Comment on Dame et al, page 4282
Dame and colleagues have provided insight into the physiologic consequences of Epo gene regulation by their demonstration that transcription depends in part on the Wilms tumor suppressor protein, Wt1.
The biologic repertoire of erythropoietin (Epo) extends way beyond hormonal stimulation of red blood cell production. In addition to its well-understood endocrine role in erythropoiesis, Epo may also act in a para-crine and perhaps autocrine manner to protect the central nervous system (CNS) and myocardium against ischemic damage, preserve endothelial and smooth muscle integrity, and maintain reproductive function.FIG1
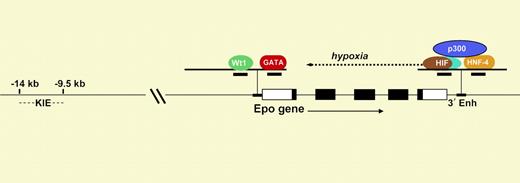
Diagram of the Epo gene. The 5 exons are shown as rectangles. The Epo promoter contains response elements that bind to Wt1 and GATA, and the 3′ enhancer contains elements that bind to HIF and HNF-4.
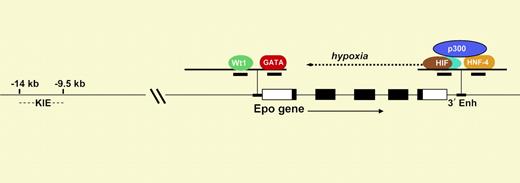
Diagram of the Epo gene. The 5 exons are shown as rectangles. The Epo promoter contains response elements that bind to Wt1 and GATA, and the 3′ enhancer contains elements that bind to HIF and HNF-4.
Like many genes, Epo regulation must be considered in a 3-dimensional framework involving time, place, and environmental stimulus. The best-understood level of regulation is the dramatic enhancement of Epo transcription at low oxygen tension, mediated through the hypoxia-inducible transcription factor HIF.1 As shown in the figure, this αβ heterodimer binds to a cognate hypoxia response element in a crucial enhancer located just 3′ to Epo's polyadenylation site (3′ Enh). HIF is expressed in nearly all cells and organs and serves as the master orchestrator of oxygen-dependent expression of a number of physiologically important genes. At normal oxygen tension, the α-subunit of HIF is posttranslationally modified by proline hydroxylation so that it is rapidly degraded.2 Only in hypoxic cells can HIF-α survive, allowing nuclear translocation, αβ dimer assembly, and induction (or repression) of gene expression.
Mechanisms underlying developmental and tissue-specific regulation of Epo expression are less clear.3 In humans and other mammals, during fetal development, the liver is the primary site of Epo production, with a switch to the kidney at birth. A previous study by Dame et al4 showed that the binding of the transcription factor GATA-4 to a cognate site on the Epo promoter contributes to expression in the fetal liver. In the paper published in this issue, Dame and colleagues demonstrate that the interaction of an isoform of Wt1 (Wt1(–KTS)) at a downstream site on the Epo promoter (see figure) is associated with transcriptional activation. They found that in mice deficient in Wt1, the expression of Epo mRNA in the fetal liver was about 60% of that in wild-type mice.
The determinants of tissue-specific expression of Epo appear to be complex.3 The robust hypoxic induction of Epo in kidney and liver is due in part to cooperation in the 3′ enhancer between HIF and HNF-4, a nuclear receptor that is preferentially expressed in these organs. Both HIF and HNF-4 bind to the transcriptional adapter p300. In addition, as shown in the figure, there is a kidney-inducible element 9 to 14 kb upstream of the promoter. The paper of Dame et al suggests that lower-level production of Epo in the brain and testes is due to coexpression of Wt1 at these sites. These results help set the stage for understanding the biologic significance of low-level Epo production at sites beyond the kidney and liver. ▪