Abstract
Although recent advances have enabled hematopoietic stem cells (HSCs) to be enriched to near purity, more information about their characteristics will improve our understanding of their development and stage-related functions. Here, using microarray technology, we identified endothelial cell-selective adhesion molecule (ESAM) as a novel marker for murine HSCs in fetal liver. Esam was expressed at high levels within a Rag1− c-kitHi Sca1+ HSC-enriched fraction, but sharply down-regulated with activation of the Rag1 locus, a valid marker for the most primitive lymphoid progenitors in E14.5 liver. The HSC-enriched fraction could be subdivided into 2 on the basis of ESAM levels. Among endothelial antigens on hematopoietic progenitors, ESAM expression showed intimate correlation with HSC activity. The ESAMHi population was highly enriched for multipotent myeloid-erythroid progenitors and primitive progenitors with lymphopoietic activity, and exclusively reconstituted long-term lymphohematopoiesis in lethally irradiated recipients. Tie2+ c-kit+ lymphohematopoietic cells in the E9.5–10.5 aorta-gonad-mesonephros region also expressed high levels of ESAM. Furthermore, ESAM was detected on primitive hematopoietic progenitors in adult bone marrow. Interestingly, ESAM expression in the HSC-enriched fraction was up-regulated in aged mice. We conclude that ESAM marks HSC in murine fetal liver and will facilitate studies of hematopoiesis throughout life.
Introduction
Hematopoietic stem cells (HSCs) are defined as cells with the capacity for self-renewal as well as differentiation into multilineage blood cells, maintaining the immune system throughout life. A large body of information exists about molecular mechanisms involved in maintaining their integrity, and many studies have attempted to identify unique markers associated with these extremely rare cells. In bone marrow of adult mice, the Lin− c-kitHi Sca1+ CD34−/Lo Thy1.1Lo subset is known to include HSCs with long-term repopulating capacity.1 However, several of these parameters differ between strains of mice, change dramatically during developmental age or inflammation, and are expressed on many non-HSCs.2-4 The recent identification of CD150/SLAM as stable markers made it possible to increase the purity of HSCs even in aged mice or cyclophosphamide/granulocyte colony-stimulating factor-treated mice with mobilized progenitors.5 However, even the most highly purified HSCs are heterogeneous, and it may eventually be possible to associate discrete functions or activity states with subpopulations. Additional authentic HSC markers could have utility in attempts to rescue hematopoietic disorders using hematopoietic progenitors obtained from reprogrammed adult tissues.6,7
HSCs are thought to arise initially from hemogenic endothelium, which can produce hematopoietic cells as well as endothelial cells. Therefore, it is not surprising that HSCs share some endothelial properties at early developmental stages.8,9 For example, the CD34 sialomucin and Tie2, an angiopoietin receptor, are expressed on HSCs in E10–11 embryos.10,11 Endoglin and vascular-endothelial cadherin are additional endothelial markers found on fetal HSCs.8,12 However, the expression of many of these antigens declines on HSCs at later stages of development.3,4,13 It is interesting that the expression of CD34 is restored when adult HSCs are driven into cycle by 5-fluorouracil or granulocyte colony-stimulating factor administration.14,15 CD11b/Mac-1 is an adhesion molecule that is similarly dependent on developmental age and activation status.16 In contrast to these patterns, endomucin is a CD34-like sialomucin that marks HSCs from E10 and throughout subsequent development.17 Each of these advances offered the promise of learning more about how HSCs arise de novo and function throughout life.
We previously determined that the most primitive cells with lymphopoietic potential first develop in the para-aortic splanchnopleura (PSp)/aorta-gonad-mesonephros (AGM) region of embryos, and we tracked expression of the Rag1 lymphoid gene.18,19 To extend those findings, we searched for genes that might be differentially expressed at the very earliest stages of lymphopoiesis. We here show that endothelial cell-selective adhesion molecule (ESAM) is a durable and effective marker of HSCs. Indeed, ESAM was expressed throughout life and could be used as a gating parameter for sorting long-term repopulating HSCs.
Methods
Animals
Rag1/GFP knockin mice (CD45.2 alloantigen) were described.20,21 Mice of the corresponding wild-type (WT) C57BL/6 were obtained from Japan Clea (Shizuoka, Japan). Mating homozygous male Rag1/GFP knockin mice with WT C57BL/6 female mice generated heterozygous Rag1/GFP knockin fetuses. The day of vaginal plug observation was considered as day 0.5 postcoitum (E0.5). In some experiments, we purchased pregnant C57BL/6 mice from Japan Clea and used their fetuses. The congenic C57BL/6 strain (C57BL/6SJL; CD45.1 alloantigen) was purchased from The Jackson Laboratory (Bar Harbor, ME) and used in transplantation experiments. The experimental designs of this study were approved by the committee of Osaka University for animal studies.
Antibodies
Phycoerythrin (PE)–conjugated anti-Sca1 (Ly6A/E; D7), CD48 (HM48-1), CD11b/Mac-1 (M1/70), Gr-1 (RB6-8C5), CD19 (1D3), CD4 (L3T4), and CD8a (53-6.7) monoclonal antibodies (mAbs), biotinylated anti-CD45.2(104) mAb, allophycocyanin (APC)–conjugated anti-CD11b/Mac-1 (M1/70) and c-kit (2B8) mAbs, and PE-Texas red tandem-conjugated (PE-TR) streptavidin were purchased from BD Biosciences PharMingen (San Diego, CA). PE-conjugated anti-CD34 (RAM34), CD31/PECAM-1(390), CD105/Endoglin (MJ7/18), and Tie2 (TEK4) mAbs, PE-Cy7–conjugated anti-Sca1 (Ly6A/E; D7) mAb, and APC-conjugated anti CD45.1 (A20) mAbs were purchased from eBioscience (San Diego, CA). A rat anti–mouse ESAM mAb (1G8), a rabbit anti–mouse ESAM polyclonal Ab (VE19), and a rabbit preimmune IgG were prepared in our hands.22 A fluorescein isothiocyanate (FITC)–conjugated goat anti–rat IgG (H+L) Ab purchased from Southern Biotechnology (Birmingham, AL), a PE-conjugated goat antirat Ig Ab purchased from BD Biosciences PharMingen, or AlexaFluor 488 goat anti–rabbit IgG (H+L) Ab purchased from Invitrogen (Carlsbad, CA) was used as a second Ab for the anti-ESAM Abs. A PE-conjugated hamster IgG1 was purchased from BD Biosciences PharMingen and used as a control for a PE-conjugated anti-CD48. FITC-conjugated anti-CD11b/Mac-1 (M1/70), Gr-1 (RB6–8C5), TER-119, CD45R/B220 (RA3-6B2), and CD3e (145-2C11) were purchased from BD Biosciences PharMingen and used as lineage markers in adult studies.
Cell sorting
Fetal liver or cells obtained from adult femurs and tibias of heterozygous Rag1/GFP knockin mice were harvested and subjected to cell sorting as previously described.18 In the first step, Rag1− or Rag1Lo cells were sorted according to levels of green fluorescent protein (GFP) expression. Background auto-fluorescence was discriminated from authentic GFP by collecting data in 2 fluorescence channels without compensation. Under these conditions, even extremely low levels of fluorescence specific to GFP knockin mice were obvious on 2-color diagonal plots. The purity of the sorted cells in the first step was more than 95%. The sorted cells were incubated with anti-FcR (2.4G2) before staining with PE-anti-Sca1 and APC-anti-c-kit antibodies, suspended in 7-amino-actinomycin D (7-AAD)–containing buffer, and subjected to a second round of sorting. Dead cells stained with 7-AAD and a few contaminating cells with inappropriate GFP levels were gated out, and the cells were then fractionated according to Sca1 and c-kit to obtain Rag1− c-kitHi Sca1+ HSCs and Rag1Lo ckitHi Sca1+, early lymphoid progenitors (ELPs). The cell sorting was performed with FACSaria using the FACSDiva program (BD Biosciences, San Jose, CA). The sorting gates used to isolate HSCs and ELPs from E14.5 fetal liver for gene array experiments (“Gene arrays”) are shown in Figure S1 (available on the Blood website; see the Supplemental Materials link at the top of the online article).
In the sorting experiments with ESAM expression, Rag1/GFP− cells were first sorted from fetal tissues or adult bone marrow. Then the cells were stained with rat anti–mouse ESAM mAb (1G8) followed by FITC-goat anti–rat IgG or PE-goat anti–rat Ig, respectively. After the staining for ESAM, E14.5 fetal liver Rag1/GFP− cells were stained with PE–anti-Sca1 and APC-anti–c-kit antibodies. For PSp/AGM or yolk sac (YS) cells of E9.5-10.5 embryos, a PE-anti–Tie2 Ab was used instead of a PE-anti–Sca1 Ab. The adult marrow Rag1/GFP− cells were stained with FITC-anti-Lin (Mac-1, Gr-1, TER119, CD45R/B220, CD3e), PE-Cy7–anti-Sca1, and APC-anti–c-kit Abs. Then the cells were suspended in 7-AAD–containing buffer and subjected to a second round of sorting.
Gene arrays
Total RNAs were isolated from the Rag1− ckitHi Sca1+, HSC-enriched fraction and the Rag1Lo ckitHi Sca1+, ELP-enriched fraction, both of which were isolated from mouse E14.5 fetal liver. From the aliquot of each RNA (24 ng), biotin-labeled cRNA was prepared using GeneChip 2-Cycle Target Labeling and Control Reagents (Affymetrix, Santa Clara, CA) according to the manual. The cRNA was fragmented and hybridized to Mouse Genome 430 2.0 Array (Affymetrix) using a GeneChip Hybridization, Wash, and Stain Kit (Affymetrix). Fluorescent signals were scanned by GeneChip Scanner 3000, and the data were analyzed by GeneSpring GX software (Agilent Technologies, Palo Alto, CA). Bacillus subtilis genes on the arrays were used as negative controls for background subtraction. All values of the genes in each array were divided by the value of glyceraldehyde-3-phosphate dehydrogenase (Gapdh) gene. The Cross-Gene Error Model was used to estimate measurement precision by combining variability of gene expression data. The experiments were duplicated to confirm reproducibility of the data. All microarray data have been deposited with CIBEX, National Institute of Genetics DDBJ (DNA Data Bank of Japan) under the accession number CBX73.
Flow cytometry
Rag1/GFP− cells of E14.5 fetal liver were incubated with rat anti–mouse ESAM mAb (1G8) followed by FITC-goat anti–rat IgG. Then the cells were incubated with a rat anti–mouse FcRII/III (2.4G2) and subsequently stained with PE-Cy7–anti-Sca1 and APC-anti–c-kit in combination with PE-anti–CD34, CD31/PECAM1, CD105/Endoglin, or Tie2 mAbs. The cells were suspended in 7-AAD–containing buffer and subjected to flow cytometry analyses, performed with FACSaria using the FACSDiva program (BD Biosciences).
In the other experiments, cultured cells or recovered cells from transplanted mice were incubated with anti-FcR and then stained with PE-, APC-conjugated mAbs and/or a biotinylated Ab followed by PE-TR-streptavidin as indicated in each figure. The flow cytometry analyses were performed with FACScalibur using the Cellquest program (BD Biosciences). The data analyses were done with FlowJo software (TreeStar, Ashland, OR).
Methylcellulose culture
A total of 500 or 100 cells of each sorted fraction (Rag1/GFP− ckitHi Sca1+ ESAM1−/Lo or Rag1/GFP− ckitHi Sca1+ ESAM1Hi of fetal liver or adult marrow, respectively) were cultured in Iscove modified Dulbecco medium-based methylcellulose medium supplemented with 50 ng/mL of recombinant mouse (rm) stem cell factor (SCF), 10 ng/mL of rm interleukin-3 (IL-3), 10 ng/mL of recombinant human IL-6, and 3 U/mL of recombinant human erythropoietin (Methocult GF 3434; StemCell Technologies, Vancouver, BC). After 8 to 10 days, colonies were enumerated and classified as granulocyte colony-forming units (CFU-G); macrophage colony-forming units (CFU-M); granulocyte-macrophage colony-forming units (CFU-GM); erythroid burst-forming units (BFU-E); or mixed erythroid-myeloid colony-forming units (CFU-Mix) according to shape and color under an inverted microscope. In some experiments, colony types were morphologically certified by examination of cytospin preparations after May-Grünwald/Giemsa staining.
Stromal coculture and limiting dilution assays
The murine bone marrow stromal cell line MS-5 was generously provided by Dr J. Mori (Niigata University, Niigata, Japan). Nonirradiated MS-5 stromal cells were prepared at a concentration of 5 × 104 cells/well in 24-well tissue plates 1 day before the seeding of sorted cells at a concentration of 50 cells/mL. Cells were cultured in α-minimum essential medium (Invitrogen) supplemented with 10% fetal calf serum (FCS), rm SCF (10 ng/mL), rm Flt3-ligand (20 ng/mL), and rm IL-7 (1 ng/mL). The cultures were fed every 4 days by removing half of the medium and replacing it with fresh medium, and maintained for 6 or 10 days. Cytokines were freshly added with each feeding.
The frequencies of lymphohematopoietic progenitors in the fetal liver–derived Rag1/GFP− ckitHi Sca1+ ESAM−/Lo or Rag1/GFP− ckitHi Sca1+ ESAMHi fraction were determined by plating the sorted cells in limiting dilution assays using 96-well flat bottom plates. Pre-established MS-5 stromal cell layers were plated with 1, 2, 4, 8, or 50 cells each using the Automated Cell Deposition Unit of the FACSaria (BD Biosciences). Cells were cultured in α-minimum essential medium supplemented with 10% FCS, rm SCF (10 ng/mL), rm Flt3-ligand (20 ng/mL), and rm IL-7 (1 ng/mL). At 10 days of culture, wells were inspected for the presence of hematopoietic clones. Positive wells were harvested, stained, and analyzed by flow cytometry for the presence of CD19+ Gr-1− B lineage cells. The frequencies of progenitors were calculated by linear regression analysis on the basis of Poisson distribution as the reciprocal of the concentration of test cells that gave 37% negative cultures.
Competitive repopulation assay
The Ly5 system was adapted to a competitive repopulation assay. A total of 1000 Rag1/GFP− c-kitHi Sca1+ ESAM−/Lo or Rag1/GFP− c-kitHi Sca1+ ESAMHi cells sorted from fetal liver of E14.5 Rag1/GFP heterozygous embryos (CD45.2) were mixed with 2 × 105 unfractionated adult bone marrow cells obtained from WT C57BL/6-Ly5.1 (CD45.1) mice and were transplanted into C57BL/6-Ly5.1 mice irradiated at a dose of 10 Gy. At 5 weeks after transplantation, peripheral blood cells of the recipients were colleted by retro-orbital bleeding and were stained with APC-conjugated anti-CD45.1 and biotinylated anti-CD45.2 Abs followed by PE-TR streptavidin. The cells were simultaneously stained with PE-conjugated anti–Mac-1 and Gr-1 Abs, or PE-conjugated anti-CD19 Ab, or PE-conjugated anti-CD4 and CD8a Abs. Twenty weeks after transplantation, all recipients were killed, and reconstitution of CD45.2+ myeloid, B, or T cells was confirmed by flow cytometry in the bone marrow, spleen, and thymus, respectively. For the second transplantation, bone marrow cells of primary recipients with CD45.2 engraftment were transferred into 10 Gy-irradiated C57BL/6-Ly5.1 mice (106 whole bone marrow cells per recipient). After 20 weeks, contribution of CD45.2 cells to the hematopoietic reconstitution was evaluated in the second recipients.
Cell preparation from PSp/AGM and YS
To examine the phenotype or function of hematopoietic progenitors in the PSp/AGM and the YS of early embryos, cells were prepared as previously described.19 Briefly, tissues were dissociated by incubation with dispase II (Roche Diagnostics, Mannheim, Germany) for 20 minutes at 37°C and cell dissociation buffer (Invitrogen) for 20 minutes at 37°C followed by vigorous pipetting. The cells were then suspended in phosphate-buffered saline containing 3% FCS and stained with the Abs indicated in each experiment.
Statistical methods
Statistical analysis was carried out by standard Student t tests. Error bars used throughout indicate SD of the mean.
Results
Identification of ESAM as a marker of primitive hematopoietic cells
Microarrays were performed as part of an effort to characterize the initial transition of fetal HSCs to primitive lymphopoietic cells. The comparisons involved mRNA from Rag1Lo ckitHi Sca1+, ELPs, and the HSC-enriched Rag1− ckitHi Sca1+ fraction isolated from E14.5 fetal liver (Figure S1).
Consistent with previous analyses,12,23,24 Evi1, CD41, and Endoglin were highly expressed by fetal HSCs (Figure 1). Esam transcripts were conspicuous in the HSC fraction and attracted attention because of a sharp down-regulation on differentiation to ELP (Figure 1). Real-time PCR using Esam specific primers verified the results of these microarrays (data not shown).
Esam gene is preferentially expressed in the HSC fraction of E14.5 fetal liver. Microarray analyses comparing HSC-enriched and ELP-enriched populations were performed. Two independent tests depicted the Esam gene as preferentially expressed in the HSC-enriched population. Results are shown as relative expression levels of each gene comparing with that of Gapdh of which value is 100. Ratio was calculated by [HSC level]/[ELP level] in each gene. The relative expression levels of c-kit were also shown as internal quality controls.
Esam gene is preferentially expressed in the HSC fraction of E14.5 fetal liver. Microarray analyses comparing HSC-enriched and ELP-enriched populations were performed. Two independent tests depicted the Esam gene as preferentially expressed in the HSC-enriched population. Results are shown as relative expression levels of each gene comparing with that of Gapdh of which value is 100. Ratio was calculated by [HSC level]/[ELP level] in each gene. The relative expression levels of c-kit were also shown as internal quality controls.
ESAM can be used to subdivide hematopoietic progenitors in fetal liver
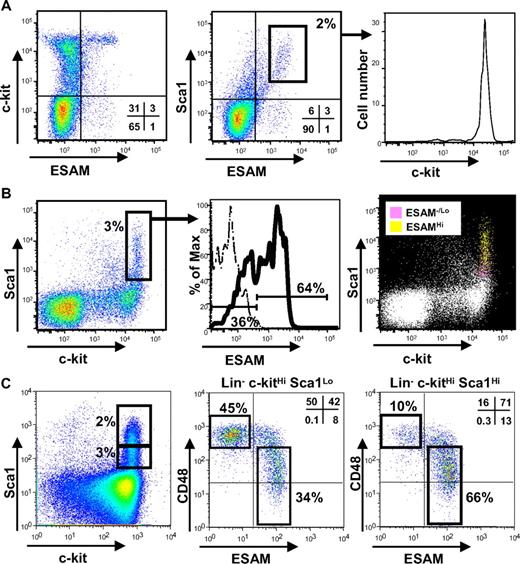
The availability of a rat antimouse ESAM mAb (clone 1G8) facilitated characterization of mononuclear cells obtained from E14.5 fetal liver. A majority of ESAM+ cells were found in the c-kitHi fraction (Figure 2A left). ESAM expression also correlated with Sca1 (Figure 2A middle). These observations suggested that ESAM might substitute for at least one of the 2 most widely used HSC markers. Indeed, cells in the Sca1Hi ESAMHi gate were almost homogeneous with respect to high c-kit expression (Figure 2A right).
Specific expression of ESAM on HSC-enriched fraction of E14.5 fetal liver. Flow cytometric analysis was performed for Rag1/GFP− cells of E14.5 fetal liver using anti-c-kit, anti-Sca1, and anti-ESAM Abs. First, Rag1/GFP− cells were sorted from E14.5 fetal liver of Rag1/GFP knockin heterozygous fetuses with high purity. The sorted cells were incubated with a purified rat anti–mouse ESAM Ab (1G8) followed by goat anti–rat IgG-FITC. The cells were then stained with anti-c-kit-APC, anti-Sca1-PE, and 7-AAD. To minimize the nonspecific binding of anti-c-kit and Sca1 mAbs to the cells wearing goat anti–rat IgG-FITC, the cells were incubated with a rat anti–mouse FcRII/III Ab before the anti-c-kit and Sca1 staining. (A) The Rag1/GFP− cells were analyzed with respect to expression of ESAM, c-kit, and Sca1 (Left, middle). Expression of c-kit in the Sca1Hi ESAMHi cells (middle, inset) is presented (right). (B) The conventional c-kitHi Sca1+ fraction (left, inset) could be divided into 2 fractions, ESAM−/Lo and ESAMHi (middle). The cells were stained with an isotype control IgG (dashed line) or with the anti-ESAM Ab (solid line). The ESAMHi cells (yellow) were found as c-kitHi Sca1Hi, whereas the ESAM−/Lo cells (pink) were c-kitHi Sca1Lo (right). (C) Six-color flow cytometric analysis using an anti-ESAM Ab followed by goat anti–rat IgG-FITC, a PE-anti-CD48 Ab, biotin-anti-lineage marker Abs (TER119, Gr1, CD3, CD45R/B220) followed by SA-PETR, a PE-Cy7-anti-Sca1 Ab, an APC-anti-c-kit, and 7-AAD was performed for E14.5 fetal liver cells of WT C57B6 embryos. The profile of Lin− cells regarding c-kit and Sca1 expression is shown in the left. The Lin− c-kitHi Sca1Lo and Lin− c-kitHi Sca1Hi fractions gated in the left panel were analyzed with respect to expression of ESAM and CD48 (middle and right). The percentage of cells in each gate is indicated in each panel.
Specific expression of ESAM on HSC-enriched fraction of E14.5 fetal liver. Flow cytometric analysis was performed for Rag1/GFP− cells of E14.5 fetal liver using anti-c-kit, anti-Sca1, and anti-ESAM Abs. First, Rag1/GFP− cells were sorted from E14.5 fetal liver of Rag1/GFP knockin heterozygous fetuses with high purity. The sorted cells were incubated with a purified rat anti–mouse ESAM Ab (1G8) followed by goat anti–rat IgG-FITC. The cells were then stained with anti-c-kit-APC, anti-Sca1-PE, and 7-AAD. To minimize the nonspecific binding of anti-c-kit and Sca1 mAbs to the cells wearing goat anti–rat IgG-FITC, the cells were incubated with a rat anti–mouse FcRII/III Ab before the anti-c-kit and Sca1 staining. (A) The Rag1/GFP− cells were analyzed with respect to expression of ESAM, c-kit, and Sca1 (Left, middle). Expression of c-kit in the Sca1Hi ESAMHi cells (middle, inset) is presented (right). (B) The conventional c-kitHi Sca1+ fraction (left, inset) could be divided into 2 fractions, ESAM−/Lo and ESAMHi (middle). The cells were stained with an isotype control IgG (dashed line) or with the anti-ESAM Ab (solid line). The ESAMHi cells (yellow) were found as c-kitHi Sca1Hi, whereas the ESAM−/Lo cells (pink) were c-kitHi Sca1Lo (right). (C) Six-color flow cytometric analysis using an anti-ESAM Ab followed by goat anti–rat IgG-FITC, a PE-anti-CD48 Ab, biotin-anti-lineage marker Abs (TER119, Gr1, CD3, CD45R/B220) followed by SA-PETR, a PE-Cy7-anti-Sca1 Ab, an APC-anti-c-kit, and 7-AAD was performed for E14.5 fetal liver cells of WT C57B6 embryos. The profile of Lin− cells regarding c-kit and Sca1 expression is shown in the left. The Lin− c-kitHi Sca1Lo and Lin− c-kitHi Sca1Hi fractions gated in the left panel were analyzed with respect to expression of ESAM and CD48 (middle and right). The percentage of cells in each gate is indicated in each panel.
The c-kitHi Sca1+ fraction, known to include all conventional HSCs, was divided into 2 categories according to ESAM staining (Figure 2B left and middle). The subpopulation with the highest density of ESAM was enriched for c-kitHi Sca1Hi cells, whereas ones with negative or low levels of ESAM were found in the c-kitHi Sca1Lo subset (Figure 2B right). The latter are thought to be enriched with respect to committed myelo-erythroid progenitors.25 Similar results were obtained with a rabbit antimouse ESAM polyclonal Ab (VE19) (data not shown).
When E14.5 fetal liver cells of WT C57B6 mice and traditional markers of fetal liver HSCs were used instead of the Rag1/GFP knockin mice, essentially identical results were obtained (Figure 2C). This analysis was performed with CD48 expression, one of the SLAM family members recently shown to conversely relate with HSC activity in fetal liver.26 Lin− c-kitHi Sca1+ cells could be divided into ESAM− and ESAM+ fractions, and a subpopulation with higher Sca1 expression was more enriched with ESAM+ cells (Figure 2C middle and right). CD48 expression tended to increase along with down-regulation of ESAM. Indeed, the Lin− c-kitHi Sca1+ fraction was found to consist of 2 major subpopulations, CD48Hi ESAM− and CD48−/Lo ESAM+. Thus, ESAM is conspicuously expressed on immature hematopoietic cells in fetal liver and seems to conversely relate to lineage progression.
ESAM expression is closely associated with fetal HSCs among endothelial markers
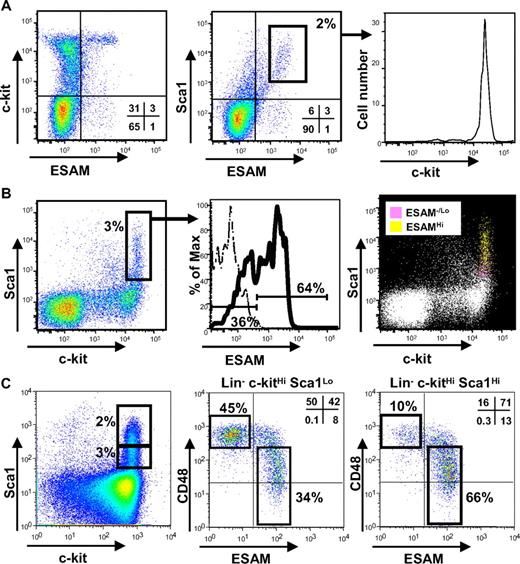
Next we evaluated how ESAM corresponds to other endothelial antigens that were previously identified on fetal hematopoietic progenitors. CD34 and CD31/PECAM1 were uniformly present on Rag1− c-kitHi Sca1+ cells in E14.5 fetal liver (Figure 3A,B). Neither could resolve the Rag1− c-kitHi Sca1+ cells into ESAMHi and ESAM−/Lo fractions, and even showed a slightly inverse relationship with ESAM expression on early progenitors. Expression profiles of Endoglin and Tie2 did correlate with ESAM (Figure 3C,D). However, although the primitive ESAMHi fraction uniformly expressed high levels of Endoglin and Tie2, many of the more differentiated ESAM−/Lo cells still retained the 2 markers. These results suggest that ESAM might be a more useful marker of HSCs than other endothelial antigens and could represent an important tool for fetal stem cell studies.
Expression of ESAM and other endothelial antigens on fetal liver HSC fraction. The expression pattern of ESAM on Rag1/GFP− c-kitHi Sca1+ cells of E14.5 fetal liver was compared with other endothelial cell-related antigens, CD34 (A), CD31/PECAM1 (B), Endoglin (C), and Tie2 (D). The percentage of cells in subpopulation is shown. In the histograms, the staining patterns of ESAM−/Lo cells are indicated with dashed lines; those of ESAMHi cells are tinted and indicated with solid lines.
Expression of ESAM and other endothelial antigens on fetal liver HSC fraction. The expression pattern of ESAM on Rag1/GFP− c-kitHi Sca1+ cells of E14.5 fetal liver was compared with other endothelial cell-related antigens, CD34 (A), CD31/PECAM1 (B), Endoglin (C), and Tie2 (D). The percentage of cells in subpopulation is shown. In the histograms, the staining patterns of ESAM−/Lo cells are indicated with dashed lines; those of ESAMHi cells are tinted and indicated with solid lines.
ESAM is useful to enrich primitive multipotent progenitors from fetal liver
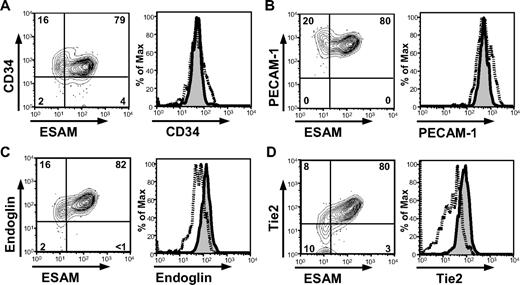
To evaluate whether ESAM expression can enrich primitive progenitors in fetuses, we compared the clonogenic potential in methylcellulose cultures of the ESAM−/Lo and ESAMHi cells in the Rag1− ckitHi Sca1+ HSC fraction of E14.5 fetal liver. Cells in the ESAMHi fraction formed more colonies with larger size than those in the ESAM−/Lo fraction (Figure 4). In particular, most CFU-Mix, primitive progenitors with both myeloid and erythroid potential, were found in the ESAMHi fraction (Figure 4A).
ESAM expression correlates with CFU activity. ESAM−/Lo or ESAMHi cells of the Rag1/GFP− ckitHi Sca1+ fraction of E14.5 fetal liver were sorted and subjected to methylcellulose colony formation assay. Numbers of CFUs (A) and morphology of the colonies derived from the indicated CFUs (B) are shown. The results in panel A are shown as mean plus or minus SD. A black bar in panel B represents 500 mm. The data are from one of 3 independent experiments that gave similar results. Significant difference between the 2 population is indicated (**P < .01).
ESAM expression correlates with CFU activity. ESAM−/Lo or ESAMHi cells of the Rag1/GFP− ckitHi Sca1+ fraction of E14.5 fetal liver were sorted and subjected to methylcellulose colony formation assay. Numbers of CFUs (A) and morphology of the colonies derived from the indicated CFUs (B) are shown. The results in panel A are shown as mean plus or minus SD. A black bar in panel B represents 500 mm. The data are from one of 3 independent experiments that gave similar results. Significant difference between the 2 population is indicated (**P < .01).
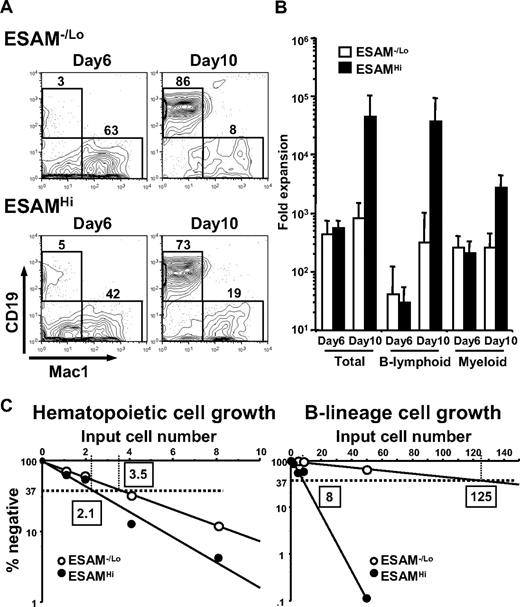
Next we analyzed the lymphopoietic potential of cells resolved on the basis of ESAM in cocultures with the MS5 bone marrow stromal cell line. The culture media contained SCF, Flt3-ligand, and IL-7, factors that effectively generate CD19+ B lymphoid cells as well as Mac1+ myeloid cells.18 When 500 cells were cultured in individual wells of 6-well plates, both ESAM−/Lo and ESAMHi fractions produced CD19+ cells and Mac1+ cells (Figure 5A). However, we observed that most of the hematopoietic colonies from ESAMHi cells grew beneath the MS5 stromal cell layer, although this was not the case with ESAM−/Lo cells (data not shown). Moreover, the lymphohematopoietic cells from ESAMHi cells continued to expand explosively after 6 days of coculture and gave rise to approximately 50 000 cells per input progenitor by day 10 (Figure 5B).
ESAM expression enriches primitive progenitors endowed with lymphopoietic activity. (A,B) A total of 500 ESAM−/Lo or ESAMHi cells of the Rag1/GFP− ckitHi Sca1+ fraction of E14.5 fetal liver were cultured with MS5 stromal cells in the presence of SCF, Flt3-ligand, and IL-7. At the indicated period, recovered cells were counted and subjected to flow cytometry (A). Yields of total cells, CD19+ Mac1− B-lineage cells, and Mac1+ CD19− myeloid-lineage cells per 1 input ESAM−/Lo or ESAMHi progenitor were calculated and given as averages with SD bars (B). (C) Limiting dilution analyses were performed in the MS5 coculture system to determine the frequency of hematopoietic progenitors (left) and that of progenitors endowed with lymphopoietic potential (right).
ESAM expression enriches primitive progenitors endowed with lymphopoietic activity. (A,B) A total of 500 ESAM−/Lo or ESAMHi cells of the Rag1/GFP− ckitHi Sca1+ fraction of E14.5 fetal liver were cultured with MS5 stromal cells in the presence of SCF, Flt3-ligand, and IL-7. At the indicated period, recovered cells were counted and subjected to flow cytometry (A). Yields of total cells, CD19+ Mac1− B-lineage cells, and Mac1+ CD19− myeloid-lineage cells per 1 input ESAM−/Lo or ESAMHi progenitor were calculated and given as averages with SD bars (B). (C) Limiting dilution analyses were performed in the MS5 coculture system to determine the frequency of hematopoietic progenitors (left) and that of progenitors endowed with lymphopoietic potential (right).
Authentic HSCs are characterized as having both lymphoid and myeloid potential.8,27 To compare numbers of primitive lymphohematopoietic progenitors in the ESAMHi and ESAM−/Lo fractions, we performed in vitro limiting dilution assays in MS5 cocultures. One in 2.1 ESAMHi cells and 1 in 3.5 ESAM−/Lo cells gave rise to blood cells, indicating that both fractions are extremely potent sources of hematopoietic progenitors (Figure 5C left). However, we observed drastic differences between the 2 fractions regarding the frequencies of primitive progenitors with lymphopoietic potential. Whereas 1 in 8 ESAMHi cells produced CD19+ B lineage cells, only 1 in 125 ESAM−/Lo cells were lymphopoietic under these conditions (Figure 5C right). These results suggest that primitive stem/progenitor cells, which are multipotent for myeloid, erythroid, and lymphoid lineages, are present in the ESAMHi fraction of fetal liver.
Long-term reconstitution activity is exclusive to ESAMHi cells
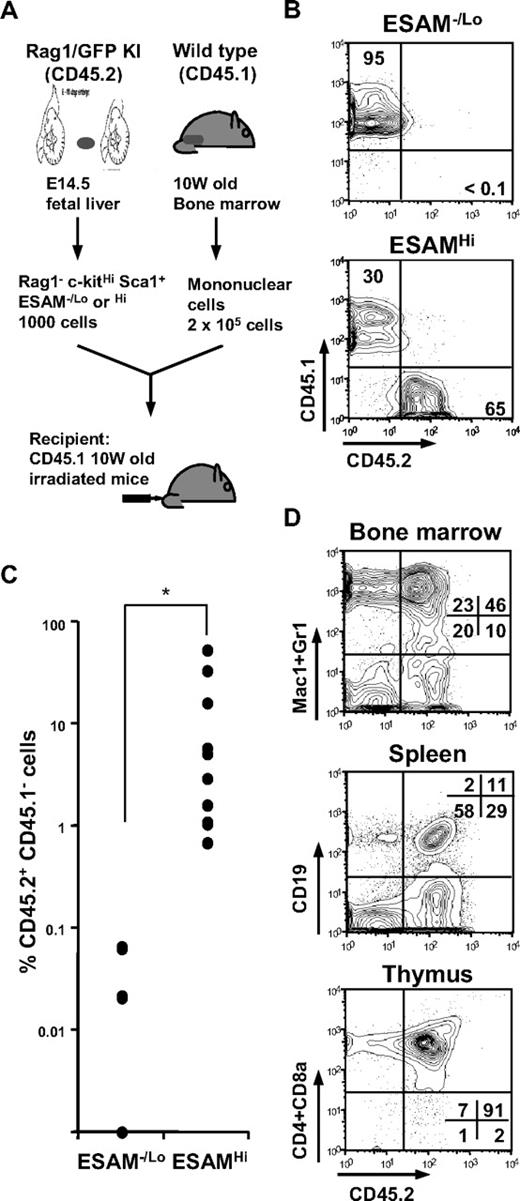
To evaluate ESAM expression on long-term reconstituting HSCs in E14.5 fetal liver, we transplanted cells of the ESAM−/Lo or ESAMHi fraction into lethally irradiated mice (Figure 6A). Five weeks after transplantation, it was obvious that CD45.2+ ESAMHi cells contributed highly to the recovery of hematopoiesis in recipients, but no chimerism was detected in mice transplanted with ESAM−/Lo cells (Figure 6B). Indeed, although 9 of 11 mice transplanted with 1000 ESAMHi cells had clear donor CD45.2+ populations (> 1.0%) among peripheral leukocytes, none of 10 mice given 1000 ESAM−/Lo cells had evidence of chimerism. Five months after transplantation, we analyzed the contribution of CD45.2+ cells to long-term lymphohematopoietic reconstitution of the recipients. Although most of the 11 mice transplanted with ESAMHi cells had clear CD45.2+ populations in bone marrow (mean ± SD of percentage CD45.2+ CD45.1− in total CD45+ cells; 16.3% ± 22.0%), none of the ESAM−/Lo-transplanted mice contained detectable CD45.2+ cells in that organ (mean ± SD of percentage CD45.2+ CD45.1− in total CD45+ cells; 0.02% ± 0.03%) (Figure 6C). Furthermore, multilineage recovery was observed in the bone marrow, spleen, and thymus of mice transplanted with ESAMHi cells (Figure 6D). In addition, bone marrow cells recovered from primary recipients with ESAMHi cell transplantation effectively reconstituted CD45.2+ lymphohematopoietic cells in secondary recipients at 5 months after transplantation (data not shown). These results indicate that high levels of ESAM expression correspond to fetal HSCs with long-term repopulating potential.
Long-term hematopoiesis-reconstituting activity is excusive to ESAMHi fraction. (A) The Rag1/GFP− Sca1+ c-kitHi cells (CD45.2+) of E14.5 fetal liver were sorted into 2 fractions, ESAM−/Lo and ESAMHi. Then, 1000 cells of each fraction were mixed with 2 × 105 CD45.1+ whole adult bone marrow cells of 10-week-old mice and transplanted to a lethally irradiated CD45.1 mouse (A). (B) Flow cytometry analyses for peripheral leukocytes were performed at 5 weeks after transplantation. In the 2 independent experiments, 9 of 11 recipients with ESAMHi cells were clearly reconstituted by CD45.2+ cells (> 1.0% in all of myeloid, T, and B lineages), whereas none of 11 recipients with ESAM−/Lo cells had CD45.2+ cells detectable in the flow cytometry. The figure shows representative results in each group. (C,D) Twenty weeks after transplantation, all the recipients were killed, and the contribution of CD45.2+ ESAMHi cells was evaluated in lymphohematopoietic organs. Percentages of CD45.2+ CD45.1− population among total CD45+ cells in bone marrow of each recipient were plotted (C). The long-term reconstitution of CD45.2+ ESAMHi cells was confirmed with respect to myeloid, B lymphoid, or T lymphoid lineages in the bone marrow, spleen, and thymus, respectively (D).
Long-term hematopoiesis-reconstituting activity is excusive to ESAMHi fraction. (A) The Rag1/GFP− Sca1+ c-kitHi cells (CD45.2+) of E14.5 fetal liver were sorted into 2 fractions, ESAM−/Lo and ESAMHi. Then, 1000 cells of each fraction were mixed with 2 × 105 CD45.1+ whole adult bone marrow cells of 10-week-old mice and transplanted to a lethally irradiated CD45.1 mouse (A). (B) Flow cytometry analyses for peripheral leukocytes were performed at 5 weeks after transplantation. In the 2 independent experiments, 9 of 11 recipients with ESAMHi cells were clearly reconstituted by CD45.2+ cells (> 1.0% in all of myeloid, T, and B lineages), whereas none of 11 recipients with ESAM−/Lo cells had CD45.2+ cells detectable in the flow cytometry. The figure shows representative results in each group. (C,D) Twenty weeks after transplantation, all the recipients were killed, and the contribution of CD45.2+ ESAMHi cells was evaluated in lymphohematopoietic organs. Percentages of CD45.2+ CD45.1− population among total CD45+ cells in bone marrow of each recipient were plotted (C). The long-term reconstitution of CD45.2+ ESAMHi cells was confirmed with respect to myeloid, B lymphoid, or T lymphoid lineages in the bone marrow, spleen, and thymus, respectively (D).
ESAM expression marks cells thought to represent HSCs in fetal and aged adult tissues
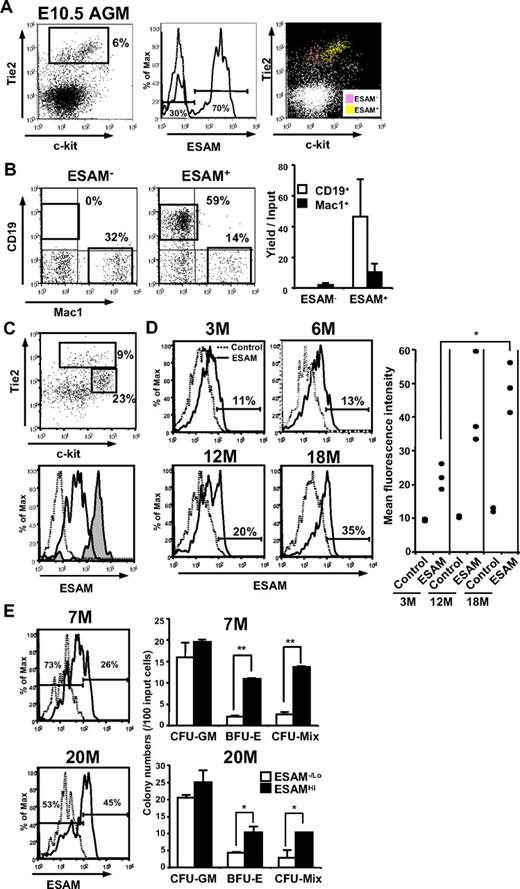
The definitive HSCs that account for lymphohematopoieisis in adults first arise in the AGM region.28,29 To examine whether ESAM was present on those HSCs, flow cytometry analyses were performed with the Rag1/GFP− cells isolated from E10.5 embryos. Tie2 and c-kit were used as additional parameters because we previously identified lymphohematopoietic cells in the early embryos as Rag1− Tie2+ c-kit+.19 A small but conspicuous Tie2Hi population was detected in E10.5 AGM cells (Figure 7A left). Interestingly, the Tie2Hi AGM cells were clearly divided into 2 discrete populations according to ESAM expression (Figure 7A middle). When the 2 populations were back-plotted on the Tie2 and c-kit profile, ESAM+ cells were exclusively c-kit+ (Figure 7A right). In addition, lymphohematopoietic potential in the MS5 cocultures was highly enriched in the ESAM+ fraction among the Tie2Hi cells. That is, the Tie2Hi ESAM+ fraction could effectively produce both CD19+ lymphoid cells and Mac1+ myeloid cells, although the Tie2Hi ESAM− fraction generated only a small number of Mac1+ cells (Figure 7B). We concluded from these analyses that ESAM marks the primitive hematopoietic cells endowed with lymphopoietic activity in the E10.5 AGM as well as in the E14.5 liver.
ESAM marks early hematopoietic progenitors throughout life. (A) Rag1/GFP− cells were sorted from AGM of E10.5 Rag1/GFP knockin heterozygous fetuses with high purity. The sorted Rag1/GFP− cells were incubated with the anti-ESAM Ab (1G8) followed by goat antirat IgG-FITC. The cells were then stained with anti-c-kit-APC, anti-Tie2-PE, and 7-AAD. The profile of Tie2 and c-kit expression in AGM cells was shown in a left panel. ESAM expression (solid line) in the Tie2Hi fraction of AGM and its control level with an isotype-matched IgG (dashed line) are presented (middle). The Tie2Hi ESAM+ cells (yellow) were c-kit+, whereas the Tie2Hi ESAM− cells (pink) were c-kit− (right). (B) The Tie2Hi ESAM− and Tie2Hi ESAM+ cells were sorted from E10.5 AGM and cultured on MS5 for 10 days. The recovered cells were counted and stained for the markers, including CD19 and Mac1. (C) Rag1/GFP− cells sorted from E10.5 YS were stained in the same manner as for the E10.5 AGM cells described in panel A. The profile of Tie2 and c-kit expression is shown in the top panel. In the lower panel, ESAM expression in the Tie2Hi c-kitLo fraction (tinted histogram) and the Tie2Lo c-kitHi fraction (open histogram) is shown. A dashed line shows the background fluorescence with an isotype-matched IgG. (D) ESAM expression of Rag1/GFP− Lin− ckitHi Sca1+ cells of adult bone marrow of 3-, 6-, 12-, and 18-month-old mice was analyzed. Representative flow cytometry results in 3 to 5 mice of each age were shown. ESAM expression on Rag1/GFP− Lin− c-kitHi Sca1+ cells of adult bone marrow of 3-, 12-, and 18-month-old mice (n = 3 in each) were simultaneously analyzed, and the data were summarized with respect to the mean fluorescence intensity with an anti-ESAM Ab or its control Ab (right panel). (E) ESAM−/Lo or ESAMHi cells were sorted from the Rag1/GFP− Lin− ckitHi Sca1+ fraction of the indicated adult bone marrow (7 and 20 months old, respectively) and subjected to methylcellulose colony assay. The data are from one of 2 independent experiments that gave similar results. Statistical significance: *P < .05, **P < .01. The percentages of cells in each gate are indicated in each panel.
ESAM marks early hematopoietic progenitors throughout life. (A) Rag1/GFP− cells were sorted from AGM of E10.5 Rag1/GFP knockin heterozygous fetuses with high purity. The sorted Rag1/GFP− cells were incubated with the anti-ESAM Ab (1G8) followed by goat antirat IgG-FITC. The cells were then stained with anti-c-kit-APC, anti-Tie2-PE, and 7-AAD. The profile of Tie2 and c-kit expression in AGM cells was shown in a left panel. ESAM expression (solid line) in the Tie2Hi fraction of AGM and its control level with an isotype-matched IgG (dashed line) are presented (middle). The Tie2Hi ESAM+ cells (yellow) were c-kit+, whereas the Tie2Hi ESAM− cells (pink) were c-kit− (right). (B) The Tie2Hi ESAM− and Tie2Hi ESAM+ cells were sorted from E10.5 AGM and cultured on MS5 for 10 days. The recovered cells were counted and stained for the markers, including CD19 and Mac1. (C) Rag1/GFP− cells sorted from E10.5 YS were stained in the same manner as for the E10.5 AGM cells described in panel A. The profile of Tie2 and c-kit expression is shown in the top panel. In the lower panel, ESAM expression in the Tie2Hi c-kitLo fraction (tinted histogram) and the Tie2Lo c-kitHi fraction (open histogram) is shown. A dashed line shows the background fluorescence with an isotype-matched IgG. (D) ESAM expression of Rag1/GFP− Lin− ckitHi Sca1+ cells of adult bone marrow of 3-, 6-, 12-, and 18-month-old mice was analyzed. Representative flow cytometry results in 3 to 5 mice of each age were shown. ESAM expression on Rag1/GFP− Lin− c-kitHi Sca1+ cells of adult bone marrow of 3-, 12-, and 18-month-old mice (n = 3 in each) were simultaneously analyzed, and the data were summarized with respect to the mean fluorescence intensity with an anti-ESAM Ab or its control Ab (right panel). (E) ESAM−/Lo or ESAMHi cells were sorted from the Rag1/GFP− Lin− ckitHi Sca1+ fraction of the indicated adult bone marrow (7 and 20 months old, respectively) and subjected to methylcellulose colony assay. The data are from one of 2 independent experiments that gave similar results. Statistical significance: *P < .05, **P < .01. The percentages of cells in each gate are indicated in each panel.
A small number of Tie2Hi c-kitLo ESAMHi cells were also observed among extraembryonic YS cells (Figure 7C). In addition, the E10.5 YS contained a conspicuous population whose phenotype was Tie2Lo c-kitHi, and cells in that fraction expressed low levels of ESAM (Figure 7C). Cells with the same phenotype were also clearly observed in the E9.5 YS, although they were absent in the caudal half of the embryo proper (Figure S2A). These Tie2Lo c-kitHi ESAMLo cells in the YS effectively produced myeloid and/or erythroid colonies in methylcellulose culture but showed little lymphopoietic potential (Figure S2B,C). Importantly, lymphopoietic activity was exclusive to the Tie2Hi c-kitLo ESAMHi fraction of the PSp/AGM region, which showed no myeloid-erythroid potential in conventional methylcellulose assays.
It is well known that HSC properties change with developmental age. Indeed, Forsberg et al previously reported ESAM expression by adult marrow HSCs,30 but the levels were weak compared with those on fetal HSCs. Therefore, we expected that ESAM, like the other endothelial antigens, would decline with HSC aging. However, we found that ESAM expression was detectable on the Rag1− LSK fraction of bone marrow through life (Figure 7D). Indeed, the proportion of ESAMHi cells and the mean fluorescence intensity of ESAM expression in the Rag1− LSK fraction increased with age (Figure 7D). Moreover, primitive myeloid-erythroid progenitors from adult bone marrow were also enriched in the ESAMHi fraction (Figure 7E). In summary, these findings demonstrate that ESAM expression is useful for exploring the biology of hematopoietic stem/progenitor cells throughout life in mice.
Discussion
We conducted gene array analyses with the principal goal of learning more about the initial differentiation of fetal HSCs to cells in lymphoid lineages. In that regard, several informative genes were identified and will be presented elsewhere (manuscript in preparation). Our screen also identified genes whose expression was previously thought to correlate with HSCs in fetal or adult tissues. Among those, Esam was particularly noteworthy as being drastically down-regulated during differentiation of HSCs to ELPs. We now report that it represents a potent tool for identifying HSCs over a wide range of developmental age.
ESAM was originally identified as an endothelial cell-specific protein, although it was also shown to be expressed on megakaryocytes and platelets.22,31 A recent study also found ESAM transcripts in adult marrow HSCs when gene arrays were used to compare Thy1.1Lo Flk2− LSK (long-term HSC-enriched), Thy1.1Lo Flk2+ LSK (short-term HSCs), and Thy1.1− Flk2+ LSK (multiple progenitors).30 We now show how ESAM levels can be exploited to obtain highly enriched CFU-Mix, lymphopoietic cells, and long-term HSCs from the Rag1− c-kitHi Sca1+ fraction of E14.5 fetal liver. It is important to note that long-term HSCs in mouse embryos are unique in expressing markers, such as Flk2 and CD11b/Mac1, not characteristic of adult HSCs.18,32 This complicates cell sorting strategies and fetal/adult comparison. We previously found that Rag1/GFP− c-kitHi Sca1+ cells derived from E14.5 fetal liver reconstituted lymphohematopoiesis in lethally irradiated adults, whereas Rag1/GFPLo c-kitHi Sca1+ cells transiently contributed to T and B lymphopoiesis. ESAM specific Abs can be used without knockin reporters and with fewer combinations of Abs to obtain highly enriched HSCs.
Although we identified Esam as one of the highly expressed genes in HSC but not in ELP, levels of this antigen strongly correlated with lymphopoietic activity. The ESAMHi fraction in Rag1− c-kitHi Sca1+ cells of E14.5 fetal liver produced CD19+ B-lineage cells more effectively and contained lymphopoietic progenitors with even higher frequency than the ESAM−/Lo fraction. The cells also gave rise to long-term reconstitution in both T and B lymphoid lineages in lethally irradiated recipients. Furthermore, in the PSp/AGM region, B lymphopoietic activity in culture was exclusive to the ESAMHi Tie2+ fraction. From these observations, we conclude that ESAM expression indicates high lymphopoietic potential in HSCs of the fetal liver and hematopoietic cells arising in the AGM region. On the other hand, we presume from the sharp down-regulation in ELPs that ESAM is not necessary for early lymphoid differentiation.
We previously reported that the first lymphopoietic cells arise in a Tie2+ c-kit+ CD34−/Lo CD41− subset in the E8.5 PSp region.19 In this report, we showed clear ESAM expression on lymphopoietic cells contained in a Tie2Hi c-kit+ subset of the E9.5 to E10.5 PSp/AGM. Lymphopoietic activity in early embryos is thought to associate closely with HSC development. Because the first HSCs have few reliable surface markers, high ESAM expression must be useful to monitor how the first HSCs are developing and moving to other sites. As an additional finding, we detected high myeloid-erythroid but little lymphopoietic potential in a Tie2Lo ckitHi subset of the YS, whose expression level of ESAM was apparently low. We presume that ESAMHi Tie2Hi c-kit+ cells in the PSp/AGM and ESAMLo Tie2Lo c-kitHi cells in the YS differ in their origins and/or as development progresses. If combined with those cell surface markers, a newly described method to trace runt-related transcription factor 1-expressing cells would address several important issues related to “primitive” and “definitive” hematopoiesis.33
It is interesting to speculate that ESAM might be important for HSC functions. A previous study showed that ESAM mediates homophilic interactions between endothelial cells,22 and endothelial cells must represent an important component of HSC-supportive niches in bone marrow.34 A subsequent study of ESAM-deficient mice showed impaired migration of neutrophils through vascular walls after normal adhesion.35 In addition, platelets from ESAM-deficient mice were less prone to disaggregation.36 Thus, ESAM expressed by endothelial and/or stem cells could have functions associated with HSC adhesion and/or migration.
Osteoblastic stromal cells that line trabecular bone are thought to construct niches containing molecules needed to regulate HSC quiescence, proliferation, and differentiation in adult bone marrow.37,38 Other findings indicate that HSCs also interact with sinusoidal endothelial cells at some distance from bones.39,40 Osteoblastic and vascular niches probably function in complementary ways.41,42 The fetal liver has no osteoblasts, and HSC niches have not been identified in that site. High level ESAM display suggests that it should be further explored within the context of endothelial-HSC interactions.
It was an unexpected but intriguing finding that ESAM levels correlate with the stem cell rich fraction even in 22-month-old mice. Although many endothelial antigens on HSCs decline with aging, endomucin and endothelial protein C receptor (CD201) have been reported to be adult HSC markers.17,43 In humans, angiotensin-converting enzyme (CD143) was recently found to mark HSCs through embryonic and adult life.44 However, ESAM seems unique in actually increasing with aging. Many genes involved in NO-mediated signal transduction, stress responses, and inflammation have been linked to HSC aging.4 P-selectin is one of the most highly up-regulated of those stress-related genes. Of particular interest, P-selectin mediates some leukocyte-vascular endothelium interactions and leukocyte extravasation, functions also attributed to ESAM on neutrophils.35 Recent reports showed that stem/progenitor cells continuously circulate outside bone marrow and can actively participate in innate immune responses.45,46 Furthermore, platelets can use P-selectin to recruit marrow hematopoietic cells into sites of injury.47 Although direct evidence is lacking at this moment, further study might implicate elevated ESAM levels in extramedullary migration of HSCs.
Efficient HSC-based therapies and the emerging field of regenerative medicine will benefit from learning more about what defines stem cells. Although patterns of expression of transcription factors and other intracellular proteins are informative, surface markers such as ESAM that are unique to HSCs have special utility.
An Inside Blood analysis of this article appears at the front of this issue.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgment
The authors thank Drs N. Sakaguchi (Kumamoto University) and H. Igarashi (Kawasaki Medical School) for the Rag1/GFP knockin mice.
Authorship
Contribution: T.Y. conducted experiments and analyzed results; T.Y., K.O., P.W.K., and Y.K. designed the research plan and wrote the paper; S.B. and D.V. prepared anti-ESAM antibodies; K.K. and T.M. performed and analyzed microarrays.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Takafumi Yokota, Department of Hematology and Oncology, Osaka University Graduate School of Medicine, C9, 2-2 Yamadaoka, Suita, Osaka 565-0871, Japan; e-mail: yokotat@bldon.med.osaka-u.ac.jp.

![Figure 1. Esam gene is preferentially expressed in the HSC fraction of E14.5 fetal liver. Microarray analyses comparing HSC-enriched and ELP-enriched populations were performed. Two independent tests depicted the Esam gene as preferentially expressed in the HSC-enriched population. Results are shown as relative expression levels of each gene comparing with that of Gapdh of which value is 100. Ratio was calculated by [HSC level]/[ELP level] in each gene. The relative expression levels of c-kit were also shown as internal quality controls.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/113/13/10.1182_blood-2008-07-167106/4/m_zh80100932070001.jpeg?Expires=1769197381&Signature=kZ8t~IG7in3~HTnHSXxJhx-YzzqCY1Q33qCffoKypAHZQ3Vb~392XrAYZFTLgV9wYv7u40RQTOwVFaR7VjkXXu1jxxbT7xZjT4399Nwh2g2bMRgT6zMZuGPrJpi1LjZ5kO8FZ6B4z-FMDpIyVMESR~wYRR9j1n1KIxodpNIamK~UsRrpH7BenoOSmOZH6b9Li8GtlRRuBjlpwjXC8eX359uU8xE6cbS~rKJGRbaLeKBgZLfT3BUCEZxGLy55I6BhW0LkPbTNZfetZ~6jl9P2U6Kqs4YnqPCYY8baLARC~VlpCu5BWbRw0ieOm~yrxHVw-3rDHC5MGGQWpCiY4sWeAQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![Figure 1. Esam gene is preferentially expressed in the HSC fraction of E14.5 fetal liver. Microarray analyses comparing HSC-enriched and ELP-enriched populations were performed. Two independent tests depicted the Esam gene as preferentially expressed in the HSC-enriched population. Results are shown as relative expression levels of each gene comparing with that of Gapdh of which value is 100. Ratio was calculated by [HSC level]/[ELP level] in each gene. The relative expression levels of c-kit were also shown as internal quality controls.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/113/13/10.1182_blood-2008-07-167106/4/m_zh80100932070001.jpeg?Expires=1770814565&Signature=4F5aJpLaaRniJv-OAIVxWimfq2iKFoSsq1IVRFUiayOY7ccUH4-5GKgULAkDEZgBW2pSW36mnrP7T1Gh-PZWHhgWC0e9JHU9D14QsYlIybUQgpH41jZVOnfOxuX1fksa4-SBGEGKObVYVMqS~Fff8-QIWuM~QfctOWqmS06ZLZivTot979GBXsrC0rVSpkVoy2ou23vU5W8tTbEXXhwlI70vcqB-nC5FCwnMToex5wAWxLuAkrsfSg4yGpboHnPY9r5U4ss4QqnfniZMStNr-KuaYjzHec1aZ6s~uiSDk-l8sJvwKR8J-jMXlfRmTMQvJUn2x6DgPYc2WF6h-lXuMQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)