Abstract
The identification of cancer antigens that contribute to transformation and are linked with immune-mediated tumor destruction is an important goal for immunotherapy. Toward this end, we screened a murine renal cell carcinoma cDNA expression library with sera from mice vaccinated with irradiated tumor cells engineered to secrete granulocyte macrophage colony-stimulating factor (GM-CSF). Multiple nonmutated, overexpressed proteins that function in tumor cell migration, protein/nucleic acid homeostasis, metabolism, and stress responses were detected. Among these, the most frequently recognized clone was protein disulfide isomerase (PDI). High titer antibodies to human PDI were similarly induced in an acute myeloid leukemia patient who achieved a complete response after vac-cination with irradiated, autologous GM-CSF–secreting tumor cells in the setting of nonmyeloablative allogeneic bone marrow transplantation. Moreover, ERp5, a closely related disulfide isomerase involved in major histocompatibility complex (MHC) class I chain-related protein A (MICA) shedding, also evoked potent humoral reactions in diverse solid and hematologic malignancy patients who responded to GM-CSF–secreting tumor cell vaccines or antibody blockade of cytotoxic T lymphocyte–associated antigen 4 (CTLA-4). Together, these findings reveal the unexpected immunogenicity of PDIs and raise the possibility that these gene products might serve as targets for therapeutic monoclonal antibodies.
Introduction
Cancer cells typically provoke innate and adaptive immune recognition, but multiple immunosuppressive mechanisms operative in the tumor microenvironment restrain the breadth and magnitude of host reactions.1,2 While the formation of clinically evident disease denotes a failure of endogenous immunity, intra-tumoral lymphocyte infiltrates that are enriched for CD8+ cytotoxic T cells and deficient in FoxP3+ regulatory T cells are tightly correlated with improved patient outcomes after standard oncologic therapy.3-5 Variants of the innate pattern recognition receptor TLR4, which bind avidly to high-mobility-group box 1 (HMGB1) released from tumor cells upon chemotherapy triggered death, are similarly linked to the clinical benefits of conventional cancer treatments.6 Together, these findings suggest a potential contribution for nascent host responses in modulating disease outcome
To enhance the potency of anti-tumor immunity, several groups have devised therapeutic strategies that augment dendritic cell-mediated cancer antigen presentation.7 Among these, vaccination with irradiated tumor cells engineered to secrete granulocyte macrophage colony-stimulating factor (GM-CSF) increases the capacity of CD11b+ dendritic cells to acquire and present cancer antigens to tumor-reactive CD4+ and CD8+ T cells, CD1d-restricted invariant natural killer T cells (NKT cells), and B cells.8-12 Several phase 1 and 2 clinical trials of this immunization scheme in patients with various solid and hematologic malignancies demonstrated the generation of a coordinated humoral and cellular anti-tumor response that effectuated substantial tumor necrosis.13
Although a minority of vaccinated subjects achieved prolonged survival in these studies, most eventually succumbed to progressive disease, implying that additional immune defects remain to be addressed. In this context, substantial evidence delineates cytotoxic T lymphocyte–associated antigen 4 (CTLA-4) as a critical negative regulator of endogenous and vaccine engendered anti-tumor immunity.14 CTLA-4 engagement by B7-1 and B7-2 limits T-cell receptor signal transduction and thereby attenuates lymphocyte clonal expansion and effector activities.15-17 In murine models, the administration of blocking antibodies to CTLA-4 potentiates anti-tumor immunity, particularly in combination with cancer vaccines.18-20 Moreover, the infusion of a fully human anti–CTLA-4 monoclonal antibody (ipilumimab) to advanced melanoma and ovarian carcinoma patients previously immunized with irradiated, autologous, GM-CSF–secreting tumor cells evokes dense T- and B-cell infiltrates in metastatic lesions, which accomplish further tumor destruction.21
The detailed investigation of subjects achieving clinically meaningful benefits on these early-stage trials has yielded important insights into the mechanisms underlying protective tumor immunity in humans.13 The screening of cDNA expression libraries, constructed from responding metastases, with patient sera collected after immunotherapy has led to the identification of specific gene products associated with immune-mediated tumor necrosis. Indeed, humoral reactions to major histocompatibility complex (MHC) class I chain-related protein A (MICA), an NKG2D ligand expressed in tumor cells as part of the DNA damage response,22,23 antagonized the immunosuppressive effects of shed MICA and intensified innate and adaptive anti-tumor cytotoxicity.24 The serologic approach similarly established melanoma inhibitor of apoptosis protein (ML-IAP) as a tumor rejection antigen with the capacity to provoke a coordinated antibody, CD4+, and CD8+ T-cell reaction.25 Likewise, humoral responses to ATP6S1, a putative accessory unit of the vacuolar H+-ATPase complex, were correlated with clinical benefits in some patients, while this gene product was also targeted by vaccination with GM-CSF–secreting B16 melanoma cells in a murine model.26
Based upon the induction of high-titer antibodies to ATP6S1 as a function of immunotherapy in both humans and mice, we wondered whether a detailed analysis of vaccine reactions in a murine tumor model might uncover specific gene products that would prove relevant to the evaluation of anti-tumor immunity in patients. Toward this end, we characterized the antigens that elicited humoral responses after tumor cell vaccination in the RENCA murine renal cell carcinoma model. One experimental advantage of this system is that immunization with irradiated, wild-type RENCA cells affords moderate levels of resistance against subsequent tumor challenge, whereas GM-CSF transduction enhances the intensity of protective immunity.8 Consequently, a comparison of the gene products identified with sera from mice vaccinated with parental or GM-CSF–secreting RENCA cells might also help elucidate the impact of GM-CSF on immune recognition of cancer cells. Here we show that GM-CSF increases the breadth of the tumor antigen repertoire targeted by antibodies, and this unexpectedly includes protein disulfide isomerases (PDIs) in mice and patients.
Methods
Murine tumor model
Renal cell carcinoma (RENCA) cells were cultured in Dulbecco modified Eagle medium (DMEM) containing 10% (vol/vol) inactivated fetal calf serum (FCS), 100 U/mL penicillin/streptomycin, 1 mM nonessential amino acids, and 10 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (HEPES) buffer, pH 7.4. Syngeneic, female BALB/c mice from 8 to 12 weeks of age were obtained from Taconic Farms (Hudson, NY). Animals were immunized subcutaneously on the abdominal wall with 5 × 105 irradiated (35 Gy), GM-CSF–secreting or wild-type RENCA cells at weekly intervals, as previously described.8 Sera were obtained at varying times during vaccination by eye bleeding. All mouse experiments were conducted under a protocol approved by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)–accredited Dana-Farber Cancer Institute Institutional Animal Care and Use Committee (IACUC).
Flow cytometry
RENCA cells (5 × 105) were incubated with sera (1:100 dilution) from naive or vaccinated mice or an immunoglobulin G (IgG) isotype control for 3 hours at room temperature, washed, and then stained with a secondary, phycoerythrin (PE)–conjugated goat anti–mouse IgG (Jackson ImmunoResearch Laboratories, West Grove, PA). In some experiments, cells were stained with PE-conjugated goat anti–mouse pan-IgG, IgG1, IgG2a, IgG2b, or IgG3 secondary antibodies diluted at 1/100 (Santa Cruz Biotechnology, Santa Cruz, CA). Cells were then analyzed with a FACScan cytometer (Becton Dickinson, Franklin Lakes, NJ).
Complement-dependent tumor lysis
RENCA cells cultured in 3% FCS in DMEM were incubated in mouse sera (diluted 1:5) at room temperature. After 60 minutes Low-Tox rabbit complement (1/10; Cedarlane, Burlington, ON) was added for 1 hour at 37°C. Cytotoxicity was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT; Roche Diagnostics, Indianapolis, IN). Cells were incubated for 4 hours at 37°C in 96-well plates. After 24 hours, the intensity of the MTT product was measured at 550 nm using a microplate reader.
cDNA library construction and screening
A cDNA expression library was generated from RENCA cells using previously described methods.26 In brief, total RNA was isolated using guanidine isothiacyanate, mRNA purified over oligo-dT cellulose columns, and cDNA synthesized with Superscript II Reverse Transcriptase (RT; Invitrogen, Carlsbad, CA). The cDNA was cloned into the Lambda Zap vector, and the library was screened according to the manufacturer's instructions (ZAP-cDNA Gigapack III Gold cloning and picoBlue Immunoscreening kits; Stratagene, La Jolla, CA). Sera were pooled from 5 vaccinated mice and preabsorbed against Escherichia coli lysed with nonrecombinant phage. Plaques (106) were plated and screened with sera diluted 1:300 in Tris-buffered saline (TBS)/0.1% Tween-20/2% nonfat dried milk (NFDM), and 0.01% (wt/vol) sodium azide. Reactive clones were detected with a goat anti–mouse pan IgG antibody conjugated to alkaline phosphatase (Jackson ImmunoResearch Laboratories) and plaque-purified through sequential replatings. Plasmid DNA from positive clones was isolated, and the cDNA inserts were sequenced (Molecular Biology Core Facility, Dana-Farber Cancer Institute, Boston, MA) and analyzed with the GenBank BLASTN and BLASTX algorithms (National Center for Biotechnology Information) and the Cancer Immunome Database (http://ludwig-sun5.unil.ch/CancerImmunomeDB).
Tumor antigen expression
Trizol (GIBCO/BRL, Invitrogen) was used to isolate total RNA from tumor cells and normal tissues. Total RNA (10 μg) was electrophoresed through an agarose formaldehyde gel in 3-[N-morpholino]propanesulphonic acid (MOPS) running buffer, transferred to nylon membranes (Hybond-XL; Amersham Biosciences, Piscataway, NJ), and cross-linked with a UV Stratalinker 2400 (Stratagene). 32P-labeled (NEN/Perkin Elmer Life Sciences, Waltham, MA) probes ranging from 500 to 1500 nucleotides were prepared with 25 ng template DNA and the Prime-It II Random Primer Labeling kit (Stratagene). The 18S ribosomal RNA was used as a loading control. Hybridizations were performed overnight at 68°C, and then the filters were extensively washed and developed.
Clinical protocols
The phase I trials of vaccination with lethally irradiated, autologous tumor cells engineered to secrete GM-CSF in advanced melanoma, and non-small cell lung carcinoma patients have been described.27-29 The studies of this same immunization strategy in patients with acute myeloid leukemia/advanced myelodysplasia and ovarian carcinoma will be reported elsewhere (manuscripts in preparation). The phase 1 trial of the fully human anti–CTLA-4 blocking monoclonal antibody (ipilumimiab) in previously vaccinated melanoma and ovarian carcinoma patients has been described.21 All clinical protocols received approval from the Dana-Farber/Harvard Cancer Center Institutional Review Board, the Food and Drug Administration, and the Recombinant DNA Advisory Committee, and informed consent was obtained in accordance with the Declaration of Helsinki for these studies.
Enzyme-linked immunosorbent assays
Anti-PDI antibodies were measured by coating enzyme-linked immunosorbent assay (ELISA) plates (Nunc, Thermo Fisher Scientific, Rochester, NY) overnight at 4°C with 1 to 5 μg/mL histidine-tagged recombinant human PDI protein (ProSpec-TechnoGene, Rehovot, Israel) or control histidine peptide (New England Peptide, Gardner, MA) dissolved in a carbonate buffer, pH 9.6. Next, the wells were blocked overnight at 4°C with 2% NFDM/phosphate-buffered saline (PBS), washed, and then incubated in triplicate with 100 μL patient sera diluted 1:100 in 2% NFDM/PBS overnight at 4°C. A goat anti–human IgG conjugated to alkaline-phosphatase (Jackson ImmunoResearch Laboratories) was added at room temperature, and the plate was developed with pNPP substrate (Sigma-Aldrich, St Louis, MO). The absorbance of PDI minus control histidine peptide at 405 nm was determined.
Anti-ERp5 antibodies were assayed by coating the ELISA plates with 1 μg/mL gluthathione S-transferase-ERp5 (GST-ERp5; Abnova, Walnut, CA) or GST recombinant protein produced with the PGEX 5X-3 vector (Amersham Pharmacia). After blocking overnight with 2% NFDM/PBS at 4°C, the wells were washed in PBS plus Tween-20 (PBST) and incubated in triplicate overnight at 4°C with patient sera diluted 1:50 or a rabbit polyclonal anti–human ERp5 antibody diluted 1:500 (Axxora, San Diego, CA) as a positive control. A goat anti–human IgG conjugated to alkaline-phosphatase (Jackson ImmunoResearch Laboratories) was added at room temperature, and the plate was developed with pNPP substrate (Sigma-Aldrich). The absorbance of ERp5 minus GST at 405 nm was determined.
Statistics
The Student t test was used to compare groups in the complement-dependent lysis experiments.
Results
GM-CSF secreting RENCA cell vaccines stimulate a broad humoral response
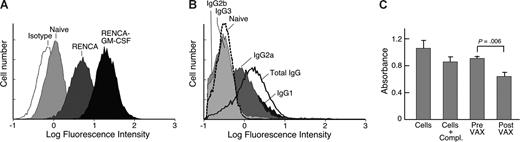
Vaccination with irradiated, GM-CSF–secreting RENCA cells generates more efficient protection against live RENCA cell challenge than vaccination with irradiated, wild-type RENCA cells.8 Tumor rejection involves the activities of CD4+ T cells that secrete a broad profile of Th1 and Th2 cytokines, CD8+ cytotoxic T cells, and CD1d-restricted NKT cells.8,10,11,30 To examine whether immunization also triggers a humoral response, we collected sera from vaccinated mice and evaluated recognition of wild-type RENCA cells with flow cytometry. An anti–mouse pan-IgG secondary antibody was used to measure reactions that typically depend upon CD4+ T-cell help for class switching.31 Both types of RENCA vaccines administered at weekly intervals evoked antibody responses that increased steadily with repetitive immunizations (not shown), but irradiated, GM-CSF–secreting RENCA cells stimulated stronger reactivity than irradiated wild-type cells, while naive mice displayed minimal staining (Figure 1A).
Vaccination with irradiated, GM-CSF–secreting RENCA cells stimulates potent anti-tumor humoral immunity. Sera were collected from syngeneic Balb/c mice that were either naive or immunized 10 times at weekly intervals with irradiated GM-CSF–secreting or parental RENCA cells. (A) Sera were diluted 1:100 and evaluated for reactivity against live RENCA cells by flow cytometry. An anti–mouse pan-IgG secondary antibody was used to detect isotypes that depend upon CD4+ T cell help for class switching. (B) IgG2a and IgG1 are the main isotypes stimulated by vaccination. (C) Sera obtained after vaccination mediates more potent complement-dependent lysis of RENCA cells than sera from naive mice. Representative of 4 independent experiments.
Vaccination with irradiated, GM-CSF–secreting RENCA cells stimulates potent anti-tumor humoral immunity. Sera were collected from syngeneic Balb/c mice that were either naive or immunized 10 times at weekly intervals with irradiated GM-CSF–secreting or parental RENCA cells. (A) Sera were diluted 1:100 and evaluated for reactivity against live RENCA cells by flow cytometry. An anti–mouse pan-IgG secondary antibody was used to detect isotypes that depend upon CD4+ T cell help for class switching. (B) IgG2a and IgG1 are the main isotypes stimulated by vaccination. (C) Sera obtained after vaccination mediates more potent complement-dependent lysis of RENCA cells than sera from naive mice. Representative of 4 independent experiments.
To examine whether these humoral responses might contribute to anti-tumor activity, we determined the isotypes stimulated with vaccination. IgG2a and IgG1 antibodies were readily demonstrated, whereas IgG2b and IgG3 reactions were not detected (Figure 1B). Consistent with the ability of IgG2a antibodies to activate complement, sera obtained after vaccination mediated increased complement-dependent killing of RENCA cells compared with sera obtained before vaccination (P = .006; Figure 1C). These results suggest that the vaccine-induced antibodies are biologically active.
To identify the targets of these humoral responses, we constructed a cDNA expression library in bacteriophage using mRNA isolated from wild-type RENCA cells. Sera collected from mice immunized 10 times with irradiated, wild-type, or GM-CSF–secreting RENCA cells were diluted 1:300 and used for the library screening. Immunoreactive plaques were detected with an anti-mouse IgG secondary antibody and purified through further replatings. The screening with sera from mice vaccinated with wild-type RENCA cells yielded only 3 clones that represented 2 distinct gene products, but GM-CSF stimulated the detection of 177 clones, which represented 26 distinct gene products. Additional phage plate assays revealed that sera from mice immunized with GM-CSF–secreting cells also recognized the 2 antigens identified with parental RENCA vaccines, while sera from mice immunized with wild-type cells failed to detect any of the GM-CSF–associated targets. Moreover, sera from naive mice did not react with any of the 28 antigens characterized in the 2 screens. Together, these findings indicate that although vaccination with irradiated wild-type cells modestly enhances humoral recognition of RENCA cells, GM-CSF secretion results in a marked diversification of the antigenic repertoire targeted by antibodies.
RENCA vaccine targets are involved in oncogenic pathways
Of the 28 distinct antigens recognized by sera from mice immunized with GM-CSF–producing cells, 21 encode known proteins, many of which have been previously implicated in transformation (Table 1). These gene products can be grouped based upon putative roles in cell function. One set of antigens contributes to tumor cell migration. Among these, the cell surface protein CD44, which is also aberrantly expressed in human renal cell carcinomas, mediates tumor cell adhesion to endothelial cells and the extra-cellular matrix, while rho-associated coiled-coil forming kinase-2 (ROCK2) modifies the cellular cytoskeleton to promote metastasis.32,33 A second group of targets, which includes eukaryotic translational initiation factor 4A1 (eIF4A-1), ribosomal protein L15 (RPL15), and the proteasome subunit, β type 5 (PSMB5), are involved in protein translation and degradation, 2 pathways that are critical to the regulation of tumor cell growth and survival.34,35 A third set of antigens consists of heat-responsive protein 12 (HRP12), autophagy-related 3 (atg3), and autophagy-related 12 (atg12), which enhance the resistance of tumor cells to various forms of stress.36,37 A fourth group of targets participates in intermediary metabolism and includes farnesyl diphosphate synthetase (FDPS), a key enzyme in the mevalonate biosynthetic pathway that generates cholesterol and isoprenoids, and ATP synthase mitochondrial F1 complex δ subunit (atp5d), an important component of oxidative phosphorylation.38,39 A final set of antigens encompasses nuclear proteins that contribute to chromatin, DNA repair, and transcriptional control.
The sequencing of the 21 known vaccine targets failed to reveal any alterations compared with the GenBank database, eliminating mutation as a possible mechanism for their immunogenicity. Nonetheless, previous work suggested that the increased expression of normal differentiation proteins, such as Her2/neu and MART-1, might provoke immune recognition of tumor cells.40 Because transformation may involve the aberrant activation of specific oncogenic pathways, we thus characterized the expression of selected RENCA antigens by northern analysis (Figure 2). All of the gene products examined, including ROCK2, FDPS, guanine nucleotide-binding protein β2 subunit (GNB2), transcription elongation factor A (SII) 1 (TCEA1), structure-specific recognition protein 1 (SSRP1), IQGAP1, and CD44, displayed increased transcript levels in RENCA cells compared with normal kidney, liver, and spleen (with the exception of FDPS that was expressed to a comparable degree in the liver, the major site of cholesterol biosynthesis).38 High levels of SSRP1 and surface CD44 protein were also confirmed with western analysis and flow cytometry of RENCA cells, respectively (not shown). Together, these results indicate that the humoral response stimulated by vaccination with GM-CSF–secreting RENCA cells targets gene products that are overexpressed and involved in diverse oncogenic pathways.
RENCA vaccine targets show enhanced expression in tumor cells compared with normal tissues. RNA was isolated from RENCA cells and normal kidney, liver, and spleen, and the transcript levels for multiple RENCA antigens were determined with northern analysis. The 18S ribosomal RNA served as a loading control.
RENCA vaccine targets show enhanced expression in tumor cells compared with normal tissues. RNA was isolated from RENCA cells and normal kidney, liver, and spleen, and the transcript levels for multiple RENCA antigens were determined with northern analysis. The 18S ribosomal RNA served as a loading control.
PDI is immunogenic in myeloid leukemia
The human orthologs of ROCK2, SSRP1, eIF4A, IQGAP, aldose reductase (Akr1b3), acetyl-coenzyme A acetytransferase 2 (ACAT2), and heterogeneous nuclear ribonucleoprotein C (hnRNPC1) were previously deposited in the Cancer Immunome Database (http://ludwig-sun5.unil.ch/CancerImmunomeDB), reflecting their earlier identification as antibody targets in cancer patients through the screening of human tumor-derived cDNA expression libraries.41-43 This conservation of immunogenicity supports our idea that the characterization of vaccine responses in a murine model might prove informative for the analysis of human anti-tumor immunity. The most frequently recognized antigen in the RENCA library screening was PDI, which accounted for 86% of the clones detected with sera from mice immunized with GM-CSF–secreting cells. PDI is an abundant resident of the endoplasmic reticulum, where it functions as a disulfide isomerase and chaperone to facilitate proper folding of nascent proteins.44-47 Some evidence suggests that PDI may also be localized to the cell membrane in tumor cells and there modulate integrin-dependent adhesion and sensitivity to chemotherapy.48-50
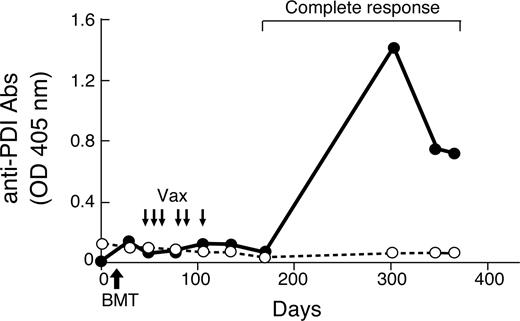
To examine whether PDI is immunogenic in cancer patients, we established an ELISA with recombinant full-length human protein and evaluated sera from 46 metastatic melanoma, 22 metastatic non–small cell lung carcinoma, 2 metastatic ovarian carcinoma, and 12 acute myeloid leukemia patients who were enrolled on phase I clinical trials of vaccination with irradiated, autologous tumor cells engineered to secrete GM-CSF. We used a pan-human IgG secondary antibody to measure preferentially those isotypes dependent upon CD4+ T-cell help for class switching. Humoral responses to PDI proved uncommon in this cohort, as only one melanoma and one leukemia patient were reactive, whereas 30 healthy donors were negative. Although the anti-PDI antibodies in the melanoma patient were detectable before vaccination and not altered with therapy, a longitudinal analysis of the leukemia subject uncovered an intriguing correlation between humoral immunity and clinical outcome (Figure 3).
Anti-human PDI antibodies are associated with the induction of a clinical response in a patient with acute myeloid leukemia. A longitudinal analysis of humoral reactivity to human PDI (•) and human PBEF (○) was performed with serial sera samples diluted 1:100 and a secondary anti–human pan-IgG secondary antibody. This leukemia patient first underwent a nonmyeloablative allogeneic bone marrow transplant (BMT) and then was vaccinated with irradiated, autogous GM-CSF–secreting leukemia cells as indicated. Reduction of the immunosuppression for prophylaxis of graft-versus-host disease preceded the complete hematologic response, which has been maintained with 17 months of follow-up.
Anti-human PDI antibodies are associated with the induction of a clinical response in a patient with acute myeloid leukemia. A longitudinal analysis of humoral reactivity to human PDI (•) and human PBEF (○) was performed with serial sera samples diluted 1:100 and a secondary anti–human pan-IgG secondary antibody. This leukemia patient first underwent a nonmyeloablative allogeneic bone marrow transplant (BMT) and then was vaccinated with irradiated, autogous GM-CSF–secreting leukemia cells as indicated. Reduction of the immunosuppression for prophylaxis of graft-versus-host disease preceded the complete hematologic response, which has been maintained with 17 months of follow-up.
This subject with refractory acute myeloid leukemia underwent a nonmyeloablative allogeneic bone marrow transplant and then received irradiated, autologous GM-CSF–secreting tumor cells early posttransplant (the full clinical trial will be presented in detail separately). This sequential strategy is based on experiments that established the ability of GM-CSF–producing tumor cells to enhance protective immunity after allogeneic bone marrow transplantation in murine models.51,52 After the completion of vaccination in the human protocol, the immunosuppressive therapy for prophylaxis against graft-versus-host disease was reduced, and the leukemia patient achieved a complete hematologic response that was temporally associated with the development of high titer anti-PDI antibodies. No humoral reactivity against human pre-B-cell colony–enhancing factor (PBEF), an ortholog of another RENCA antigen identified in the murine screen (Table 1), was observed, however, establishing the specificity of the antibody response. While further studies are required to elucidate the precise contributions of graft-versus-leukemia and vaccine-induced immunity to this patient's clinical course, our findings nonetheless underscore the immunogenicity of human PDI in the context of tumor destruction.
Humoral responses to the related disulfide isomerase ERp5
Recent work has delineated a key role for ERp5, a closely related PDI, in promoting the shedding of MICA from tumor cells.46,47,53 MICA is a ligand for NKG2D, a receptor expressed on NK cells, γδ T cells, and CD8+ T cells that upon engagement triggers perforin dependent cytolysis and provides costimulation.23,54,55 MICA is induced in tumor cells through the DNA damage response, but the surface localization of ERp5 renders the α3 domain of MICA susceptible to proteolysis; the release of soluble ligand in turn provokes the down-regulation of NKG2D.22,53 We previously reported that some advanced melanoma and non–small cell lung carcinoma patients who responded to vaccination with irradiated, autologous, GM-CSF–secreting tumor cells and antibody blockade of CTLA-4 generated high-titer antibodies to MICA.24 These therapy-induced humoral reactions antagonized the immunosuppressive effects of shed MICA and augmented innate and adaptive anti-tumor cytotoxicity.
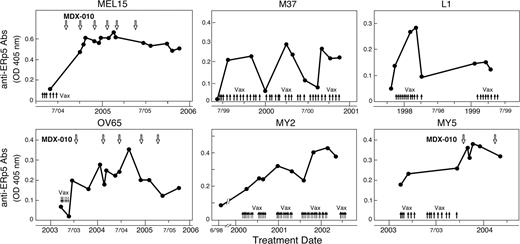
Based on these findings and the immunogenicity of PDI, we wondered whether the disulfide isomerase ERp5 might similarly evoke antibody responses. To explore this idea, we established an ELISA with recombinant human ERp5 protein and evaluated the development of antigen-specific IgG antibodies in the same patient cohort used for the PDI analysis. Remarkably, 3 of the 4 subjects previously shown to mount anti-MICA antibodies as a function of immunotherapy also generated humoral responses to ERp5 (Figure 4 top row). Metastatic melanoma patient MEL15 achieved an ongoing near complete response to vaccination and CTLA-4 blockade (36+ months); metastatic melanoma subject M37 remains in a complete response 8 years after vaccination; and metastatic non–small cell lung carcinoma patient L1 achieved stable disease of 2 years duration after immunization. Furthermore, we identified 3 additional subjects without anti-MICA antibodies who similarly developed humoral reactions to ERp5 as a consequence of GM-CSF–secreting vaccines and/or CTLA-4 antibody blockade (Figure 4 bottom row). These included an advanced ovarian carcinoma patient who achieved an ongoing partial response (3.5+ years) and 2 advanced myeloid leukemia subjects who demonstrated prolonged stable disease (30 and 18 months); these leukemia patients were enrolled in a vaccination protocol that did not involve allogeneic bone marrow transplantation (this trial will be reported in detail elsewhere; V.H., M.V., Haesook Kim, Mildred Pasek, Corey Cutler, John Koreth, Edwin Alyea, Stefanie Sarantopoulos, Joseph H. Antin, J.R., Christine Canning, Jeffery Kutok, Martin C. Mihm, G.D., and R.S., manuscript submitted). Together, these investigations delineate an association between humoral reactions to ERp5 and immune-mediated tumor destruction that is operative in 4 different types of human malignancies.
Humoral responses to ERp5 are associated with immune-mediated tumor destruction in diverse solid and hematologic malignancies. A longitudinal analysis of antibodies to human ERp5 was performed with serial sera samples diluted 1:50 and a secondary anti–human pan-IgG antibody. MEL15 and M37 are metastatic melanoma patients, L1 is a metastatic non-small cell lung carcinoma patient, OV65 is a metastatic ovarian carcinoma patient, and MY2 and MY5 are acute myeloid leukemia patients. denotes vaccination with irradiated, autologous, GM-CSF–secreting tumor cells. indicates the infusion of a fully human anti–CTLA-4 blocking monoclonal antibody (MDX-010; ipilumimiab).
Humoral responses to ERp5 are associated with immune-mediated tumor destruction in diverse solid and hematologic malignancies. A longitudinal analysis of antibodies to human ERp5 was performed with serial sera samples diluted 1:50 and a secondary anti–human pan-IgG antibody. MEL15 and M37 are metastatic melanoma patients, L1 is a metastatic non-small cell lung carcinoma patient, OV65 is a metastatic ovarian carcinoma patient, and MY2 and MY5 are acute myeloid leukemia patients. denotes vaccination with irradiated, autologous, GM-CSF–secreting tumor cells. indicates the infusion of a fully human anti–CTLA-4 blocking monoclonal antibody (MDX-010; ipilumimiab).
Discussion
The crafting of genetic and biochemical strategies to characterize cancer antigens has yielded the insight that most patients generate antibody and T-cell responses against tumors.1 The targets of these adaptive immune reactions include mutated gene products, cancer-testes antigens, overexpressed normal differentiation proteins, and aberrantly translated intronic sequences.40 Notwithstanding the breadth of targets capable of provoking immune recognition of cancer cells, relatively little is known regarding which gene products are associated with immune-mediated tumor destruction and whether these gene products are shared among different tumor types. In this study, we undertook a detailed analysis of vaccination responses in a murine tumor model and in patients enrolled in early-stage clinical trials to learn more about the specific antigens linked with protective tumor immunity.
Our studies revealed that the engineering of tumor cells to secrete GM-CSF resulted in a marked increase in the repertoire of cancer antigens recognized by antibodies. The vaccine targets proved to be overexpressed proteins involved in diverse oncogenic pathways. Among the antigens, the human ortholog of PDI manifested immunogenicity in a limited number of cancer patients, whereas the closely related disulfide isomerase ERp5 elicited humoral reactions that were temporally associated with clinically significant anti-tumor effects in 4 different solid and hematologic malignancies. Because ERp5 facilitates immune escape through the enhancement of MICA shedding53 and may also promote tumor cell invasion through modifying surface thiols,56,57 our results raise the possibility that this disulfide isomerase may be a useful target for cancer immunotherapy.
Most of the gene products characterized in the RENCA library screening were intracellular, nonmutated proteins. These could be grouped based on putative function into several oncogenic pathways, including tumor cell migration, protein homeostasis, stress responses, intermediary metabolism, and chromatin/transcriptional control. The dependence of cancer cells on these networks may account for their increased expression, which in turn likely contributes, at least in part, to their immunogenicity.40 The physical association of some of the vaccine targets with RNA and DNA might further promote humoral reactivity through the concurrent engagement of TLR7 and TLR9, respectively, as has been demonstrated in murine models of systemic lupus erythematosis.58,59 Consistent with this idea, the human orthologs of SSRP1, hnRNPC1, and H1 histone family member 0 (H1[0]) were previously identified as targets of antibody responses in patients with autoimmune diseases.60-62 Moreover, the ability of GM-CSF to enhance the acquisition and processing of dying tumor cells by recruited dendritic cells and macrophages is likely to be another important factor in diversifying the antibody response.63 Additional studies are required to determine, however, whether the RENCA targets also elicit a coordinated helper and cytotoxic T-cell reaction.
While the antibody responses to intracellular moieties may enhance tumor immunity through the cross-presentation of antigen-antibody complexes to dendritic cells,64 humoral reactions against cell surface proteins might mediate direct anti-tumor effects as well. Consistent with these findings, our characterization of the RENCA model demonstrated that sera obtained after vaccination mediates more potent complement-dependent tumor lysis than sera from naive mice. While earlier reports suggested that PDI was present at the surface of some tumor cells,48-50 we were unable to document specific membrane staining in RENCA cells using 2 different commercially available monoclonal antibodies. Other surface proteins such as CD44 and PBEF might serve as the relevant targets for the antibody-dependent cytotoxicity of RENCA cells, although this will require additional investigation. Nonetheless, we confirmed the strong surface expression of PDI and ERp5 in multiple murine and human cancer cell lines,53 including tumor types similar to the patients reported here. Unfortunately, autologous cancer cells were not available for characterization, as these had been used for vaccine manufacture. The membrane localization and association of ERp5-specific humoral immunity with tumor destruction raises the possibility that disulfide isomerase might be an attractive target for therapeutic monoclonal antibodies. However, our efforts to evaluate the ability of patient sera to mediate direct anti-tumor effects through binding ERp5 have been limited thus far by an inability to engineer stable ERp5 surface expression (not shown). Whether anti-ERp5 antibodies modulate MICA shedding or antagonize tumor cell invasion are other intriguing issues that remain for further investigation. Lastly, the concordance of humoral reactions to ERp5 and MICA in some patients who achieve clinically significant benefits in response to immunotherapy underscores the tumor suppressor activities of the NKG2D pathway in humans.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank the National Institutes of Health (Bethesda, MD), the Leukemia & Lymphoma Society (White Plains, NY), the GABBA Graduate Program (Oporto University), and the Portuguese Foundation for Science and Technology for their educational and financial support.
National Institutes of Health
Authorship
Contribution: C.F., R.S., V.H., M.V., M.J., J.R., R.S., and D.D. performed research; C.F., R.S., V.H., M.V., M.J., J.R., R.S., D.D., and G.D. analyzed data; and C.F. and G.D. wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Glenn Dranoff, Dana-Farber Cancer Institute, Dana 520C, 44 Binney Street, Boston, MA 02115; e-mail: glenn_dranoff@dfci.harvard.edu.