Abstract
CD4+CD25+ regulatory T lymphocytes (Tregs) are specialized T cells playing a key role in the control of immune homeostasis. Here, we show that human Tregs constitutively express tyrosine hydroxylase (TH, EC 1.14.16.2), the rate-limiting enzyme in the synthesis of catecholamines, and contain substantial amounts of dopamine, norepinephrine, and epinephrine, which are released upon treatment with reserpine. Catecholamine release results in reduced production of interleukin-10 and transforming growth factor-β by Tregs, and in down-regulation of Treg-dependent inhibition of effector T-lymphocyte (Teff) proliferation, which occurs without affecting the production of tumor necrosis factor-α or interferon-γ. Tregs and Teffs express on the cell membrane both D1-like and D2-like dopaminergic receptors to a similar extent (12%-29% of the cells). Catecholamine-dependent down-regulation of Tregs is, however, selectively reversed by pharmacological blockade of dopaminergic D1-like receptors, which in Tregs only (and not in Teffs) are also expressed at the level of mRNA and are functionally coupled to intracellular production of cAMP. These findings indicate that in human Tregs endogenous catecholamines subserve an autocrine/paracrine loop involving dopaminergic pathways and resulting in down-regulation of Treg function.
Introduction
CD4+CD25+ regulatory T lymphocytes (Tregs) are specialized T cells that play crucial roles in the control of immune homeostasis, thus contributing to the maintenance of immunologic self-tolerance and being critically involved in immunologic diseases, tumor immunity, and transplantation tolerance.1 Tregs represent approximately 5% to 10% of circulating T cells in both mice and humans, and are also termed “naturally occurring,” in contrast to other types of T-regulatory cells such as Tr1 and Th3, which likely develop from conventional CD4+CD25− T lymphocytes (Teffs) in the periphery, and are thought to represent altered T-cell differentiation states rather than a unique T-cell lineage.2-4 Tregs are usually considered “naturally anergic,” although some recent studies may challenge this view5 ; nonetheless, they express several surface markers of activation, including CD25 itself (the interleukin-2 [IL-2] high-affinity receptor α chain), CTLA-4 (CD152), and glucocorticoid-induced tumor necrosis factor (TNF) receptor (GITR), among others (reviewed in Piccirillo and Thornton4 ). Such molecules are not expressed (or, eventually, are expressed only to a minor extent) in peripheral, naive Teffs, but they are induced after activation, thus rendering conventional activated CD4+ T cells virtually undistinguishable from Tregs on the simple basis of the pattern of surface molecules.
Until a few years ago, the catecholamines dopamine (DA), norepinephrine (NE), and epinephrine (E) were considered mainly as conventional neurotransmitters and neuroendocrine mediators; however, there is now robust evidence that they are also produced and used by cells of the immune system.6-14 Over the last decade, data have been accumulating about the functional relevance of the endogenous production of catecholamines in immune system cells.7,15 We have recently shown that in human peripheral blood mononuclear cells stimulated with the T-cell preferential polyclonal mitogen phytohemagglutinin (PHA) the synthesis of catecholamines is effectively induced through a protein kinase C–dependent mechanism that triggers the de novo expression of the mRNA for tyrosine hydroxylase (TH, EC 1.14.16.2, the first and rate-limiting enzyme in the synthesis of catecholamines) and its subsequent activation in T and B lymphocytes, resulting in intracellular accumulation of substantial amounts of such mediators.16,17 In these conditions, pharmacological inhibition of TH results in decreased activation-induced apoptosis,18 supporting a role for endogenous catecholamines in the functional modulation of lymphocyte activation. Intracellular catecholamines accumulating in activated lymphocytes are nonetheless also likely to provide these cells with a supply of mediators, which in turn may act upon lymphocytes themselves and/or upon neighboring cells.19
Lymphocytes are indeed a privileged target for catecholamines, inasmuch as they express both adrenoceptors (ARs)20,21 and dopaminergic receptors.22-26 NE released from sympathetic nerve terminals in lymphoid organs is widely accepted as a key mediator in the interplay between the brain and the immune system.21 In particular, functional β2-ARs expressed on immune cells regulate CD4+ T-cell–mediated immunity as well as antibody production by B cells (recently reviewed in Kin and Sanders27 ). DA, on the other hand, has been only recently identified as a direct modulator of the immune response.28 Nonetheless, over the last few years, evidence has rapidly increased regarding the ability of DA to affect the function of lymphocytes,22,24-26,29,30 including murine Tregs.31
Evidence thus supports the existence in lymphocytes of an endogenous catecholaminergic system. Interestingly, following cellular activation the various components of the system seem to undergo up-regulation, including de novo expression of functional α1-ARs,32 increased expression of β-ARs,33 increased response to DA (through D3 receptors),24 and increased TH expression and accumulation of catecholamines.16
Up-regulation of the endogenous catecholaminergic system in activated lymphocytes, together with the notion that Tregs express the general phenotype of activated cells, led us to hypothesize that Tregs could possess an array of functional catecholamine receptors, constitutively express TH, and consequently contain amounts of catecholamines significantly higher than naive Teffs. The present study was therefore undertaken to test such hypotheses and to investigate the possible role of endogenous catecholamines in the functional modulation of human Tregs.
Materials and methods
Substances
The following substances were purchased from Sigma (St Louis, MO): dopamine (DA) hydrochloride, norepinephrine (NE) bitartrate, epinephrine (E) hydrochloride, l-dihydroxyphenylalanine (l-DOPA), 3-methoxytyramine (3-MT) hydrochloride, dl-3,4-dihydroxyphenylacetic acid (DOPAC), homovanillic acid (HVA), nor-metanephrine (NM), 3-methoxy-4-hydroxymandelic acid (VMA), 3-methoxy-4-hydroxyphenylglycol (MHPG), dl-3,4-dihydroxyphenylglycol (DHPG), metanephrine (MET), reserpine, SCH 23390 (R(+)-7-Chloro-8-hydroxy-3-methyl-1-phenyl-2,3,4,5-tetrahydro-1H-3-benzazepine hydrochloride, L-741626 ((±)-3-[4-(4-Chlorophenyl)-4-hydroxypiperidinyl]methylindole), U-99194A (5,6-dimethoxy-2-(di-n-propylamino)indan maleate), L-741741 ((±)-3-[4-(4-Chlorophenyl)-4-hydroxypiperidinyl]methylindole), prazosin hydrochloride, CGP 20712A ((±)-2-hydroxy-5-[2-[2-hydroxy-3-[4-[1-methyl-4-(trifluoromethyl)-1H-imidazol-2-yl]phenoxy]propyl] amino]ethoxy]-benzamide methanesulfonate), ICI-118551 ((±)-1-[2,3-(dihydro-7-methyl-1H-inden-4-yl)oxy]-3-[(1-methylethyl)amino]-2-butanol hydrochloride), bovine serum albumin, HEPES, and PHA. RPMI 1640, heat-inactivated fetal calf serum (FCS), glutamine, and penicillin/streptomycin were from Celbio (Milan, Italy). Dextran and Ficoll-Paque Plus were from Pharmacia Biotech (Uppsala, Sweden). Rabbit anti–human D1, D3, D4, and D5 receptor polyclonal IgG were from Calbiochem (Merk Biosciences, Darmstadt, Germany), rabbit anti–human D2 receptor polyclonal IgG was from Santa Cruz Biotechnology (Santa Cruz, CA), and fluorescein isothiocyanate (FITC)–conjugated goat anti–rabbit IgG antibody was from Caltag Laboratories (Burlingame, CA). All other reagents and solvents were of high-performance liquid chromatography (HPLC) grade and were obtained from Merck Biosciences.
Purification of human Tregs and Teffs
Tregs and Teffs were isolated from buffy coat preparations derived from the whole blood of healthy donors by immunomagnetic sorting using the Dynal CD4+CD25+ Treg Kit (Dynal, Oslo, Norway), according to the manufacturer's instructions. Both Tregs and Teffs were finally free from residual beads (as assessed by electron microscopy), their purity was always more than 95% by flow cytometry, and their vitality was more than 99% by the Trypan Blue exclusion test. The artificial activation of the cells was excluded by cytofluorimetric analysis of surface expression of CD69, which was always less than 1%. For subsequent culture, cells were resuspended in RPMI 1640 medium supplemented with 10% heat-inactivated FCS, 2 mM glutamine, and 100 U/mL penicillin/streptomycin, at the final concentration of 1 × 106 cells/mL, and incubated at 37°C in a moist atmosphere of 5% CO2.
High-performance liquid chromatography (HPLC) with multielectrode electrochemical detection
Catecholamines in the cells and in the culture medium were assayed by high-performance liquid chromatography with multielectrode electrochemical detection (HPLC-ED). To this end, 1 × 106 cells were processed as described previously,14 and 30 μL recovered supernatant was injected into the chromatographic system. The medium was added with HClO4 0.4 N (1:1 vol/vol), kept on ice for 30 minutes, and then centrifuged for 5 minutes at 15 000g and 20°C. Again, the supernatant was recovered and filtered, and 30 μL was injected into the HPLC system. The system consisted of a dual-piston pump (model LC10ADVP; Shimadzu, Kyoto, Japan), a Beckman Ultrasphere IP C18 5-μm analytical column equipped with an Ultrasphere IP C18 guard cartridge (Beckman Instruments, Fullerton, CA), an autosampler (model HT 300L; HTA, Brescia, Italy) equipped with a 6-port injector valve (W Type; Vici Valco Instruments, Houston, TX), and a multielectrode electrochemical detector (Coularray 5600A; ESA, Bedford, MA) with a 4-channel sensor cell (model 6210; ESA). The mobile phase was composed as follows: ultrapure water–acetonitrile (90.7:9.3 vol/vol), 0.049 M KH2PO4, 0.349 mM Na2EDTA, and 0.6264 mM octansolfonic acid as the counter ion. The pH was adjusted to 3.20 with H3PO4. Freshly prepared mobile phase was filtered (0.45 μm) and degassed under vacuum for 10 minutes. The flow rate was 0.8 mL/min. The multielectrode detector operated in the oxidation-reduction mode, with the potentials of the 4 electrodes set at 150 mV, 250 mV, 300 mV, and −200 mV. The method allowed the simultaneous resolution and measurement over 22 minutes of the following catecholic compounds: l-DOPA; DA; NE; E; the DA metabolites DOPAC, 3-MT, and HVA; the NE metabolites MHPG, DHPG, VMA, and NM; and the E metabolite MET. The chromatograms were collected, stored, and processed with the application software Coularray for Windows (ESA). Catecholamines and metabolites in the samples were quantified by using the peak heights of a standard curve generated by injecting known samples (3 fmol to 3 pmol), and values were finally normalized for cell number. By use of this method, the lowest amounts of catecholamines and metabolites that could be assayed were 0.02 to 0.10 × 10−12 mol/106 cells or per milliliter.
Reverse transcriptase–polymerase chain reaction (RT-PCR)
Total RNA was extracted from 100 000 cells by Perfect RNA Eukaryotic Mini Kit (Eppendorf, Hamburg, Germany). RNA samples were then treated with DNAase I (Gibco, Eggenstein, Germany) for 15 minutes at room temperature. Extracted RNA was quantified by spectrophotometry at 260 nm and reverse transcribed, and cDNA was amplified using a 1-step RT-PCR reaction kit (Finnzymes, Espoo, Finland). PCR was performed according to the following steps: 30 minutes at 48°C for the reverse transcription, then 45 cycles (15 seconds at 95°C, 1 minute at 58°C, 80 seconds at 72°C), and a final 5-minute extension period at 72°C. Samples were then chilled at −4°C until analysis, which was performed on 10 μL RT-PCR product in 2% agarose gel in the presence of ethidium bromide (BIORAD, Hercules, CA). Quantification of the products was accomplished by densitometric analysis of photographic negatives of the gel, using a commercial software (Multi-Analyst; BIORAD). Data were finally expressed as optical density (OD) in arbitrary units and normalized for the expression of β-actin mRNA. Primers were obtained from Invitrogen (San Giuliano Milanese, MI, Italy), and their sequences are shown in Table 1
Enzyme-linked immunosorbent assays (ELISAs)
Concentrations of cytokines in culture supernatants and intracellular cAMP levels were determined by using commercial ELISA kits (Amersham, Buckinghamshire, United Kingdom).
Proliferation and inhibition assay
PHA (10 μg/mL) was added to 1 × 105 Tregs and 1 × 105 Teffs, alone or placed in the same well (Tregs/Teffs ratio = 1:1), and proliferation was measured after 2 days of culture by using a colorimetric immunoassay for the quantification of cell proliferation, based on the ELISA measurement of BrdU incorporated during DNA synthesis (Amersham).
Stimulation with anti-CD3 (10 μg/mL) and anti-CD28 (10 μg/mL) (Becton Dickinson Biosciences, Franklin Lakes, NJ) was performed on 5 × 104 Tregs and 5 × 104 Teffs, alone or placed in the same well (Tregs/Teffs ratio = 1:1). Experiments were run in triplicate in 96-well round-bottomed plates with and without stimuli for 96 hours and then pulsed for an additional 18 hours with 1 μCi (0.037 MBq) 3H thymidine (3H-TdR)/well (Pharmacia Biotech). Then, cells were harvested and the radionuclide uptake was measured by scintillation counting. To calculate the rate of suppression of Tregs, the following formula was used: [(cpm of Teffs − cpm of coculture)/cpm of Teffs] × 100.
Flow cytometric analysis of dopamine receptors on Teffs and Tregs
Freshly isolated Teffs and Tregs were subjected to an indirect immunofluorescence staining, using rabbit polyclonal IgG directed against human dopaminergic D1-like (D1 and D5) and D2-like (D3, D4, and D5) receptors. Briefly, 1 × 106 cells were incubated with each antibody at 1:25 dilution in 100 μL for 30 minutes on ice. Cells were then resuspended in 100 μL PBS and stained with an FITC-conjugated goat anti–rabbit IgG antibody (10 μL at 1:10 dilution). In each analysis, samples of the same specimens were stained with the secondary antibody alone (negative control) to assess nonspecific fluorescence intensity values.
Flow cytometric analysis was performed on an EPICS Elite ESP (Beckman Coulter, Miami, FL), and a minimum of 10 000 cells was analyzed from each sample. The results were finally expressed as percentage of positive cells (%) and as receptor density on positive cells (mean channel fluorescence intensity [MFI], ie, the median value of fluorescence intensity distribution, in arbitrary units).
Statistics
Data are presented as mean ± SD, with n indicating the number of observations. Statistical significance of the differences between groups was assessed by 2-tailed Student t test or 1-way analysis of variance followed by Bonferroni or Dunnet posttest for paired or unpaired data, as appropriate. Calculations were performed using a commercial software (GraphPad Prism version 4.00 for Windows; GraphPad Software, San Diego, CA).
Results
Tregs contain high levels of intracellular catecholamines and express tyrosine hydroxylase, vesicular monoamine transporters, dopaminergic receptors, and adrenoceptors
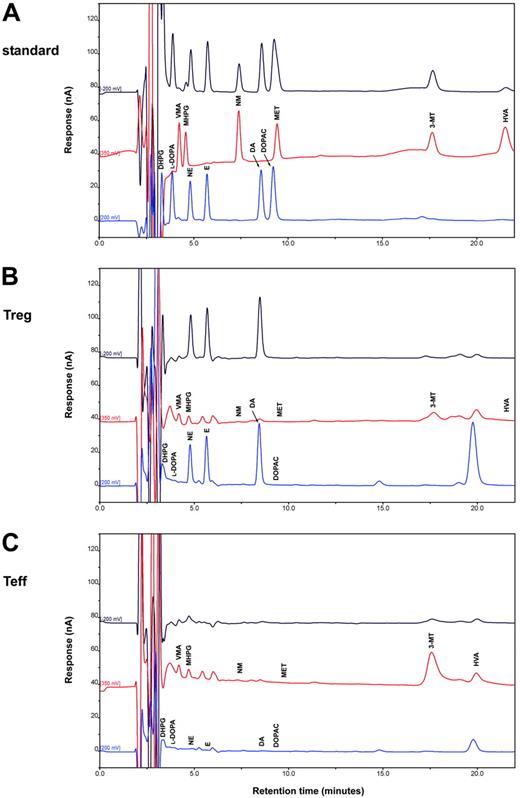
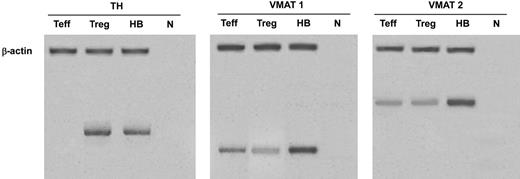
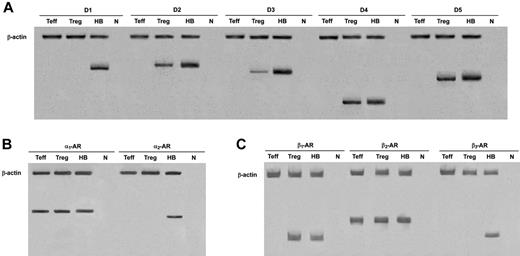
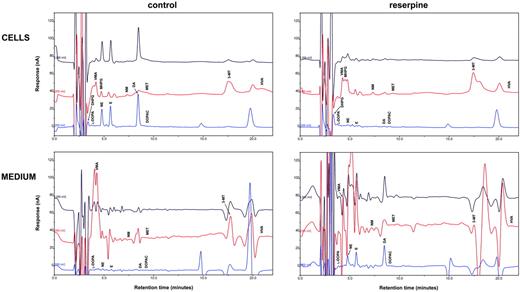
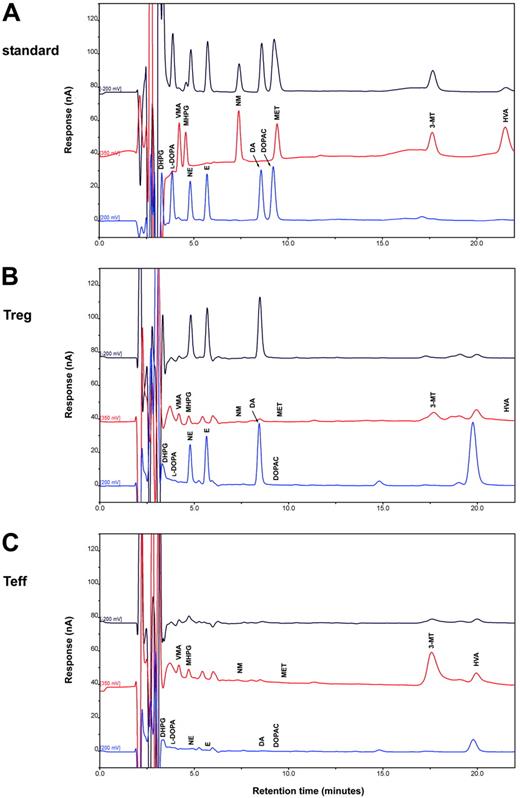
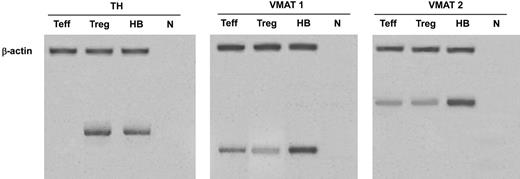
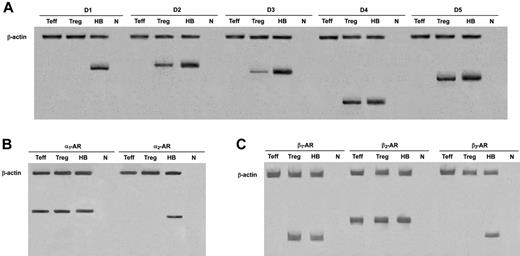
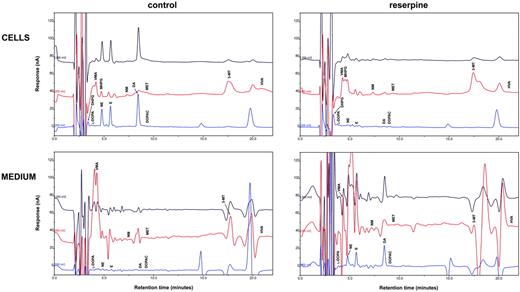
In human Tregs, HPLC-ED analysis detected DA, NE, and E, together with their common precursor l-DOPA and several of their major metabolites (Figure 1). In comparison with Teffs, in Tregs both l-DOPA and metabolite levels were similar, but DA, NE, and E were significantly higher (Table 2), from 19-fold (in the case of NE) to 83-fold (as for E) and even 170-fold (as in the case of DA), and reached values that are very close to those that we previously reported to occur in human lymphocytes after mitogen activation.16 Tregs also expressed the mRNA for TH, the rate-limiting enzyme in the synthesis of catecholamines, for vesicular monoamine transporter 1 (VMAT-1) and VMAT-2 (Figure 2), and the mRNAs for D2, D3, D4, and D5 dopaminergic receptors, as well as for α1-, β1-, and β2-ARs (Figure 3). No evidence was found for the expression of mRNAs for the D1-like D1 dopaminergic receptor or for α2- or β3-ARs (Figure 3). For comparison, Teffs expressed mRNA for both VMAT-1 and VMAT-2, but no detectable levels of mRNA for TH, and mRNA for α1- and β2-ARs, but neither for α2-, β1-, nor for β3-ARs or for any type of dopaminergic receptors (Figures 2–3).
HPLC-ED assay of endogenous catecholamines in Tregs and Teffs. Chromatograms of a standard sample containing 300 fmol catecholamines and of each of their metabolites (A) and of representative samples containing endogenous catecholamines and metabolites extracted from 1 × 106 human Tregs (B) and 1 × 106 Teffs (C). Results in Tregs and in Teffs are from 1 representative experiment of 6 separate experiments. For quantitative data, see Table 3.
HPLC-ED assay of endogenous catecholamines in Tregs and Teffs. Chromatograms of a standard sample containing 300 fmol catecholamines and of each of their metabolites (A) and of representative samples containing endogenous catecholamines and metabolites extracted from 1 × 106 human Tregs (B) and 1 × 106 Teffs (C). Results in Tregs and in Teffs are from 1 representative experiment of 6 separate experiments. For quantitative data, see Table 3.
Expression of mRNA for TH (left), VMAT 1 (center), and VMAT 2 (right) in Tregs and Teffs. HB indicates human brain (positive control); n, no RNA (negative control). Data are from 1 representative experiment of 3 separate experiments.
Expression of mRNA for TH (left), VMAT 1 (center), and VMAT 2 (right) in Tregs and Teffs. HB indicates human brain (positive control); n, no RNA (negative control). Data are from 1 representative experiment of 3 separate experiments.
Expression of mRNA for dopaminergic receptors, and α- and β-ARs in Tregs and Teffs. HB indicates human brain (positive control); n, no RNA (negative control). Data are from 1 representative experiment of 3 separate experiments.
Expression of mRNA for dopaminergic receptors, and α- and β-ARs in Tregs and Teffs. HB indicates human brain (positive control); n, no RNA (negative control). Data are from 1 representative experiment of 3 separate experiments.
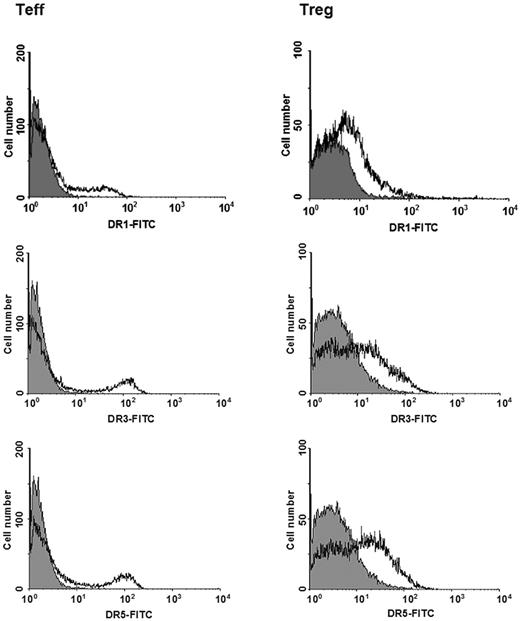
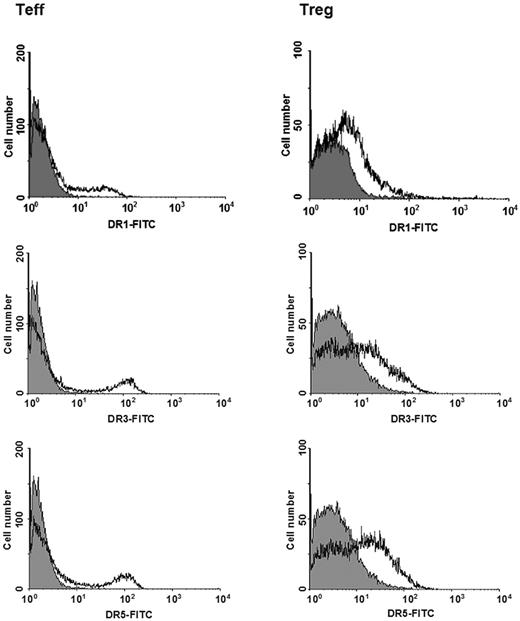
Expression of dopaminergic receptors was further explored by means of flow cytometry. Figure 4 shows 1 representative of 3 experiments performed on Teffs and Tregs obtained from different individuals, and Table 3 reports detailed results indicating that consistent subsets (20.9%-29.0% of total cells) of both Teffs and Tregs expressed dopaminergic receptors of the D2, D3, D4, and D5 subtypes. For all of these subtypes, percentage of positive cells and receptor density were not significantly different in Teffs and in Tregs. D1 receptors, on the contrary, were expressed in only 15.6% of Teffs and 11.6% of Tregs, and in these latter cells receptor density was also significantly lower than those of D1 in Teffs or of D3 and D5 in Tregs (Table 3).
Expression of dopaminergic D1, D3, and D5 receptors in Teffs and in Tregs. Open histograms indicate specific staining; solid histograms, negative control. Data are from 1 representative experiment of 3 separate experiments. For quantitative data, see Table 3.
Expression of dopaminergic D1, D3, and D5 receptors in Teffs and in Tregs. Open histograms indicate specific staining; solid histograms, negative control. Data are from 1 representative experiment of 3 separate experiments. For quantitative data, see Table 3.
Reserpine induces the release of catecholamines from Tregs
The intracellular levels of catecholamines and of their metabolites did not differ in freshly isolated Tregs and in Tregs cultured for 30 minutes (data shown in Tables 2 Only DOPAC levels increased from 0.21 ± 0.26 10−12 mol/106 cells (n = 6) to 0.54 ± 0.08 10−12 mol/106 cells (n = 6) (P < .05). As shown in Figure 5 and in Table 4, 1-hour incubation of Tregs with 1 μM reserpine (an agent that inhibits the uptake of catecholamines into chromaffin granules) induced a huge decrease of intracellular catecholamines, without affecting their metabolites, with the exception of the NE metabolite MHPG, which increased. The decrease was likely due to an overflow of catecholamines from the cells, as suggested by the concurrent increase of DA, NE, and E in the medium, where also the NE metabolite NM increased, while the DA metabolite 3-MT decreased (Table 4). When the total content per well (ie, cells + medium) of each catecholamine and metabolite was considered in the absence and in the presence of reserpine, as shown in the right-side columns of Table 4, it became evident that treatment with reserpine induced an overall increase of DA and E, as well as of the NE metabolite MHPG, and a decrease of the DA metabolite 3-MT.
Effect of reserpine on endogenous catecholamines in Tregs. Chromatograms showing the effect of 1-hour incubation with 10 μM reserpine on endogenous catecholamines and their metabolites in human Tregs. Results are from 1 representative of 6 separate experiments. For quantitative data, see Table 4.
Effect of reserpine on endogenous catecholamines in Tregs. Chromatograms showing the effect of 1-hour incubation with 10 μM reserpine on endogenous catecholamines and their metabolites in human Tregs. Results are from 1 representative of 6 separate experiments. For quantitative data, see Table 4.
Incubation of Treg/Teff coculture with reserpine impairs the suppression exerted by Tregs on the proliferation, but not cytokine secretion, of Teffs
Coculture of Teffs with Tregs 1:1 resulted in suppression of the proliferation of Teffs induced by either PHA (by 77.6% ± 14.1% of Teffs alone, n = 6; P < .001 versus Teffs alone) or anti-CD3/anti-CD28 antibodies (by 56.0% ± 4.3%, n = 6; P < .001 vs Teffs alone). Incubation with 1 μM reserpine significantly reduced the suppressive effect of Tregs, down to 1.2% ± 43.5% (stimulation with PHA) and to 22.1% ± 7.8% (stimulation with anti-CD3/anti-CD28 antibodies; in both cases, P < .001 versus Teffs + Tregs without reserpine, n = 3-6). Coculture of Teffs and Tregs also resulted in about 50% reduction of mitogen-induced production of TNF-α and interferon-γ (IFN-γ), but incubation with reserpine did not modify this effect (Table 5) In separate experiments, reserpine did not affect the percentage of viable cells in cultured Tregs and Teffs, alone and together, at rest and after mitogen stimulation (data not shown).
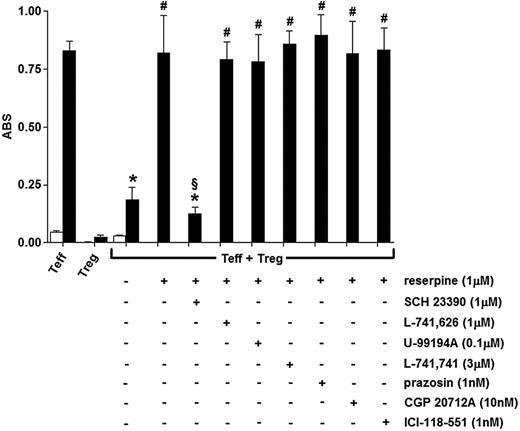
Dopaminergic D1/D5 receptor pathways mediate reserpine-induced suppression of Treg function
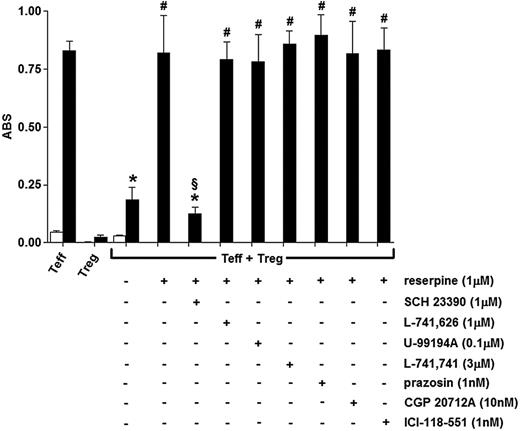
To assess whether the observed effect of reserpine was exerted through dopaminergic receptor– and/or AR-mediated pathways, the cocultures of Tregs with Teffs were incubated with 1 μM reserpine alone or in the presence of antagonists selective for D2, D3, D4, or D5 dopaminergic receptor subtypes or for α1-, β1-, and β2-ARs (ie, all the catecholamine receptors that were found to be expressed in Tregs). As shown in Figure 6, the effect of reserpine was completely reverted only in the presence of the D1/D5 selective antagonist SCH 23390, while none of the other antagonists had any significant effect. In separate experiments, 1 μM SCH 23390 alone had no effect per se on either Treg or Teff viability, or on mitogen-induced proliferation of Teffs (data not shown).
Effect of reserpine on the Treg-dependent suppression of mitogen-induced Teff proliferation. Cells were incubated for 48 hours alone (□) or in the presence of 10 μg/mL PHA (▪). Each column is the mean ± SEM of 5 separate experiments. *P < .01 versus Teffs + PHA; #P < .01 versus Teffs + Tregs + PHA; §P < .05 versus Teffs + Tregs + PHA + reserpine.
Effect of reserpine on the Treg-dependent suppression of mitogen-induced Teff proliferation. Cells were incubated for 48 hours alone (□) or in the presence of 10 μg/mL PHA (▪). Each column is the mean ± SEM of 5 separate experiments. *P < .01 versus Teffs + PHA; #P < .01 versus Teffs + Tregs + PHA; §P < .05 versus Teffs + Tregs + PHA + reserpine.
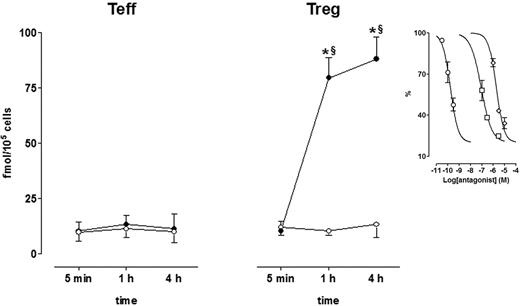
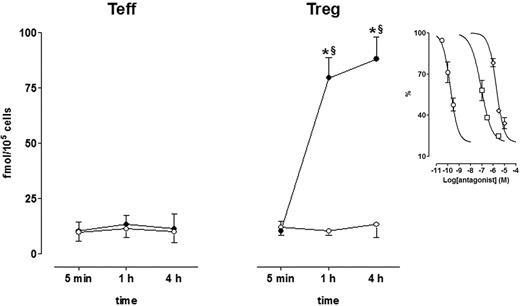
Dopaminergic D1/D5 receptors are functionally coupled to adenylate cyclase,45 and therefore, to investigate their function, intracellular cAMP levels were measured in Teffs and in Tregs in the absence and in the presence of DA. As shown in Figure 7, cAMP levels were similarly low in both Tregs and Teffs, and incubation with DA had no effect in Teffs but induced a huge and sustained increase of intracellular cAMP in Tregs, which was already evident at 5 minutes and could be detected still after 4 hours. DA-induced cAMP rise in Tregs was concentration-dependently inhibited by dopaminergic receptor antagonists (Figure 7) with the following IC50 values (with 95% confidence intervals) in nanomoles per liter: SCH 23390 = 0.17 (0.11-0.26); haloperidol = 87.3 (15.8-481.6); and spiperone = 1877 (81.5-4321).
Effect of DA on intracellular cAMP in Teffs and in Tregs. Cells were cultured alone (○, control) or in the presence of 1 μM DA (•). Each point is the mean ± SD of 3 separate experiments. *P < .01 versus 0 minutes; §P < .01 versus control at the corresponding time interval. (Inset) Effect of the dopaminergic receptor antagonists SCH 23390 (circles), haloperidol (squares), and spiperone (diamonds) on 1 μM DA–induced increase of cAMP after 4-hour culture. Data are expressed as percentage of the values obtained in the presence of DA alone. Each point is the mean ± SD of 3 separate experiments. See “Results” for IC50 values.
Effect of DA on intracellular cAMP in Teffs and in Tregs. Cells were cultured alone (○, control) or in the presence of 1 μM DA (•). Each point is the mean ± SD of 3 separate experiments. *P < .01 versus 0 minutes; §P < .01 versus control at the corresponding time interval. (Inset) Effect of the dopaminergic receptor antagonists SCH 23390 (circles), haloperidol (squares), and spiperone (diamonds) on 1 μM DA–induced increase of cAMP after 4-hour culture. Data are expressed as percentage of the values obtained in the presence of DA alone. Each point is the mean ± SD of 3 separate experiments. See “Results” for IC50 values.
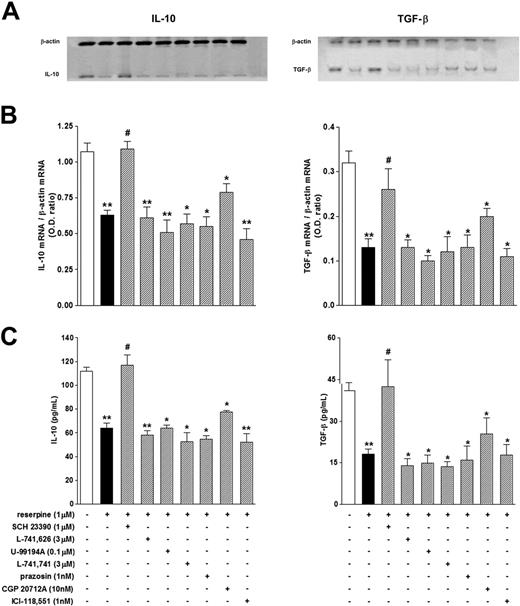
Effect of reserpine on IL-10 and TGF-β production by Tregs and Teffs and on FoxP3 mRNA expression in Tregs
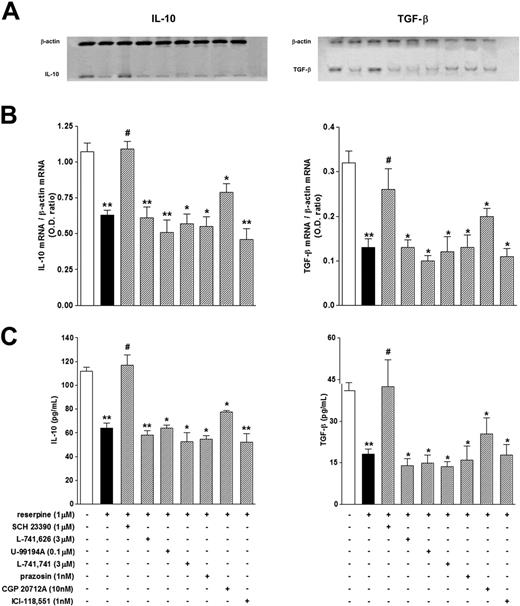
As shown in Table 6, both Tregs and Teffs produced IL-10 and TGF-β, but production of both cytokines was higher in Tregs than in Teffs. Production of IL-10 and TGF-β in Teffs was not affected by reserpine, but in Tregs it was significantly reduced, both at the mRNA and at the protein level (Table 6; Figure 8). As in the case of reserpine-induced reduction of the suppressive effect of Tregs, reserpine-induced inhibition of IL-10 and TGF-β production was also selectively reverted by pharmacological blockade of the D1/D5 receptor pathways, but not by blockade of α1-, β1-, and β2-AR pathways (Figure 8). FoxP3 mRNA was not detected in Teffs, while in Tregs its levels were 0.55 ± 0.09 (n = 9) and were not affected to any significant extent by 1 μM reserpine (0.48 ± 0.14, n = 9; P > .05).
Effect of reserpine on the production of IL-10 and of TGF-β by Tregs. Cells were incubated for 24 hours alone (□) or in the presence of reserpine without (▪) or with (▨) antagonists selective for dopaminergic receptors and ARs. Each column is the mean ± SEM of 3 to 9 separate experiments. *P < .05 and **P < .01 versus control; #P < .05 versus reserpine alone.
Effect of reserpine on the production of IL-10 and of TGF-β by Tregs. Cells were incubated for 24 hours alone (□) or in the presence of reserpine without (▪) or with (▨) antagonists selective for dopaminergic receptors and ARs. Each column is the mean ± SEM of 3 to 9 separate experiments. *P < .05 and **P < .01 versus control; #P < .05 versus reserpine alone.
Discussion
The main finding of the present study is that human Tregs selectively express TH, the key enzyme in the synthesis of catecholamines, and contain high amounts of catecholamines stored in reserpine-sensitive compartments, which, upon release, subserve an autocrine/paracrine modulatory loop leading to impaired suppressive activity of Tregs toward mitogen-induced Teff proliferation. Pharmacological evidence indicates that functional impairment of Tregs depends upon the activation of dopaminergic D1-like receptor pathways and is due to inhibition of both IL-10 and TGF-β production by these cells.
Endogenous catecholamines in human Tregs and Teffs
Although trace amounts of catecholamines are usually detected in naive lymphocytes,11 we previously reported that mitogen stimulation is needed to trigger the expression of TH and the subsequent synthesis of DA, NE, and E,16 up to amounts that are able to affect lymphocyte function to a significant extent.18 Expression of TH and high intracellular levels of catecholamines in Tregs are therefore in line with the notion that these cells express the phenotype of activated lymphocytes.4 Catecholamines in human Tregs likely accumulate into specific storage sites, as suggested by the expression in these cells of the mRNA for VMAT-1 and VMAT-2 and by the outflow of catecholamines following incubation with reserpine, which inhibits the uptake of catecholamines into chromaffin granules. Depletion of intracellular stores (eg, after treatment with reserpine) is likely to trigger the whole catecholaminergic synthetic pathway, as indicated by the overall increase of DA (+70%) and E (+89%).
In this regard, the consequences of reserpine treatment on Treg function provide the first experimental evidence of the existence in human Tregs of an autocrine/paracrine loop subserved by endogenous catecholamines acting on the receptors expressed on these cells. This is indeed the most likely interpretation of the results showing the ability of reserpine, on one hand, to induce a complete overflow of endogenous catecholamines from Tregs and, on the other hand, to profoundly impair the inhibitory activity of Tregs on mitogen-induced proliferation of Teffs, apparently through reduced production of both IL-10 and TGF-β, which are the main immunosuppressive cytokines produced by Tregs.1
Human Teffs, on the other hand, contain only trace amounts of catecholamines that, due to their paucity, are unlikely to succeed in subserving any autocrine/paracrine regulatory loop, as indicated by the lack of effect of reserpine on IL-10 and TGF-β production in these cells. This is also in line with previous observations showing that only high intracellular levels of catecholamines occurring after lymphocyte activation are able to affect survival and death in these cells.18
Receptors for catecholamines in Tregs and Teffs
Our results show that Tregs express a wide array of dopaminergic receptors and of ARs, which are likely to represent suitable targets for endogenous catecholamines (and even for exogenous ligands). By use of RT-PCR, we detected mRNA for several subtypes of dopaminergic receptors (namely, the D1-like D5 and the D2-like D2, D3, and D4), as well as for α1-, β1-, and β2-ARs. Teffs, on the other hand, expressed mRNAs for only α1- and β2-ARs. As for ARs, the occurrence of α1- and β2-ARs on human T lymphocytes has been reported previously, and in particular β2-ARs are considered the main interface between the nervous system and the immune system (see, eg, Kohm and Sanders20 and Elenkov et al21 ). The occurrence of β1-ARs in Tregs was unanticipated, however, since to the best of our knowledge this particular AR type has never been detected in human lymphocytes. Until now, we were nonetheless unable to obtain evidence for any role of AR-operated pathways, since in pilot experiments neither NE nor E exerted any effect on the parameters under study up to micromolar concentrations (M.C., unpublished observations, March 2004).
Indeed, several lines of evidence indicate that the suppressive effect of reserpine on Tregs is exerted through dopaminergic D1-like receptor pathways, since it was selectively sensitive to blockade of D1-like receptors but not of other dopaminergic receptors and/or ARs, and in preliminary experiments it was mimicked by DA and selective D1-like receptor agonists, but not by agonists selective for D2-like receptors or ARs, nor by NE or E (M.C., unpublished observations, March 2004). Recently, it was shown that DA down-regulates Treg function in the mouse via D1-like dopaminergic receptors.31 Mouse lymphocytes indeed contain endogenous catecholamines6 ; however, at present no evidence exists for any autocrine/paracrine loop in these cells.
In view of their functional relevance, we investigated more in-depth dopaminergic receptors on Tregs and Teffs. First, by means of flow cytometry, we demonstrated membrane expression of dopaminergic receptors of both the D1-like (D1 and D5 on both Teffs and Tregs) and the D2-like (D2, D3, and D4 on Teffs, and at least D3 on Tregs; D2 and D4 were not investigated on these cells due to the limitations imposed by the reduced number of cells recovered at each separation) subtypes (Table 2). Both the percentage of positive cells as well as receptor density per cell were similar in Tregs and Teffs (with the only notable exception of D1 receptors on Tregs, which were expressed to a very limited extent). This finding was surprising, as in our experiments Tregs and Teffs clearly differ in both expression of receptor mRNA and response to dopaminergic agents (both observed in Tregs only). We turned therefore to receptor function by measuring intracellular levels of cAMP, which is the second messenger acted upon by D1-like receptors (see, eg, Seeman45 ), and such experiments unequivocally showed that receptor–second messenger coupling occurs in Tregs only (Figure 7). We cannot exclude that dopaminergic receptors on Teffs may respond; however, our results do not support this possibility.
Currently available pharmacological tools do not allow one to distinguish between D1 and D5 receptors: SCH 23390 shows similar affinities for D1 (Ki ∼ 0.2 nM) and D5 (Ki = 0.3 nM).46 We therefore tried to obtain some indication about the relative importance of these 2 subtypes in Tregs by using 2 D2-like selective antagonists that nonetheless clearly differ in their relative affinity for D1 and D5 receptors, namely haloperidol (Ki at D1 ∼ 80 nM, and at D5 ∼ 100 nM) and spiperone (Ki at D1 ∼ 350 nM, and at D5 ∼ 3500 nM).46 The spiperone-haloperidol affinity ratio is therefore approximately 4.4 at D1 and approximately 35 at D5, and the spiperone/SCH 23390 affinity ratio is approximately 1750 at D1 and 11 667 at D5. In our experiments, the IC50s of haloperidol, spiperone, and SCH 23390 on DA-induced cAMP increase in Tregs were 87.3 nM, 1877 nM, and 0.17 nM, respectively, with a spiperone-haloperidol potency ratio of approximately 21.5 and a spiperone/SCH 23390 potency ratio of approximately 11 041. These results, together with the observation that on Tregs D1 receptors are expressed to a more limited extent than D5, suggest that D5 receptors are the main receptor responsible for dopaminergic inhibition of Treg function. In any case, in our experimental conditions we could not account for a number of factors possibly influencing receptor response (eg, high/low-affinity state of the receptors, receptor coupling to G proteins46 ), thus our data should be taken only as a preliminary indication. The future availability of new agents with a better D1/D5 selectivity profile will eventually allow for a better definition of the relative role of D1-like receptor subtypes on human Tregs.
Dopaminergic modulation of Treg and Teff function
The effects of reserpine and of dopaminergic agents support the existence of a dopaminergic inhibitory loop sustained by endogenous catecholamines regulating Treg function. Dopaminergic effects include inhibition of IL-10 and TGF-β production and impairment of the ability of Tregs to suppress Teff proliferation. The effects seem to be selectively exerted on Tregs, since in our experiments, reserpine affected neither Teff production of IL-10 or TGF-β, nor Teff proliferation in the absence of Tregs. In human T cells, DA triggers the production of IL-10.25 However Teffs contain only trace amounts of DA (and of other catecholamines), therefore endogenous autocrine/paracrine loops are unlikely. Reserpine also had no effect on TNF-α or IFN-γ production in Teff/Treg cocultures, suggesting that Treg-induced inhibition of the production of these cytokines is independent from the dopaminergic control of these cells. As in the case of IL-10, TNF-α production is also induced by DA in T lymphocytes.25 Failure of reserpine to affect TNF-α (and IFN-γ) production in Teff/Treg cocultures, where DA is released by Tregs themselves, may imply that Treg-derived DA is released in amounts sufficient to act in an autocrine way on Tregs themselves but not in a paracrine way on neighboring Teffs, and/or that TNF-α (and IFN-γ) production by Teffs is not modulated by dopaminergic receptors. Teffs are exclusively CD4+ cells, and it cannot be excluded that reported results in human T lymphocytes25 may depend upon the activity of CD4 subsets.
DA and dopaminergic agents may indeed profoundly influence the immune response acting at several levels,28 thus the net result is likely to depend on the integration of the effects exerted on the various components of the response. Moreover, dopaminergic receptor function may depend upon the status of T lymphocytes (eg, resting or activated): D3 receptors mediate stronger responses in activated in comparison with resting T lymphocytes,24 and we have recently reproduced and extended those findings to the D2, D4, and D5 subtypes (M.F., unpublished observations, October 2005). Lymphocyte activation may therefore profoundly affect the response to dopaminergic agents, a phenomenon that could contribute to explain the heterogeneous and sometimes conflicting results so far obtained.22,24,29,30,47
Dependence of the functional response upon time of exposure and functional status of the cells has been studied in depth for noradrenergic pathways. Murine CD4+ effector T cells differentially express β2-ARs, both at rest and following activation, with detectable expression on Th1 but not on Th2 cells.48 Interestingly, although both NE and E inhibit PHA-induced synthesis of IFN-γ by murine immune spleen cells,49 more IFN-γ is produced by Th1 cells generated from NE-exposed naive T cells.50 Moreover, exposure of Th1 cells to NE or to the β2-AR agonist terbutaline decreases IL-2 production upon the first activation but not upon restimulation.51 TH-dependent production of catecholamines in human lymphocytes is triggered upon activation,16 affects activation-dependent apoptosis,18 and is down-regulated by D1-like receptor activation,17 an observation that further adds to the complexity of the catecholaminergic network in immune cells. Indeed, it cannot be excluded that catecholamines released by Tregs, while suppressing Tregs themselves, may also result in direct activation of Teffs.22,25
Clinical implications of dopaminergic modulation of Treg function
Although much investigation is still needed to unravel the complex mechanisms acted upon by catecholamines in the immune system, the results of the present study support dopaminergic D1-like receptor pathways as targets for the functional modulation of human Tregs. In view of the key role played by Tregs in the control of immune homeostasis, this finding has a potentially very wide array of clinical implications. Hereafter, we will briefly discuss those related to antitumor immunity and to autoimmune disease, since in such conditions the therapeutic use of dopaminergic agents has often been attempted, although with disappointing results. DA has been proposed to regulate malignant cell proliferation and to control immune functions both directly and indirectly (reviewed in Basu and Dasgupta52 ). More than 2 decades ago, DA was suggested as a novel antitumor agent against, for example, melanoma53 ; however, its possible mechanism(s) of action remained ill defined.54 Recently, in a mouse model of carcinoma, DA improved the antitumor efficacy of cyclophosphamide while reducing its hematotoxicity.55 Further investigations are needed to confirm those results, as well as to assess their clinical relevance, but the possibility that DA as an antitumor agent may also act through down-regulation of Tregs that maintain tolerance toward tumor cells cannot be excluded.56 Interestingly, we have recently reported that activation of IFN type I receptors effectively induces catecholamine release from activated lymphocytes,57 and we are currently studying the effects of type I IFNs on human Tregs; the hypothesis that the antitumor efficacy of IFN-α58,59 may be attributed at least in part to down-regulation of Treg function through DA release leading to activation of D1-like receptors is quite intriguing. As for autoimmunity, the involvement of dopaminergic mechanisms has usually been explained taking into account the ability of dopaminergic agonists to suppress the pituitary secretion of prolactin, a hormone that is considered to have a pathogenic role—for example, in rheumatoid arthritis, systemic lupus erythematosus, and multiple sclerosis.60 However, autoimmune disease seems to be associated with altered brain dopaminergic neurotransmission in animal models,61 as well as with impaired dopaminergic mechanisms in peripheral lymphocytes, in particular in multiple sclerosis patients.18,62 Thus the contribution of dopaminergic pathways to autoimmunity is likely to be more complex than previously thought, also involving peripheral mechanisms. Failure to replicate in clinical trials the usually favorable results obtained with dopaminergic agents in animal models of autoimmune disease could possibly also be ascribed to the still-limited comprehension of the contribution of such mechanisms.
Exploitation of endogenous catecholamines in human lymphocytes as novel targets for therapeutic interventions still requires clarification of the functional network sustained by these mediators (Figure 9), which nonetheless could be approached by several different pharmacological strategies. Lymphocyte catecholamines can be modulated at the following levels: (a) intracellular storage (as shown in the present study); (b) receptor activation/blockade by selective agonists/antagonists; (c) inhibition of catecholamine synthesis (eg, using the antihypertensive agent α-methyl-p-tyrosine, a TH selective inhibitor); and (d) enhancement of catecholamine synthesis (eg, using the anti-Parkinson drug l-DOPA, the direct DA precursor). Reserpine is currently used as an antihypertensive agent, although its use is limited by a burden of adverse effects. Other therapeutic agents, however, have been shown at least in vitro to interfere with the storage of catecholamines in lymphocytes, such as tetrabenazine63 and type I IFNs.57 Interestingly, IFN-β restores dopaminergic signaling in lymphocytes,62 indicating that its effect on lymphocyte catecholamines is exerted at several levels.
The cellular network sustained by endogenous catecholamines in human lymphocytes. Speculative scheme depicting the possible actions of endogenous catecholamines produced and released by human lymphocytes in the development of the immune response. Tregs constitutively express TH, the key enzyme in the synthesis of catecholamines, dopaminergic receptors, and α- and β-ARs, and contain high amounts of catecholamines stored in reserpine-sensitive compartments. Upon release, endogenous catecholamines (likely DA) subserve an autocrine/paracrine modulatory loop involving the activation of dopaminergic D1-like (D1/D5) receptors, leading to impaired suppressive activity of Tregs toward mitogen-induced Teff proliferation. In addition to the chromaffin granule depletant reserpine (used in the present study), candidate agents that may induce the release of catecholamines from lymphocytes include type I IFNs, tetrabenazine, as well as even high [K+]e. The picture also summarizes data from available literature, showing that, in the absence of stimulation, effector T lymphocytes express dopaminergic receptors and α- and β-ARs and contain trace amounts of DA, NE, and E. Upon stimulation, intracellular catecholamines increase by several 10-fold, and expression and function of both dopaminergic receptors and ARs may also undergo significant changes. Under these conditions, endogenous catecholamines either may directly affect cell survival and apoptosis from within the cell (lightning bolts), or they can be released (red arrows) to act upon lymphocytes themselves and/or upon neighboring cells. Dopaminergic receptors and α- and β-ARs on Teffs may also represent the targets for catecholamines released by nearby Tregs, which might also result in direct activation of nearby Teffs, resulting, for example, in cytokine secretion and integrin-mediated T-cell adhesion to the extracellular matrix (and, in turn, activated Teffs themselves may represent local sources of DA acting upon Tregs and finally resulting in down-regulation of their inhibitory function). Question marks highlight various issues that await clarification. For the sake of clarity and simplicity, the picture does not include the potential role of catecholamines that are normally present in the extracellular space or that can be released from sympathoadrenergic terminals innervating lymphoid organs and tissues, nor does it include catecholamines, which lymphocytes can encounter when they enter the brain in physiological (or pathological) situations. Please see “Discussion” for a detailed discussion of the various aspects depicted in the figure as well as for specific bibliographic references.
The cellular network sustained by endogenous catecholamines in human lymphocytes. Speculative scheme depicting the possible actions of endogenous catecholamines produced and released by human lymphocytes in the development of the immune response. Tregs constitutively express TH, the key enzyme in the synthesis of catecholamines, dopaminergic receptors, and α- and β-ARs, and contain high amounts of catecholamines stored in reserpine-sensitive compartments. Upon release, endogenous catecholamines (likely DA) subserve an autocrine/paracrine modulatory loop involving the activation of dopaminergic D1-like (D1/D5) receptors, leading to impaired suppressive activity of Tregs toward mitogen-induced Teff proliferation. In addition to the chromaffin granule depletant reserpine (used in the present study), candidate agents that may induce the release of catecholamines from lymphocytes include type I IFNs, tetrabenazine, as well as even high [K+]e. The picture also summarizes data from available literature, showing that, in the absence of stimulation, effector T lymphocytes express dopaminergic receptors and α- and β-ARs and contain trace amounts of DA, NE, and E. Upon stimulation, intracellular catecholamines increase by several 10-fold, and expression and function of both dopaminergic receptors and ARs may also undergo significant changes. Under these conditions, endogenous catecholamines either may directly affect cell survival and apoptosis from within the cell (lightning bolts), or they can be released (red arrows) to act upon lymphocytes themselves and/or upon neighboring cells. Dopaminergic receptors and α- and β-ARs on Teffs may also represent the targets for catecholamines released by nearby Tregs, which might also result in direct activation of nearby Teffs, resulting, for example, in cytokine secretion and integrin-mediated T-cell adhesion to the extracellular matrix (and, in turn, activated Teffs themselves may represent local sources of DA acting upon Tregs and finally resulting in down-regulation of their inhibitory function). Question marks highlight various issues that await clarification. For the sake of clarity and simplicity, the picture does not include the potential role of catecholamines that are normally present in the extracellular space or that can be released from sympathoadrenergic terminals innervating lymphoid organs and tissues, nor does it include catecholamines, which lymphocytes can encounter when they enter the brain in physiological (or pathological) situations. Please see “Discussion” for a detailed discussion of the various aspects depicted in the figure as well as for specific bibliographic references.
In view of the wide array of dopaminergic and adrenergic agents currently in clinical use for various indications and of their usually favorable therapeutic index, in-depth characterization of these mechanisms in the functional modulation of human lymphocytes could provide the rationale for further investigation examining the therapeutic potential of such agents in a number of major disease conditions.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Contribution: M.C. defined the experimental strategy, supervised the experiments, data analysis, and interpretation, and wrote the paper; A.M.F. discussed the experimental strategy, and participated in data analysis and drafting of the paper; M.F., E.R., and R.B. designed and performed the experiments, analyzed the data, and discussed and approved the paper; E.C. and F.S. performed the experiments, participated in data analysis, and approved the paper; F. Meloni, F. Marino, and S.L. discussed the experimental strategy and the interpretation of the data, and discussed and approved the paper.
Acknowledgments
This work was supported by grants from the Italian Ministry of Instruction, University and Research–MIUR (PRIN 2003, project no. 2003062975_003 to S.L.), from the Italian Foundation for Multiple Sclerosis–FISM (grant no. 2003/R/67 to M.C.), and from the University of Insubria (FAR 2004 and 2005 to M.C.).
The authors are grateful to Dr Davide Rossi and Dr Simona Cattaneo, Immunohematology and Transfusional Service, Ospedale di Circolo (Varese, Italy) who collaborated in providing human blood. The helpful assistance of Dr Ramòna Consuélo Maio in performing ELISA assays and of Ms Michela Perusin in HPLC-ED analysis of catecholamines as well as the skillful collaboration of Dr Massimiliano Legnaro in flow cytometry assays of dopaminergic receptors are gratefully acknowledged. The authors are also indebted to Dr Mauro Zaffaroni (Multiple Sclerosis Center, Hospital Sant'Antonio Abaté, Gallarate [VA], Italy) for his valuable contribution to the critical evaluation and interpretation of results.

![Figure 9. The cellular network sustained by endogenous catecholamines in human lymphocytes. Speculative scheme depicting the possible actions of endogenous catecholamines produced and released by human lymphocytes in the development of the immune response. Tregs constitutively express TH, the key enzyme in the synthesis of catecholamines, dopaminergic receptors, and α- and β-ARs, and contain high amounts of catecholamines stored in reserpine-sensitive compartments. Upon release, endogenous catecholamines (likely DA) subserve an autocrine/paracrine modulatory loop involving the activation of dopaminergic D1-like (D1/D5) receptors, leading to impaired suppressive activity of Tregs toward mitogen-induced Teff proliferation. In addition to the chromaffin granule depletant reserpine (used in the present study), candidate agents that may induce the release of catecholamines from lymphocytes include type I IFNs, tetrabenazine, as well as even high [K+]e. The picture also summarizes data from available literature, showing that, in the absence of stimulation, effector T lymphocytes express dopaminergic receptors and α- and β-ARs and contain trace amounts of DA, NE, and E. Upon stimulation, intracellular catecholamines increase by several 10-fold, and expression and function of both dopaminergic receptors and ARs may also undergo significant changes. Under these conditions, endogenous catecholamines either may directly affect cell survival and apoptosis from within the cell (lightning bolts), or they can be released (red arrows) to act upon lymphocytes themselves and/or upon neighboring cells. Dopaminergic receptors and α- and β-ARs on Teffs may also represent the targets for catecholamines released by nearby Tregs, which might also result in direct activation of nearby Teffs, resulting, for example, in cytokine secretion and integrin-mediated T-cell adhesion to the extracellular matrix (and, in turn, activated Teffs themselves may represent local sources of DA acting upon Tregs and finally resulting in down-regulation of their inhibitory function). Question marks highlight various issues that await clarification. For the sake of clarity and simplicity, the picture does not include the potential role of catecholamines that are normally present in the extracellular space or that can be released from sympathoadrenergic terminals innervating lymphoid organs and tissues, nor does it include catecholamines, which lymphocytes can encounter when they enter the brain in physiological (or pathological) situations. Please see “Discussion” for a detailed discussion of the various aspects depicted in the figure as well as for specific bibliographic references.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/109/2/10.1182_blood-2006-01-028423/4/m_zh80020706780009.jpeg?Expires=1771011805&Signature=yynUCvmEz52txL9gdNh8KIdGzZ-bjp3tvSuVXISs3sUQkw3qcHWSn~hYzWXFeK7H4KtjxwF7wfqRMSyoFJYS-jVEq-e~Q4TNUNNVPBvHZp-jZT2bWlsQzcG9oBoqB5F0D6X9syOlIxRjv22mhpKSuSl-BeqmVKjifh7afiQcTQj3Li-~1GAKuR3YHuoD~X9yEYwkEI2xa~6AcD0OLRUOUVhGG2iOG~Z43n0cypw8ZBpy9g5wbLpbVPXN1Amkb2qAQXCpAiPIHjSzx0R3-xjUs3rbXSkzonAe-xHkRtAScRfec-2jf~dCu84EREknAEhIbRJxoNXjPFjctu-uI~ABMA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![Figure 9. The cellular network sustained by endogenous catecholamines in human lymphocytes. Speculative scheme depicting the possible actions of endogenous catecholamines produced and released by human lymphocytes in the development of the immune response. Tregs constitutively express TH, the key enzyme in the synthesis of catecholamines, dopaminergic receptors, and α- and β-ARs, and contain high amounts of catecholamines stored in reserpine-sensitive compartments. Upon release, endogenous catecholamines (likely DA) subserve an autocrine/paracrine modulatory loop involving the activation of dopaminergic D1-like (D1/D5) receptors, leading to impaired suppressive activity of Tregs toward mitogen-induced Teff proliferation. In addition to the chromaffin granule depletant reserpine (used in the present study), candidate agents that may induce the release of catecholamines from lymphocytes include type I IFNs, tetrabenazine, as well as even high [K+]e. The picture also summarizes data from available literature, showing that, in the absence of stimulation, effector T lymphocytes express dopaminergic receptors and α- and β-ARs and contain trace amounts of DA, NE, and E. Upon stimulation, intracellular catecholamines increase by several 10-fold, and expression and function of both dopaminergic receptors and ARs may also undergo significant changes. Under these conditions, endogenous catecholamines either may directly affect cell survival and apoptosis from within the cell (lightning bolts), or they can be released (red arrows) to act upon lymphocytes themselves and/or upon neighboring cells. Dopaminergic receptors and α- and β-ARs on Teffs may also represent the targets for catecholamines released by nearby Tregs, which might also result in direct activation of nearby Teffs, resulting, for example, in cytokine secretion and integrin-mediated T-cell adhesion to the extracellular matrix (and, in turn, activated Teffs themselves may represent local sources of DA acting upon Tregs and finally resulting in down-regulation of their inhibitory function). Question marks highlight various issues that await clarification. For the sake of clarity and simplicity, the picture does not include the potential role of catecholamines that are normally present in the extracellular space or that can be released from sympathoadrenergic terminals innervating lymphoid organs and tissues, nor does it include catecholamines, which lymphocytes can encounter when they enter the brain in physiological (or pathological) situations. Please see “Discussion” for a detailed discussion of the various aspects depicted in the figure as well as for specific bibliographic references.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/109/2/10.1182_blood-2006-01-028423/4/m_zh80020706780009.jpeg?Expires=1771228326&Signature=vRw16Cm2Sa52vYorcUsIAkIQ9BFlmmJkU2h3Qj9Un9r0CUAWczncudQgyCkDFk6whyaRmUEMC~9AmN1JGtYIugkleDlDFjKYQ8YH4nJPO4tLQvMPuQD40PZg21I4PqUajlznNCxFV1mMF9UnLuw~j-NKjF5S7a5iBHz6mVLCLUDrI9pRyBWbd1jXTE0B1CAEht8-~eH-9XXFTNKu3LYs-POTz1-y7Nxw9MsmhexiRBSVQsmwuJOqo2weBrjUWB9yFpbcaOo0nogME~w9d011f-EmdgVLGfuBt1lMhoFsJNnTJdkU959to3qviEdNKpfdgHoC7f-inGOcPucvaZdw9A__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)