57BL/6 (H2b) mice were lethally irradiated and given transplants of bone marrow cells and splenocytes from major histocompatibility complex (MHC)–disparate BALB/c (H2d) donors. In marked contrast to the effects of bortezomib on GVHD prevention when administered immediately after BMT, delayed bortezomib administration resulted in significant acceleration of GVHD-dependent morbidity. No toxicity was observed following delayed bortezomib administration in models where donor T cells were not coadministered, indicating that these deleterious effects were critically dependent on GVHD induction. The increase in GVHD susceptibility even occurred when late administration of bortezomib was preceded by early administration. Pathologic assessment revealed that significant increases in gastrointestinal lesions occurred following delayed bortezomib administration during GVHD. This pathology correlated with significant increases of type 1 tumor necrosis factor α (TNF-α) receptor transcription in gastrointestinal cells and with significant increases of TNF-α, interleukin 1β (IL-1β), and IL-6 levels in the serum. These results indicate that the differential effects of proteasome inhibition with bortezomib on GVHD are critically dependent on the timing of bortezomib administration.
Introduction
The occurrence of acute graft-versus-host disease (GVHD) remains one of the most significant causes of morbidity following allogeneic bone marrow transplantation (BMT). GVHD is caused by administration of donor T cells into a genetically disparate recipient. The pathophysiology of GVHD is a complex process that can be conceptualized in 3 phases.1-3 In the first phase, the cytoreductive conditioning regimen causes immunosuppression of the recipients and damage to host tissues, including a self-limited burst of inflammatory cytokines. In the second phase, donor T cells recognize alloantigens on host antigen-presenting cells (APCs) and these activated T cells then proliferate and differentiate into effector cells. The second phase is critical for the amplification of the systemic inflammatory response, in which donor T cells also contribute to the inflammatory cytokine network. In the third phase, target tissues undergo apoptosis mediated by cellular effectors and inflammatory cytokines such as tumor necrosis factor α (TNF-α) and interferon γ, and further host tissue injury establishes a positive inflammatory feedback loop. Solid organs attacked during acute GVHD include the gut, liver, lungs, and skin.1-3
The proteasome is a multicatalytic proteinase complex responsible for the degradation of most intracellular proteins, including proteins crucial to cell-cycle regulation and apoptosis. Bortezomib (Velcade, formerly PS-341) is the first of its class of proteasome inhibitors to be tested in humans and has shown promising activity in several tumor types, especially in hematologic malignancies.4 It has recently been approved as a therapy in multiple myeloma.5 Bortezomib exerts numerous biologic effects that include blocking the activation of the transcription factor, nuclear factor-κB (NF-κB).6-8 NF-κB is implicated in the regulation of many genes that code for mediators of the immune and inflammatory responses.9,10
We have recently demonstrated that proteasome inhibition using bortezomib can markedly inhibit the generation of acute GVHD in mice after allogeneic BMT.11 Importantly, significant antitumor effects were maintained, suggesting that bortezomib may be of use to improve the efficacy of BMT. In that study, we found that bortezomib administered immediately after BMT reduced donor-derived T-cell expansion by inhibiting cell proliferation and selectively inducing apoptosis in the activated donor alloreactive T-cell population.11 Another mechanism by which bortezomib can limit GVHD may involve the blockade of NF-κB activity, resulting in a decrease in inflammatory cytokines produced by donor T cells and damaged host tissues after cytoreductive conditioning. However, NF-κB has both proapoptotic and antiapoptotic functions12 and can affect both proinflammatory and anti-inflammatory responses.13,14 It is possible that GVHD progression could also be enhanced by blocking NF-κB activity and thereby augmenting TNF-mediated cellular injury.15,16 Therefore, it was important to investigate the effects of bortezomib administration during ongoing GVHD.
We report here that the differential effects of proteasome inhibition with bortezomib on a murine GVHD model is determined by the timing of bortezomib administration. Delayed bortezomib administration resulted in marked acceleration and manifestation of GVHD-dependent pathology, particularly in the gut following BMT. This was correlated with increased transcription of type 1 TNF-α receptor (TNFR1) in the gut and the presence of proinflammatory cytokines in the serum. These results suggest that extreme caution must be used with delayed administration of bortezomib in combination with T cell–replete allogeneic BMT.
Materials and methods
Animals
Female BALB/c (H2d), C57BL/6 (B6, H2b), and immunodeficient C.B-17scid/scid (SCID, H2d) mice were purchased from the Animal Production Area of the National Cancer Institute (Frederick, MD). Animals were kept in specific pathogen-free conditions. All animal protocols were approved and in vivo studies were performed at each of the 2 animal facilities (National Cancer Institute, Frederick, MD, and University of Nevada, Reno). Mice were between 8 and 12 weeks of age at the start of the experiments.
Reagent
The proteasome inhibitor, bortezomib, was kindly provided by Millennium Pharmaceuticals (Cambridge, MA). Stock bortezomib solution (1 mg/mL) was prepared in Dulbecco phosphate-buffered saline solution (PBS) and stored at -70°C for up to 2 months prior to use. Bortezomib solutions were protected from light at all times. The stock solutions were thawed and diluted to 0.075 mg/mL in PBS immediately prior to use.
Cell preparation
Bone marrow cell (BMC) suspensions were prepared by gently releasing cells from the backbones, femurs, and tibiae into PBS with a mortar and pestle, filtering through a mesh filter to remove particulates, and washing the cell suspensions twice. Spleen-cell preparations were prepared by gently crushing the tissues to release the cells. Preparations were filtered to remove debris and washed twice in PBS for injection. Cell counts were performed on a Coulter Z1 cell counter (Coulter Electronics, Hialeah, FL).
In vivo studies
Induction of GVHD studies were performed at each of the 2 animal facilities (National Cancer Institute and University of Nevada) according to the guidelines of the Animal Care and Use Committees of each institution. C57BL/6 (B6, H2b) or CB-17 SCID (H2d) mice were used as recipients in the 2 GVHD model systems separately. B6 recipient mice received myeloablative doses (900-950 cGy) total body irradiation (TBI) from a 137Cs source. Irradiation was followed by the infusion of 1.0 or 1.5 × 107 BALB/c BMCs intravenously with or without BALB/c splenocytes (SCs; 5-20 × 106 cells intravenously) as a source of allogeneic T cells. CB-17 SCID recipient mice did not receive conditioning and were given transplants of B6 SCs (40 × 106 cells intravenously) as a source of allogeneic T cells. Recipient B6 or CB-17 SCID mice then received PBS or bortezomib in PBS at a dose of 15 μg (intravenously or intraperitoneally, with comparable results) at the indicated time phase after cell infusion. Mice were monitored and weighed weekly. All moribund mice were humanely killed. All experiments were performed at least 3 times with 5 to 10 mice per group.
Histology
Tissues (liver, small and large intestine) from the mice were placed in 10% formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin. Tissue sections were evaluated and graded in coded fashion by a veterinary pathologist (M.R.A.) as previously described.17 A semiquantitative scale from 0 to 4 was used where histopathologic changes were identified as minimal = 1, mild = 2, moderate = 3, and severe = 4. Cumulative histopathology scores were calculated based on the sum of individual changes of 3 to 4 parameters in each organ (villous blunting, crypt-cell hyperplasia, crypt-cell apoptosis, and inflammation in the small intestine; goblet-cell depletion, sloughing of epithelial cells into the lumen, and crypt-cell apoptosis in the colon; and vacuolation, necrosis, and oval-cell hyperplasia in the liver). Each experiment consisted of 3 to 6 mice per group and 4 independent experiments were assessed. Images were visualized using an Olympus Vanox AHBS3 microscope with an Olympus SPlan Apo × 20/0.70 NA objective (Olympus, Woodbury, NY). A Diagnostic Instrument Spot RT color digital camera using Spot software version 4.0.2 was used to acquire the images (Diagnostic Instruments, Sterling Heights, MI).
RNAse protection assay
Total small intestine and liver RNA was isolated using RNA STAT-60 (Tel-test, Friendswood, TX) according to the manufacturer's instructions. mRNA expression was then examined using the BD Riboquant RNAse protection assay (RPA) system (mouse APO-3 probe set, PharMingen, San Diego, CA) as directed. Briefly, 32P-UTP–labeled antisense RNA probes were synthesized and then hybridized with total small intestine and liver RNA overnight (16 hours). The following day, free RNA strands and excess probe were digested with RNAse A and T1, then precipitated, resuspended in 5 μL loading buffer, and run on a 4.75% polyacrylamide gel. The gel was then absorbed onto gel blot paper and dried on a Slab Gel Dryer at 80°C (Savant, Holbrook, NY), followed by overnight exposure and autoradiography using the Bio-Rad GS-525 Molecular Imager System (Bio-Rad, Hercules, CA). Band densities were determined using Molecular Analyst Software (Bio-Rad) and normalized against the L32 and glyceraldehyde phosphate dehydrogenase (GAPDH) probes for each sample. Samples for the RPA from 2 (liver) and 4 (gut) independent experiments consisted of 3 to 5 samples per group in each independent experiment.
Cytokine analysis
Serum was collected on day 13 after BMT. Serum cytokine levels were determined by multiplex analysis on a Luminex (Austin, TX), using mouse cytokine-specific bead sets and standards according to the manufacturer's instructions (R&D Systems, Minneapolis, MN). Serum samples from 3 independent experiments with 3 to 5 serum samples per group were analyzed in each independent experiment.
Statistics
Survival data were plotted by the Kaplan-Meier method and analyzed by the log-rank test. Serum and RPA data were analyzed by the unpaired Student t test with the Welch correction (2-tailed P value). Pathology scores were evaluated with the Mann-Whitney test. P less than .05 was considered significant.
Results
Delayed administration of bortezomib results in accelerated GVHD morbidity following allogeneic BMT
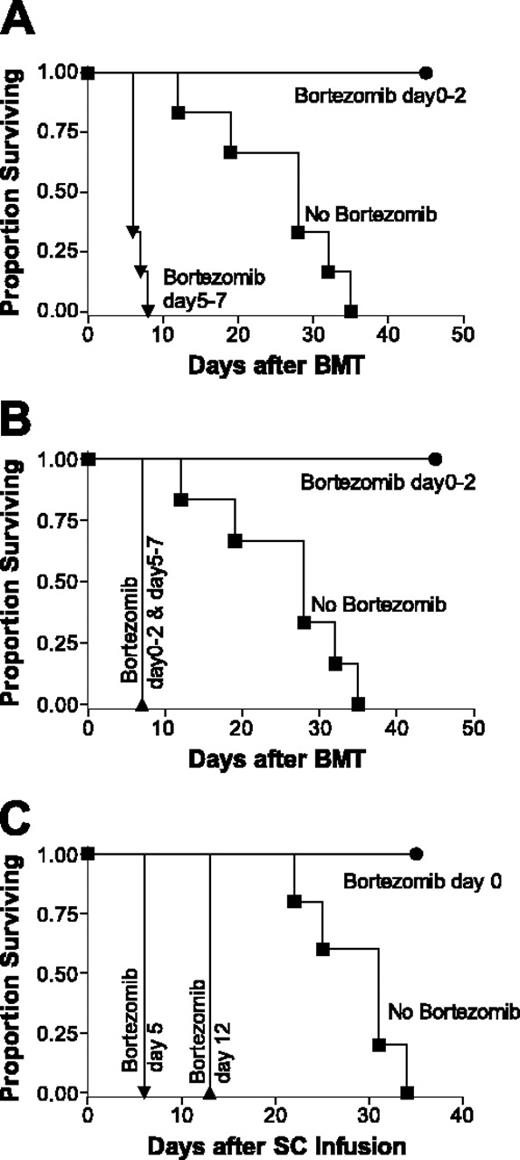
We have previously observed that bortezomib, administered immediately after allogeneic BMT, resulted in increased survival and protection from GVHD. Therefore, we next assessed the effects of bortezomib given at later time points when GVHD was ongoing. Using a fully major histocompatibility complex (MHC)–mismatched BMT model (BALB/c (H2d) BMCs and SCs lethally irradiated C57BL/6 (H2b) recipients), we found that early administration of bortezomib (day 0-2 after BMT) consistently resulted in significant protection from GVHD (Figure 1A-B). In marked contrast, when bortezomib was administered at later time points (day 5-7 after BMT), the mice rapidly died due to acute GVHD, sometimes showing overt acute GVHD within 12 hours of the first injection and all dying within several days from the first day of injection (Figure 1A). Use of donor purified T cells resulted in findings that were comparable with the use of whole SCs containing the equivalent dose of T cells (data not shown). When bortezomib was administered early to prevent GVHD and then continued with the delayed administration, significant increases in GVHD morbidity occurred (Figure 1B), suggesting the augmentation of GVHD associated with delayed bortezomib administration was dominant over the protection seen with early administration.
The paradoxic effect of proteasome inhibition with bortezomib on the alteration of GVHD development and mortality as determined by the timing of bortezomib administration. (A-B) B6 (H2b) recipients of BALB/c (H2d) 15 million bone marrow and 20 million spleen cells were treated with or without 15 μg bortezomib per dose daily. Bortezomib protected mice from GVHD mortality when administration was daily from day 0 through +2 after BMT. Significant increases in survival were observed in early bortezomib-treated mice (•) compared with GVHD control (vehicle control-treated) mice (▪; panels A and B, P < .001). Conversely, bortezomib accelerated GVHD mortality when administration was from day +5 through +7 (A) or bortezomib treatment from day 0 through +2 and day +5 through +7 (B) after BMT. Significant decreases in survival were observed in late bortezomib-treated mice (•) compared with GVHD control (vehicle control-treated) mice (▪; panel A, P < .001) and in continuous bortezomib-treated mice (▴) compared with GVHD control (vehicle control-treated) mice (▪; panel B, P < .001). Results from 1 of 3 independent experiments are presented for panels A and B. Each experiment consists of 5 to 10 mice per treatment group. (C) C.B-17 SCID (H2d) recipients of B6 (H2b) 40 million spleen cells were treated with or without 15 μg bortezomib per dose for 1 day. Significant decreases in survival were observed in day +5 bortezomib-treated mice (▾) compared with GVHD control (vehicle control-treated) mice (▪; panel A, P < .001) and in day +12 bortezomib-treated mice (▴) compared with GVHD control (vehicle control-treated) mice (▪; panel B, P < .001). Results from 1 of 3 independent experiments are presented. Each experiment consists of 4 to 10 mice per treatment group.
The paradoxic effect of proteasome inhibition with bortezomib on the alteration of GVHD development and mortality as determined by the timing of bortezomib administration. (A-B) B6 (H2b) recipients of BALB/c (H2d) 15 million bone marrow and 20 million spleen cells were treated with or without 15 μg bortezomib per dose daily. Bortezomib protected mice from GVHD mortality when administration was daily from day 0 through +2 after BMT. Significant increases in survival were observed in early bortezomib-treated mice (•) compared with GVHD control (vehicle control-treated) mice (▪; panels A and B, P < .001). Conversely, bortezomib accelerated GVHD mortality when administration was from day +5 through +7 (A) or bortezomib treatment from day 0 through +2 and day +5 through +7 (B) after BMT. Significant decreases in survival were observed in late bortezomib-treated mice (•) compared with GVHD control (vehicle control-treated) mice (▪; panel A, P < .001) and in continuous bortezomib-treated mice (▴) compared with GVHD control (vehicle control-treated) mice (▪; panel B, P < .001). Results from 1 of 3 independent experiments are presented for panels A and B. Each experiment consists of 5 to 10 mice per treatment group. (C) C.B-17 SCID (H2d) recipients of B6 (H2b) 40 million spleen cells were treated with or without 15 μg bortezomib per dose for 1 day. Significant decreases in survival were observed in day +5 bortezomib-treated mice (▾) compared with GVHD control (vehicle control-treated) mice (▪; panel A, P < .001) and in day +12 bortezomib-treated mice (▴) compared with GVHD control (vehicle control-treated) mice (▪; panel B, P < .001). Results from 1 of 3 independent experiments are presented. Each experiment consists of 4 to 10 mice per treatment group.
It has been shown that myeloablative conditioning with TBI for BMT can result in gastrointestinal damage and inflammatory cytokine production.18,19 This could be a contributing factor with the increased gut toxicity observed following delayed bortezomib administration. To exclude the influence of cytoreductive conditioning with TBI, we next performed GVHD studies using B6 (H2b) SCs injected into nonirradiated immunodeficient C.B-17 SCID (H2d) mice (Figure 1C). Administration of bortezomib on the day of cell transfer protected the mice from lethal GVHD, whereas mice that received delayed bortezomib rapidly died of GVHD. In addition, no morbidity was observed in mice that received bortezomib without adoptive transfer of allogeneic spleen cells (SCID recipients, data not shown; allogeneic BMT, Figure 2A-B). The results demonstrated that the increased GVHD-dependent morbidity following delayed bortezomib administration was not dependent on conditioning of the recipients by irradiation because the CB-17 SCID recipients also died earlier of GVHD.
Delayed administration of bortezomib results in the appearance of GVHD morbidity using doses of donor SCs alone that produce no overt pathology
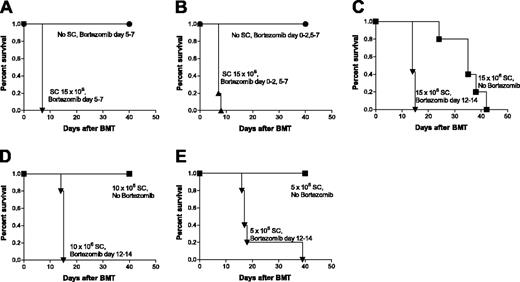
Because the delayed administration of bortezomib results in a rapid induction of GVHD mortality, we next performed dose responses of donor SCs to ascertain the extent of GVHD augmentation with delayed bortezomib administration. The results demonstrate that even at low doses of SCs, in which no outward signs of GVHD were apparent, the delayed administration of bortezomib resulted in significantly (P < .005) increased GVHD morbidity (Figure 2C-E). Thus, delayed bortezomib administration can result in the occurrence of acute GVHD in instances where no evidence of overt GVHD would normally be detected.
Pathologic evaluation indicated that recipients of SCs and delayed bortezomib treatment had marked increases in gut lesions in the small intestine and colon (Figures 3, 4) compared with control recipients. The small intestine had greater villous blunting and fusion, ulceration, and inflammation. Crypt-cell apoptosis and hyperplasia were also more extensive than in the recipients receiving SCs alone. Sections of colon tissue showed evidence of goblet-cell depletion with sloughed cells in crypt lumens, multifocal ulceration, and inflammation. As in the small intestine, cryptcell hyperplasia was more extensive than in the recipients receiving SCs alone. The presence of microscopic lesions was contingent on the administration of both SCs and delayed bortezomib administration because no pathologic damage or GVHD mortality was observed when bortezomib was administered at this or any other time point without SCs (Figures 2A-B and 3). In the liver, there were no significant differences in the lesions present between the 2 treatment groups that received SCs (Figure 3). This suggests that the gut is the primary target for this increased mortality after delayed bortezomib administration following allogeneic BMT. Thus, the data indicate that the timing of bortezomib administration has a pivotal role in protection or increased GVHD pathology after BMT and the gut is the primary GVHD target organ affected.
Dose response of donor spleen cells necessary for the increased GVHD mortality by delayed bortezomib administration. (A-B) B6 (H2b) recipients of BALB/c (H2d)15 million BMCs with or without 15 million spleen cells (SCs) were treated with 15 μg bortezomib per dose daily. Morbidity was not observed in mice that received BMCs but not SCs and delayed bortezomib administration from day +5 through +7 (A) or bortezomib treatment from day 0 through +2 and from day +5 through +7 (B) after BMT. Significant decreases in survival were observed in delayed bortezomib-treated mice with SCs (▾, ▴) compared with delayed bortezomib-treated mice without SCs mice (•; panels A and B, P < .001). (C-E) B6 (H2b) recipients of BALB/c (H2d) 15 million bone marrow with 15 million (C), 10 million (D), or 5 million (E) SCs were treated with or without 15 μg bortezomib per dose daily. Delayed bortezomib treatment from day +12 through +14 after BMT accelerates GVHD mortality. Significant decreases in survival were observed in bortezomib-treated mice (▾) compared with mice that received PBS (no bortezomib-treatment; ▪; P < .005). Results from 1 of 3 independent experiments are presented. Each experiment consisted of 5 to 10 mice per treatment group.
Dose response of donor spleen cells necessary for the increased GVHD mortality by delayed bortezomib administration. (A-B) B6 (H2b) recipients of BALB/c (H2d)15 million BMCs with or without 15 million spleen cells (SCs) were treated with 15 μg bortezomib per dose daily. Morbidity was not observed in mice that received BMCs but not SCs and delayed bortezomib administration from day +5 through +7 (A) or bortezomib treatment from day 0 through +2 and from day +5 through +7 (B) after BMT. Significant decreases in survival were observed in delayed bortezomib-treated mice with SCs (▾, ▴) compared with delayed bortezomib-treated mice without SCs mice (•; panels A and B, P < .001). (C-E) B6 (H2b) recipients of BALB/c (H2d) 15 million bone marrow with 15 million (C), 10 million (D), or 5 million (E) SCs were treated with or without 15 μg bortezomib per dose daily. Delayed bortezomib treatment from day +12 through +14 after BMT accelerates GVHD mortality. Significant decreases in survival were observed in bortezomib-treated mice (▾) compared with mice that received PBS (no bortezomib-treatment; ▪; P < .005). Results from 1 of 3 independent experiments are presented. Each experiment consisted of 5 to 10 mice per treatment group.
Effects of delayed bortezomib administration on pathologic damage to target organs of GVHD. Significant increases in histologic changes were observed in the small and large intestine, but not the liver, of mice treated with delayed bortezomib on day +12 or day +12 through day +13 at 15 μg/mouse (intravenously). Mice were assessed 16 hours after a single bortezomib or vehicle control (VC) injection (day +12) or 6 hours after a second injection (day +13) of bortezomib or VC, respectively. Three 3 to 6 mice per experimental group in each of 4 independent experiments were assessed. Tissues were assessed for histologic changes in 3 to 4 parameters as described in “Materials and methods” and the sum of these scores are represented for each tissue. Histologic changes for each parameter were assessed and graded where 0 = normal, 1 = minimal, 2 = mild, 3 = moderate, and 4 = severe. Comparison of cumulative histopathologic scores between the groups with splenocytes were analyzed by Mann-Whitney test (P < .05).
Effects of delayed bortezomib administration on pathologic damage to target organs of GVHD. Significant increases in histologic changes were observed in the small and large intestine, but not the liver, of mice treated with delayed bortezomib on day +12 or day +12 through day +13 at 15 μg/mouse (intravenously). Mice were assessed 16 hours after a single bortezomib or vehicle control (VC) injection (day +12) or 6 hours after a second injection (day +13) of bortezomib or VC, respectively. Three 3 to 6 mice per experimental group in each of 4 independent experiments were assessed. Tissues were assessed for histologic changes in 3 to 4 parameters as described in “Materials and methods” and the sum of these scores are represented for each tissue. Histologic changes for each parameter were assessed and graded where 0 = normal, 1 = minimal, 2 = mild, 3 = moderate, and 4 = severe. Comparison of cumulative histopathologic scores between the groups with splenocytes were analyzed by Mann-Whitney test (P < .05).
Delayed administration of bortezomib is associated with increased TNFR1 mRNA levels in the gut of GVHD mice
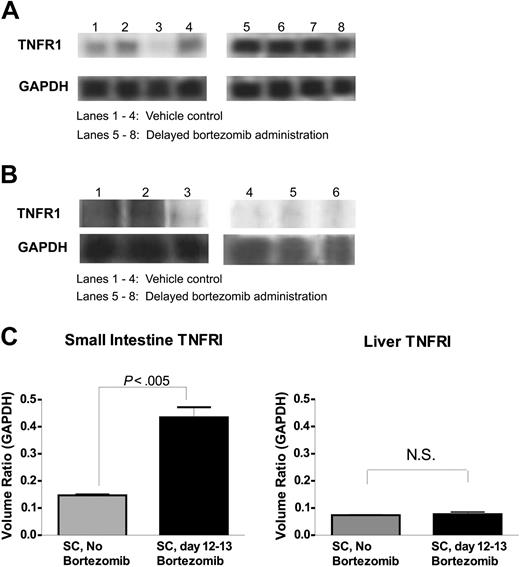
Because histopathologic examination had revealed that the gut was particularly affected with delayed bortezomib administration, we analyzed the tissue for expression of genes associated with killing pathways. At 6 hours after delayed (day 12-13 after BMT) bortezomib administration, the small intestine and liver were assessed by RPA for mRNA levels of Fas, FasL, and TNFR1 (p55). The results demonstrate that significant increases in TNFR1, the principal proinflammatory and proapoptotic receptor for TNF-α,20 were observed in the small intestine, but not the liver, of mice receiving SCs and delayed bortezomib treatment compared to recipients receiving either delayed bortezomib treatment (data not shown) or SCs alone (Figure 5A-C). No significant differences in Fas or FasL were observed (data not shown) suggesting that the TNF pathway may be particularly targeted in the gut of recipients receiving SCs after delayed bortezomib administration. Thus, up-regulating TNFR1 expression in small intestine may be, in part, the mechanism of increased susceptibility of GVHD mice to immune-mediated target damage and lethality after delayed bortezomib administration.
Increases in serum cytokine levels in GVHD mice after delayed bortezomib administration
We next assessed the levels of proinflammatory cytokines at 6 hours after delayed (day 12-13 after BMT) bortezomib administration. Serum levels of the proinflammatory cytokines TNF-α, interleukin 1β (IL-1β), and IL-6 were all significantly (P < .05) increased in the recipients receiving SCs and delayed bortezomib treatment compared with recipients receiving SCs and vehicle control (Figure 6). Delayed administration of bortezomib resulted in cytokine increases only in the recipients with SCs but not in the recipients without SCs (Figure 6). These results indicate that despite the ability of proteasome inhibition by bortezomib to block NF-κB, increased proinflammatory cytokines were detected after delayed administration of bortezomib during ongoing GVHD. Consistent with the histopathology data, increases in TNF-α, IL-1β, and IL-6 levels as well as gut tissue TNFR1 levels after delayed bortezomib administration were critically dependent on GVHD induction.
Delayed administration of bortezomib significantly increases gut histopathologic damage in mice with GVHD. B6 (H2b) recipients of BALB/c (H2d) 10 million bone marrow and 15 million spleen cells were treated with or without 15 μg bortezomib per dose daily on day 12 after BMT. The next day (day 13) mice were humanely killed and gut tissue was collected, processed, and stained with hematoxylin and eosin. (A-C) Small intestine from mice without delayed bortezomib treatment (A) have hyperplastic crypts (c) but normal villi (v). In contrast, the small intestines from mice with delayed bortezomib treatment (B-C) have villous blunting and fusion (v), hyperplastic crypts (c), and an inflammatory infiltrate (arrow). Other areas of small intestine are ulcerated (u) with a base of inflammatory cells. (D-F) Colons from mice without delayed bortezomib treatment (D) display relatively normal morphology. In contrast, colons from mice with delayed bortezomib treatment have increased (E) goblet cell depletion and inflammatory cells (arrow) in the lamina propria. Other areas of colon (F) are ulcerated (u), have sloughed cells in crypt lumens (c), and inflammation in the ulcer base and the submucosa (Sm). Original magnification × 200.
Delayed administration of bortezomib significantly increases gut histopathologic damage in mice with GVHD. B6 (H2b) recipients of BALB/c (H2d) 10 million bone marrow and 15 million spleen cells were treated with or without 15 μg bortezomib per dose daily on day 12 after BMT. The next day (day 13) mice were humanely killed and gut tissue was collected, processed, and stained with hematoxylin and eosin. (A-C) Small intestine from mice without delayed bortezomib treatment (A) have hyperplastic crypts (c) but normal villi (v). In contrast, the small intestines from mice with delayed bortezomib treatment (B-C) have villous blunting and fusion (v), hyperplastic crypts (c), and an inflammatory infiltrate (arrow). Other areas of small intestine are ulcerated (u) with a base of inflammatory cells. (D-F) Colons from mice without delayed bortezomib treatment (D) display relatively normal morphology. In contrast, colons from mice with delayed bortezomib treatment have increased (E) goblet cell depletion and inflammatory cells (arrow) in the lamina propria. Other areas of colon (F) are ulcerated (u), have sloughed cells in crypt lumens (c), and inflammation in the ulcer base and the submucosa (Sm). Original magnification × 200.
Discussion
The results presented here definitively demonstrate that proteasome inhibition with bortezomib can prevent acute GVHD when administered immediately after BMT but can also promote GVHD-dependent toxicity when administration is delayed. This increase in GVHD mortality was striking in that it could be observed even when lower amounts of donor T cells were administered such that no outward manifestations of GVHD (ie, diarrhea, posture, or grooming changes) were observed in the recipients not receiving delayed bortezomib and when GVHD was prevented by early administration of bortezomib. These results are consistent with our previous study that GVHD prevention by early administration of bortezomib is due to a lowering or dampening of the donor T-cell responses.11 Reduced T-cell responses could still be augmented by the later administration of bortezomib. This may also explain how graft-versus-tumor responses were still maintained following early bortezomib treatment.11
Up-regulation of TNFR1 mRNA expression in gut tissue following delayed bortezomib administration. B6 (H2b) recipients of BALB/c (H2d) 15 million bone marrow and 20 million spleen cells were treated with or without 15 μg bortezomib per dose daily from day +12 through +13. Small intestine and liver were collected at 6 hours after bortezomib administration (day +13) for RNA extraction. TNFR1 mRNA expression was analyzed by RPA. (A-B) Autoradiograph of TNFR1 and GAPDH bands from gut (A) and liver (B) RPA gels. (C) Quantitative levels of TNFR1 steady-state mRNA levels in the gut and liver tissue from bortezomib and vehicle control-treated animals. Quantitative levels of TNFR1 mRNA were determined by densitometric analysis and are expressed as a ratio of the band volumes of TNFR1 normalized to the GAPDH housekeeping gene. These data are representative of 2 (liver) and 4 (gut) independent experiments. NS indicates not statistically significant.
Up-regulation of TNFR1 mRNA expression in gut tissue following delayed bortezomib administration. B6 (H2b) recipients of BALB/c (H2d) 15 million bone marrow and 20 million spleen cells were treated with or without 15 μg bortezomib per dose daily from day +12 through +13. Small intestine and liver were collected at 6 hours after bortezomib administration (day +13) for RNA extraction. TNFR1 mRNA expression was analyzed by RPA. (A-B) Autoradiograph of TNFR1 and GAPDH bands from gut (A) and liver (B) RPA gels. (C) Quantitative levels of TNFR1 steady-state mRNA levels in the gut and liver tissue from bortezomib and vehicle control-treated animals. Quantitative levels of TNFR1 mRNA were determined by densitometric analysis and are expressed as a ratio of the band volumes of TNFR1 normalized to the GAPDH housekeeping gene. These data are representative of 2 (liver) and 4 (gut) independent experiments. NS indicates not statistically significant.
Delayed bortezomib administration significantly increases serum cytokine levels in mice with GVHD. B6 (H2b) recipients of BALB/c (H2d) 15 million BMCs with or without 20 million spleen cells (SCs) were treated with or without 15 μg bortezomib per dose daily, day 12 to 13 after BMT. Serum was collected at 6 hours after bortezomib administration (day +13). Significant increases in TNF-α, IL-1β, and IL-6 were observed in delayed bortezomib-treated recipients with SCs compared with vehicle control-treated recipients with SCs (P < .05). Representative data from 1 of 3 independent experiments are presented, 5 serum samples per group.
Delayed bortezomib administration significantly increases serum cytokine levels in mice with GVHD. B6 (H2b) recipients of BALB/c (H2d) 15 million BMCs with or without 20 million spleen cells (SCs) were treated with or without 15 μg bortezomib per dose daily, day 12 to 13 after BMT. Serum was collected at 6 hours after bortezomib administration (day +13). Significant increases in TNF-α, IL-1β, and IL-6 were observed in delayed bortezomib-treated recipients with SCs compared with vehicle control-treated recipients with SCs (P < .05). Representative data from 1 of 3 independent experiments are presented, 5 serum samples per group.
Bortezomib exerts numerous biologic effects that include blocking the activation of the transcription factor NF-κB.6-8 NF-κB is implicated in the regulation of many genes that code for mediators of the immune and inflammatory responses.9,10 GVHD progression is accompanied by inflammatory cytokine production and inflammatory cytokine-mediated target tissue damage.1-3 Therefore, not only immune cells (donor T cells, recipient dendritic cells) but also nonimmune cells (recipient target tissues) should be affected by delayed bortezomib administration. NF-κB plays a protective role in cell death induced by TNF-α.16 Therefore, blocking NF-κB activity in GVHD target tissues may enhance GVHD damage by enhancing TNF-mediated cellular injury. Our data indicate that the gut is the primary organ made more susceptible to GVHD attack after delayed bortezomib administration. Other studies demonstrate a strong association with NF-κB and protection from intestinal epithelial apoptosis19,21,22 and suppression of antiapoptotic molecules of the inhibitor of apoptosis protein (IAP) family up-regulation by the proteasome inhibitor MG132 with the sensitization of normal enterocytes to Fas-induced apoptosis.23 Additionally, it has been demonstrated that NF-κB can protect enterocytes from TNF-mediated death.24 Consistent with this observation, it has been demonstrated that NF-κB inhibition could prevent inflammation and yet result in increased cell injury in an intestinal ischemia model.25 NF-κB blockade may play a similar role in GVHD by suppressing the initial phase of inflammation and the later phase by increasing the susceptibility of GVHD target organs to immune-mediated apoptosis. TNFR1 is the principal proinflammatory and proapoptotic receptor for TNF-α,20,25 and the data presented here demonstrating increased mRNA levels of TNFR1 would support this pathway as being a potential component of the mechanisms by which bortezomib accelerated GVHD-induced gut pathology. It has been demonstrated that TNFR1-induced apoptosis involves 2 sequential signaling complexes. Failure to activate NF-κB (the initial step in the signaling process) results in cell death.26 Therefore, delayed bortezomib administration increased susceptibility to GVHD target organ (gut) damage and lethality may involve multiple steps. Donor T cells, as well as cytokines released by donor T cells, are necessary to trigger immune-mediated damage. Delayed bortezomib administration may act, in part, to enhance this damage by up-regulating expression of TNFR1, although bortezomib may induce apoptosis by multiple pathways, some of which may be independent of the NF-κB pathway.27 No protection from bortezomib-mediated accelerated morbidity was observed when mice deficient in TNFR1 were used (data not shown). This would suggest that TNFRII or other mediators may also contribute to the toxicity associated with delayed bortezomib administration.
A previous study has shown that NF-κB activation in leukocytes recruited during the onset of inflammation is associated with proinflammatory gene expression, whereas activation during the resolution of inflammation is associated with the expression of anti-inflammatory genes and the induction of apoptosis.13 Inhibition of NF-κB during the resolution of inflammation protracts the inflammatory response and prevents apoptosis. This suggests that NF-κB has an anti-inflammatory role in vivo involving the regulation of inflammatory resolution.13 A similar phenomenon may be occurring with donor T cells during GVHD progression in the GVHD models presented here.
Even though previous studies demonstrated that NF-κB activation provides radioprotection to the intestinal epithelium,19 we have shown that delayed bortezomib administration-dependent gut damage in our model is not dependent on radiation damage. The results showing that recipients of grafts with minimal (bone marrow source only) T cells and receiving bortezomib at any time point lack pathologic damage or morbidity indicate that bortezomib-related lethality in our model is dependent on GVHD-related immune-mediated attack. The demonstration that adoptive allogeneic lymphocyte transfer into immunodeficient SCID mice (without conditioning such as TBI) results in increased GVHD susceptibility after delayed bortezomib administration confirms that bortezomib-related lethality is due solely to GVHD processes.
Because GVHD is a complex and multistage disease process, it is perhaps not surprising that bortezomib exhibits differential effects contingent on the timing of administration. The induction phase of GVHD appears to be the most susceptible phase for successful intervention. It has been demonstrated that administration of cytokines such as IL-2,28 IL-12,29 administration of cellular therapy such as donor-type natural killer (NK) cells,17 and the presence of cytokines such as interferon-γ,30 are all protective only if administered or present early after BMT. If administration or presence of any of the cytokines or immune cells is delayed, accelerated GVHD has been reported.17,31 Given that proteasome inhibitors have been demonstrated to sensitize neoplastic cells to chemotherapeutics6 and immune lytic molecules such as TNF-related apoptosis-inducing ligand (TRAIL)32 or TNF-α,33 it is also perhaps not surprising that normal cells and tissues may also be sensitized to subsequent immune attack. It remains to be determined if augmented antitumor responses can also be observed during this stage and if there is a means to prevent the GVHD-dependent toxicity without loss of graft-versus-tumor effect.
The results presented here demonstrate the 2 facets of proteasome inhibition by bortezomib in GVHD, as determined by the timing of bortezomib administration. Our data indicate that bortezomib could be potentially used at any time following T cell–depleted allogeneic BMT. However, extreme caution must be used with delayed administration of bortezomib in combination with T cell–replete allogeneic BMT because exacerbation of GVHD-dependent pathology, particularly in the gut, may occur.
Prepublished online as Blood First Edition Paper, June 16, 2005; DOI 10.1182/blood-2004-11-4526.
Supported in part by grants R01 CA102282, R01 AI34495, R01 HL66308 and with Federal funds from the National Cancer Institute, National Institutes of Health, under contract no. N01-CO-12400. By acceptance of this article, the publisher or recipient acknowledges right of the US Government to retain a nonexclusive, royalty-free license in and to any copyright covering the article.
K.S. designed and performed in vivo studies, analyzed the data, and was the principal author; D.E.C.W. performed in vitro studies and analyzed data; M.R.A. provided histopathology services; T.J.S. designed research and assisted in the writing of the manuscript; A.P.-M. performed cytokine analysis; B.R.B. designed research and assisted in the writing of the manuscript; L.A.W. designed and performed in vivo studies, assisted in data analysis, and helped write the manuscript; and W.J.M. designed research and assisted in the writing of the manuscript.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr Ruth Gault, Qing Zhou, and William H. D. Hallett for assisting in the preparation of the manuscript and helpful discussions, and Millennium Pharmaceuticals for providing bortezomib. We thank Weihong Ma and Myra Godfrey for their technical assistance with animal studies, Melinda Berthold for her technical assistance with cytokine analysis, and Colin Beck for secretarial assistance.
The content of this publication does not necessarily reflect the views or policies of the Department of Health and Health Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the United States Government.