Abstract
To evaluate outcome and prognosis of patients with T-cell lymphoma we analyzed 343 patients treated within trials of the German High-Grade Non-Hodgkin Lymphoma Study Group (DSHNHL). Two hundred eighty-nine patients belonged to 1 of the 4 major T-cell lymphoma subtypes: anaplastic large cell lymphoma (ALCL), anaplastic large cell lymphoma kinase (ALK)–positive (n = 78); ALCL, ALK-negative (n = 113); peripheral T-cell lymphoma, unspecified (PTCLU; n = 70); and angioimmunoblastic T-cell lymphoma (AITL; n = 28). Treatment consisted of 6-8 courses of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone/prednisolone) or etoposide plus (CHOEP). Three-year event-free survival (EFS) and overall survival were 75.8% and 89.8% (ALK-positive ALCL), 50.0% and 67.5% (AITL), 45.7% and 62.1% (ALK-negative ALCL), and 41.1% and 53.9% (PTCLU), respectively. The International Prognostic Index (IPI) was effective in defining risk groups with significantly different outcomes. For patients, ≤ 60 years with lactate dehydrogenase ≤ upper normal value (UNV), etoposide improved improved 3-year EFS: 75.4% versus 51.0%, P = .003. In patients > 60 years 6 courses of CHOP administered every 3 weeks remains the standard therapy. Patients with ALK-negative ALCL, PTCLU, or AITL presenting with IPI > 1 have a poor prognosis and should be considered candidates for novel treatment strategies.
Introduction
Mature T-cell and natural killer (NK)–cell lymphomas are rare and heterogeneous diseases following an aggressive clinical course which necessitates immediate therapy. Outcome is generally believed poor although only 1 recent study1 comprises all major T-cell lymphoma subtypes according to the World Health Organization classification2,3 with spin-off studies reporting on clinical outcome and prognostic factors of various subtypes.4-6
In contrast to the progress made in the treatment of aggressive B-cell lymphoma,7-9 evidence for similar therapeutic improvements in T- and NK-cell lymphoma is largely absent. Therefore, we analyzed a large cohort of patients with T-cell lymphoma who have been treated on protocols of the German High-Grade Non-Hodgkin Lymphoma Study Group (Deutsche Studiengruppe Hochmaligne Non-Hodgkin Lymphome [DSHNHL]). We were particularly interested in the long-term results achieved with “standard” CHOP (cyclophosphamide, hydroxydaunorubicin (doxorubicin), Oncovin (vincristine), and prednisone/prednisolone) and CHOP-like therapy and wanted to define prognostic factors which should influence the decision which patients should be treated on standard or experimental protocols in the future.
Methods
Patients
Between October 1993 and May 2007, 343 patients with mature nodal or extranodal T-cell or NK-cell lymphoma2 were treated on protocols of the DSHNHL. Mandatory reference pathology was performed in 1 of the 6 German Reference Centers for Malignant Lymphomas (Berlin, Frankfurt, Kiel, Lübeck, Ulm, and Würzburg) prior to therapy. Of 343 patients, 320 could be assigned to 1 of the following subtypes: anaplastic large cell lymphoma (ALCL), anaplastic large cell lymphoma kinase-positive (ALK-positive); ALCL, ALK-negative; peripheral T-cell lymphoma unspecified (PTCLU); angioimmunoblastic T-cell lymphoma (AITL); NK/T-cell lymphoma; lymphoblastic lymphoma; enteropathy-type T-cell lymphoma; hepatosplenic γδ T-cell lymphoma; or subcutaneous panniculitis-like T-cell lymphoma. In a subset of ALCL cases the ALK status had not been determined at the time of diagnosis, because antibodies were not available at that time. In these cases, the ALK status was defined retrospectively using the ALK1 antibody from DAKO (dilution 1:80, citrate buffer pH 6.0). The definition of ALK1-negative ALCL followed the current World Health Organization classification and required characteristic large cell morphology (indistinguishable from ALK-positive ALCL), strong and consistent expression of CD30 and negativity for ALK1. Moreover, most cases had expression of at least 1 cytotoxic molecule (Perforin, Granzyme B, or TIA). Twenty-three patients were excluded from the analysis because the ALK status of some ALCL patients remained unknown (n = 11) or the T-cell lymphoma subtype could not be confirmed for technical (n = 3) or other reasons (n = 9).
All patients had thorough baseline and follow-up assessments (history, clinical evaluation, laboratory tests, chest and abdominal CT scans, and bone marrow biopsies) as required by the respective study protocol. Patients were requested to visit the study site every 3 months for the first 2 years, every 6 months in the years 3 to 5, and annually thereafter. Case report forms of each patient allowed for a detailed analysis of protocol adherence. All studies were undertaken in accordance with the Helsinki declaration, study protocols were approved by the ethics committee of each participating institution and each patient gave informed consent.
Treatment
All patients included in this analysis were treated on prospective phase II or phase III protocols of the DSHNHL. Eighty-three patients were treated on the NHL-B1 trial,10 39 patients on the NHL-B2 trial,11 12 patients on the Hi-CHOEP (cyclophosphamide, hydroxydaunorubicin (doxorubicin), Oncovin (vincristine), etoposide, and prednisone/prednisolone) phase II trial,12 59 patients on the Hi-CHOEP phase III trial,13 32 patients on the MegaCHOEP phase II trial,14,15 38 patients on the MegaCHOEP phase III trial,9,16 and 57 patients on the RICOVER-60 trial.8
For patients with T-cell lymphoma, all of the phase III trials compared the standard CHOP regimen17 to 6 or 8 courses of CHOP given every 2 weeks (CHOP-14) or to CHOP plus etoposide (CHOEP-14 or -21), or compared CHOEP to a dose-escalated (Hi-CHOEP), or a mega-dose (MegaCHOEP) variant, the latter one necessitating repeated transplantation of hematopoietic stem cells. The phase II studies were dose-finding studies using escalating doses of cyclophosphamide, doxorubicin, and etoposide compared with standard CHOP plus etoposide protocols. Radiotherapy to sites of bulky disease (≥ 7.5 cm) and to extranodal disease was part of all protocols except for the MegaCHOEP phase II trial where radiotherapy was optional.
For further details, see the respective publications in references 8 through 168-16 ; details of patient allocation are given in supplemental Table 1 (available on the Blood Web site; see the Supplemental Materials link at the top of the online article).
Statistical analysis
Event-free survival (EFS) defined as time from randomization or start of therapy to disease progression, start of salvage therapy, further (unplanned) treatment (excluding first-line chemo- and radiotherapy), relapse, or death from any cause, and overall survival (OS) defined as time from randomization or start of therapy to death from any cause, were analyzed by the Kaplan-Meier method. Estimators at 3 years for these endpoints are given with 95% confidence interval (CI) limits.
The standard International Prognostic Index (IPI) was used to classify patients as belonging to the low (IPI 0, 1), low-intermediate (IPI 2), high-intermediate (IPI 3), or high risk (IPI 4, 5) groups.18 Univariate analyses to test the IPI score and treatment effects were conducted using the log-rank test. For multivariate analyses, Cox proportional hazard models including single IPI factors or histologic subtypes adjusted for the IPI factors, respectively, were calculated. Relative risks with 95% confidence intervals and the respective P values are presented.
To test whether there were significant differences between histologic subtypes and patient characteristics we used the Kruskall-Wallis test for age and χ2 tests, or if necessary Fisher exact tests for the other items. For these comparisons, the Bonferroni correction for multiple comparisons (0.05/10) was used.
All other tests for significance were 2-sided at the 5% significance level. Statistical analyses were done with SPSS Version 15.0.
Results
Of 320 patients, 78 (24.4%) suffered from ALK-positive ALCL, 113 patients (35.3%) had ALK-negative ALCL, 70 patients (21.9%) had PTCLU, 28 patients (8.8%) had AITL, 19 patients (5.9%) had NK/T-cell lymphoma, 7 patients (2.2%) had lymphoblastic lymphoma, 2 patients each (0.6%) had enteropathy-type T-cell lymphoma or hepatosplenic γδ T-cell lymphoma, and 1 patient had subcutaneous panniculitis-like T-cell lymphoma.2
Because the low patient numbers in other disease categories precluded meaningful statistical analyses we focused on the 4 major subtypes of T-cell lymphomas: 289 patients with ALK-positive ALCL, ALK-negative ALCL, PTCLU, or AITL, which comprised 90.3% of all 320 patients. The most important clinical characteristics of these and the other remaining patients are given in Table 1. We found a male preponderance for all major subentities; the median age varied between 37 and 64 years with the ALK-positive ALCL patients representing the youngest group of patients. After adjustment for multiple comparisons we found significant differences between the major subgroups with regard to age (P < .001), lactate dehydrogenase (LDH; P = .001), stage (P = .004), IPI-score (P = .005) and B-symptoms (P = .003). Approximately 70% of patients with AITL presented with an elevated LDH while this was less frequent in PTCLU and ALCL patients, both ALK-positive or -negative. Except for the AITL patients who presented with advanced stage (III or IV) in more than 80% of cases, between 43% and 53% of patients belonging to the other subgroups presented with stage III/IV disease.
Extranodal disease was common in all subtypes (41%-56%), all localizations are given in supplemental Table 2. Highly significant (P < .001) differences were seen for involvement of soft tissues (21% in ALK-positive ALCL, 8% in ALK-negative ALCL, 0%-1% in other subtypes) and bone marrow (0% in ALK-positive ALCL, 4% in ALK-negative ALCL, 11% in PTCLU, 27% in AITL, and 13% in other subtypes) when the 4 major subgroups were compared. Although formally significant, the differences found in the frequencies of involvement of the main nasal cavity and adrenal glands should be taken with great caution because only few patients mainly from rare subtypes made the difference. B-symptoms were most frequent in AITL patients and bulky disease was seen more often in ALCL, either ALK-positive or -negative, than in other subtypes, but this was not significant after adjustment for multiple comparisons. More than half of all patients with ALCL (ALK-positive or -negative) and 46% of PTCLU but only 21% of AITL patients belonged to the IPI low-risk group.
Treatment outcome
With a median follow-up of 43.8 months for the whole group, the 3-year EFS and OS for patients in the 4 major subtypes were as follows: 75.8% (95% CI 65.8%-85.8%) and 89.8% (95% CI 82.5%-97.1%) for ALK-positive ALCL; 50.0% (95% CI 31.6%-68.4%) and 67.5% (95% CI 50.1%-84.9%) for AITL; 45.7% (95% CI 36.3%-55.1%) and 62.1% (95% CI 52.9%-71.3%) for ALK-negative ALCL; 41.1% (95% CI 29.5%-52.7%) and 53.9% (95% CI 41.7%-66.1%) for PTCLU. EFS and OS for the major subgroups and all remaining patients are shown graphically in Figure 1.
Event-free survival and overall survival of 320 patients with T-cell lymphoma according to histologic subtype.
Event-free survival and overall survival of 320 patients with T-cell lymphoma according to histologic subtype.
For patients with NK-/T-cell lymphoma, 3-year EFS was 36.1% (95% CI 14.1%-58.1%) and 3-year OS was 46.3% (95% CI 23.4%-69.2%).
Two patients with enteropathy-type T-cell lymphoma died with progressive disease 1 and 8 months after diagnosis; and 2 patients with hepatosplenic γδ T-cell lymphoma died with progressive disease 7 and 10 months after diagnosis. The patient with subcutaneous panniculitis was in CR 38 months after diagnosis.
The EFS and OS of 78 patients with ALK-positive ALCL was significantly better (P < .001 for both endpoints) than for patients with any of the other diagnoses in univariate and multivariate analysis adjusted for IPI factors. No significant differences in EFS or OS were seen between the other major subtypes (ALK-negative ALCL, PTCLU, and AITL) in univariate analysis. Using a Cox model adjusting for IPI factors we did not find a significant difference in EFS or OS for patients with PTCLU compared with patients with ALK-negative ALCL but patients with AITL did significantly better than patients with ALK-negative ALCL (relative risk 0.6, P = .046 for EFS; relative risk 0.5, P = .037 for OS; see Table 2).
Treatment results were achieved with 6 to 8 courses of the CHOP or CHOEP regimen at conventional doses except for the patients on phase II studies who received the same drugs but with escalated doses of cyclophosphamide, etoposide, and to a lesser extent also of doxorubicin. This gave us the opportunity to compare treatment results obtained with variations of the CHOP regimen.
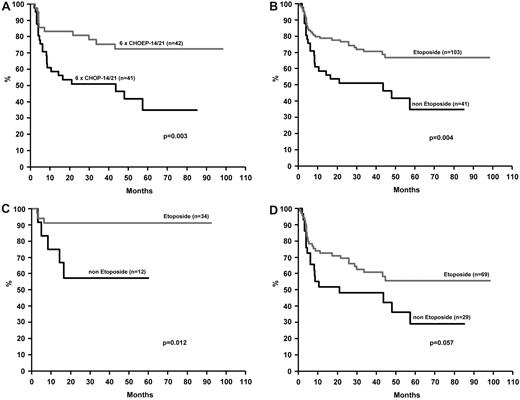
In younger patients with normal LDH the addition of etoposide to CHOP-21 or CHOP-14 significantly improved 3-year EFS (75.4% [95% CI 62.1%-88.7%] vs 51.0% [95% CI 35.7%-66.3%], P = .003, see Figure 2A). OS was not significantly affected (P = .176).10 This finding was further substantiated when all younger patients from the NHL-B1 and Hi-CHOEP phase II/III trials with normal LDH (n = 144) given CHOP plus etoposide at various doses (n = 103) were compared with patients (n = 41) receiving CHOP only (3-year EFS 70.5%, 95% CI 61.3%-79.7%) after CHOEP and 51.0% (95% CI 35.7%-66.3%) after CHOP (P = .004, see Figure 2B). OS at 3 years did not significantly differ with etoposide (81.3% [95% CI 73.5%-89.1%]) or without (75.2% [95% CI 61.9%-88.5%]; P = .285). The positive effect of etoposide on EFS was confirmed when only patients with ALK-positive ALCL were considered [3-year EFS 91.2% (95% CI 81.6%-100.0%) versus 57.1% (95% CI 28.5%-85.7%), P = .012]; the comparison showed a trend in the same direction when the other subentities were taken together (3-year EFS 60.7% [95% CI 48.7%-72.7%] vs 48.3% [95% CI 30.1%-66.5%], P = .057; see Figure 2C-D).
Event-free survival of younger patients (18-60 years, LDH ≤ UNV). Panel A refers to patients treated on the NHL-B1 trial. Panel B refer to patients treated on the NHL-B1 or Hi-CHOEP phase II/III trials. Patients who did or did not receive etoposide plus CHOP (CHOEP) are compared. Panels C and D show the EFS for patients with ALCL, ALK-positive (C), and with other major subtypes (D).
Event-free survival of younger patients (18-60 years, LDH ≤ UNV). Panel A refers to patients treated on the NHL-B1 trial. Panel B refer to patients treated on the NHL-B1 or Hi-CHOEP phase II/III trials. Patients who did or did not receive etoposide plus CHOP (CHOEP) are compared. Panels C and D show the EFS for patients with ALCL, ALK-positive (C), and with other major subtypes (D).
Neither shortening of the time interval from 3 to 2 weeks (CHOP-21 vs CHOP-14), administration of 8 instead of 6 courses of CHOP-14, or the addition of etoposide (CHOEP) significantly improved EFS and OS in elderly patients. Although the low patient numbers for some comparisons would call for further evaluation in larger cohorts of patients, for the time being CHOP-21 remains standard of care for older patients with T-cell lymphoma.
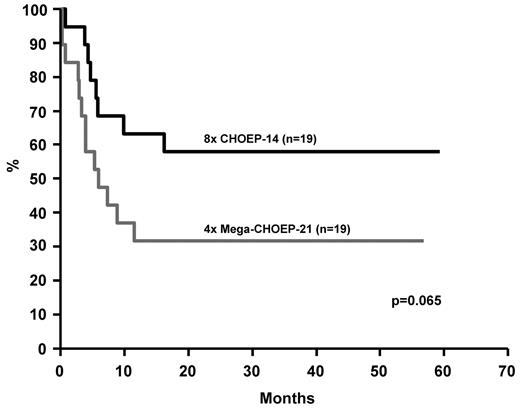
Notably, all attempts to improve treatment results for younger patients by escalating standard doses of cyclophosphamide, doxorubicin, etoposide, and prednisone failed. This extends findings from the High/CHOEP phase III trial.13 On the contrary, and similar to our recent observations in B-cell lymphoma9 younger patients with T-cell lymphoma and age-adjusted IPI 2 or 3 treated on the MegaCHOEP program did worse than those given 8 courses of standard CHOEP-14 (Figure 3). The 3-year EFS was 57.9% (95% CI 35.8%-80.0%) for patients treated with CHOEP-14 compared with 31.6% (95% CI 10.6%-52.6%; P = .065) for patients treated with MegaCHOEP. OS did not significantly differ (P = .245).
Event-free survival of younger high-risk patients (18-60 years, aaIPI 2,3) treated on the MegaCHOEP phase III trial comparing conventional (8 × CHOEP-14) with repetitive high-dose therapy
Event-free survival of younger high-risk patients (18-60 years, aaIPI 2,3) treated on the MegaCHOEP phase III trial comparing conventional (8 × CHOEP-14) with repetitive high-dose therapy
Prognostic factors
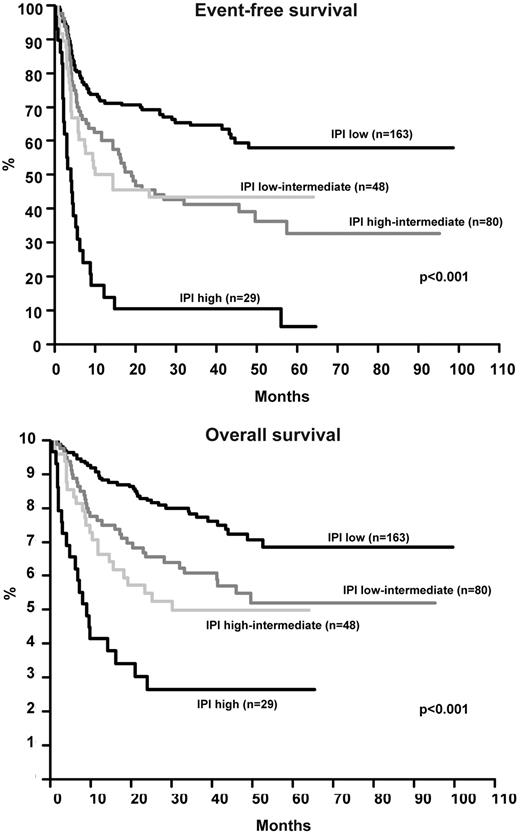
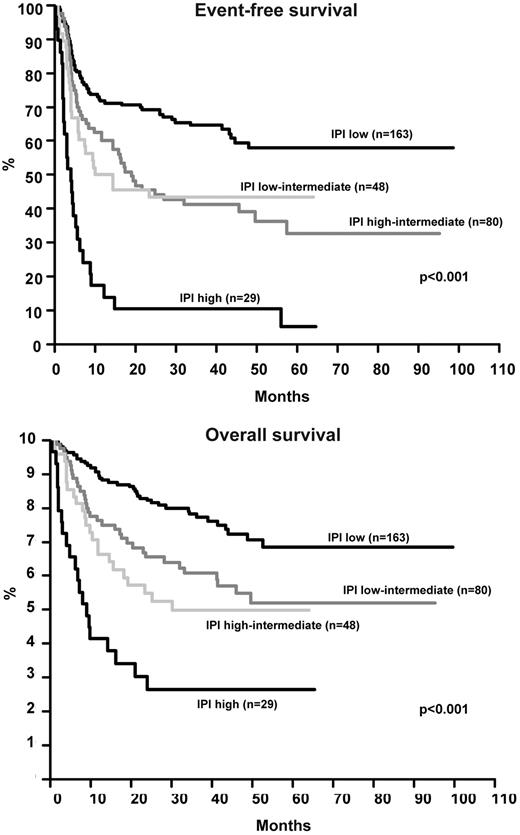
The EFS and OS of all 320 patients by IPI is shown in Figure 4. Using the Cox model, only age and LDH significantly influenced OS, all IPI factors except for stage were significant for EFS (Table 3). The relatively low number of patients with AITL and the scarcity of patients with ALK-positive ALCL and IPI > 2 (n = 12) precluded a meaningful analysis of the role of IPI in these individual subgroups. Figure 5 and Figure 6 show the EFS and OS of patients with ALK-negative ALCL and PTCLU according to IPI and confirm its validity in these subgroups. The 3-year EFS or OS for all patients excluding ALK-positive ALCL was as follows: IPI low (58.2% [95% CI 49.0%-67.4%] and 73.1% [95% CI 64.7%-81.5%]); IPI low-intermediate (33.8% [95% CI 21.6%-46.0%] and 53.0% [95% CI 39.9%-66.1%]); IPI high-intermediate (31.6% [95% CI 16.9%-46.3%] and 39.2% [95% CI 22.9%-55.5%]); and IPI high (11.1% [95% CI 0.0%-22.9%] and 22.2% [95% CI 6.5%-37.9%]). The results obtained with the Cox model (Table 3) for these patients were very similar to those for the whole patient group including the ALK-positive ALCL patients. Notably, not only patients with ALK-positive ALCL but also patients from the other subtypes had a relatively good prognosis if the IPI was 0 or 1. The patients from all subtypes other than ALK-positive ALCL showed a poor prognosis (3-year EFS ≤ 34%) if the IPI was ≥ 2.
Event-free survival and overall survival of 320 patients with T-cell lymphoma according to the IPI.
Event-free survival and overall survival of 320 patients with T-cell lymphoma according to the IPI.
Event-free survival and overall survival of 113 patients with ALK-negative ALCL according to the IPI.
Event-free survival and overall survival of 113 patients with ALK-negative ALCL according to the IPI.
Event-free survival and overall survival of 70 patients with PTCLU according to the IPI.
Event-free survival and overall survival of 70 patients with PTCLU according to the IPI.
Discussion
There are few reports on the clinical outcome of large numbers of patients with T- or NK-cell lymphoma.1,19-24 The numbers become even smaller if studies are excluded which no longer reflect modern histopathologic classification and more important, do not consider key genetic features which significantly influence prognosis. For example, older series could not take into account the ALK-status which is significantly associated with the prognosis of ALCL patients in all recent studies.25 The remaining studies (including the International Peripheral T-Cell Lymphoma Project1 ) report surprisingly low numbers of patients even with some of the more frequent T-cell subtypes and the relative frequencies of patients with any of the major subtypes vary substantially. If these differences reflect the different geographic origin of patients, changes in diagnostic criteria over time, selection bias, or other unknown reasons remains elusive. In nearly all previous reports treatment was heterogeneous comprising a plethora of drugs and modalities from steroids to transplantation. Some of the recent publications were restricted to 1 or 2 T-cell lymphoma subtypes4,5,26-29 but in many instances focused on new pathologic, cytogenetic, or molecular findings, while treatment and prognostic factors were not analyzed in detail. By far the largest study1 reported recently came from 22 referral pathologic institutions worldwide and specifically addressed histopathologic findings and their reproducibility although clinical data, prognostic factors, and treatment outcomes are also described to some extent.
In this analysis we report on patients with 1 of the 4 major subtypes of T-cell lymphoma (ALK-positive ALCL, ALK-negative ALCL, AITL, and PTCLU) who all received treatment with CHOP or CHOP plus etoposide in prospective studies of the DSHNHL. This gave us the unique opportunity to analyze outcome of significant numbers of patients with World Health Organization-classified T-cell lymphoma under “standard” therapy and describe differences in EFS and OS dependent on variations of therapy and prognostic factors.
EFS and OS were good for patients with ALK-positive ALCL. Results were especially promising in our patients because 85% of them showed a low to low-intermediate IPI. For the other subtypes, prognosis significantly differed by IPI. Patients with IPI 0 and 1 showed 3-year EFS above 50%. Almost half of our patients with T-cell lymphoma excluding ALK-positive ALCL belonged to this low risk group (IPI 0,1) and their relatively favorable outcome should be kept in mind when selecting patients for new experimental strategies. In contrast, patients with IPI ≥ 2 showed a 3-year EFS below 34% and certainly are candidates for novel treatment approaches including autologous or allogeneic transplantation if age and performance status permit.30,31
We did not find a significant difference in EFS or OS between patients with ALK-negative ALCL, PTCLU, and AITL with 1 exception: the AITL patients did better than patients with ALK-negative ALCL. This finding should be taken with caution, however, because the number of patients with AITL included in DSHNHL studies was small. We cannot confirm that patients with ALK-negative ALCL do better than patients with PTCLU although the numbers of patients with these subtypes were comparable to those analyzed by Savage et al4 We hypothesize that different treatment strategies for patients from both studies contributed to these differences.
For younger patients, the addition of etoposide to CHOP improved response rates. The difference in EFS for younger patients and ALK-positive ALCL treated with CHOP or CHOEP within the DSHNHL trials is dramatic (3-year EFS for CHOEP patients 91.2% vs. 57.1% for patients treated with CHOP, P = .012), a statistically not significant difference was seen in the remaining patients when ALK-positive ALCL was excluded (3-year EFS for CHOEP patients 60.7% vs. 48.3% for patients treated with CHOP, P = .057). This represents an important finding because more T-cell lymphoma patients may be able to make it to autologous or allogeneic transplantation after CHOEP32 while both transplant modalities are actively pursued by many institutions to improve prognosis of T-cell lymphoma. Several other studies included etoposide in various chemotherapy regimens for T-cell lymphoma33,34 with promising results. A new study by the DSHNHL, Groupe d'Etudes des Lymphomes de l'Adulte, and Groupe Ouest Est d'Etude des Leucémies et Autres Maladies du Sang will use 4 courses of CHOEP-14 before patients receive DHAP (dexamethasone, cytarabine, and cisplatin) and autologous stem cell transplantation or allogeneic transplantation.
Interestingly, no treatment was significantly better than 6 courses of CHOP given every 3 weeks in patients older than 60 years. Although patient numbers for some comparisons were small and confirmation is desirable, for the time being, 6 courses of CHOP-21 should remain the standard therapy for elderly patients with T-cell lymphoma. Convincing evidence that chemotherapy regimens other than CHOEP,33,35 antibodies36,37 or new drugs38 improve outcome of noncutaneous T-cell lymphoma is scarce.
Unexpectedly, this study and another report from our group16 suggest that dose escalation of cyclophosphamide, doxorubicin, and etoposide administered with the CHOEP regimen up to doses necessitating autologous stem cell transplantation is unable to further improve treatment results. Similar findings were reported from the MD Anderson Cancer Center23 where investigators showed that more intense chemotherapy regimens like Hyper-CVAD (cyclophosphamide, vincristine, doxorubicin, and dexamethasone) failed to improve survival of patients with T-cell lymphoma compared with CHOP. On the contrary, the MegaCHOEP trial showed better EFS and OS for patients treated with 8 courses of standard-dose CHOEP (Figure 3). This finding calls into question whether HDT/autologous stem cell transplantation will really be able to improve outcome for younger patients with T-cell lymphoma. Results of randomized trials investigating this question are eagerly awaited.
In conclusion, we demonstrate that results of standard therapy with CHOP and particularly with CHOEP for patients with ALK-positive ALCL are excellent. Moreover, also patients with ALK-negative ALCL, PTCLU, or AITL do relatively well if the IPI is low (≤ 1). If ALK-positive ALCL is excluded, patients with IPI ≥ 2 show poor EFS and OS and new strategies to improve outcome for these patients are definitely needed. Although the difference in EFS between CHOP and CHOEP was not significant (P = .057) we suggest to administer CHOP plus etoposide to younger patients with T-cell lymphoma as first-line therapy because this may help to decrease the number of patients with early progression or relapse and bring more patients to transplantation. For patients beyond 60 years of age, 6 courses of CHOP-21 should remain the standard for first-line therapy, unless randomized studies demonstrate better outcomes with novel treatment modalities.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank Deutsche Krebshilfe for supporting all of the phase III trials, Kompetenznetz Maligne Lymphome (a research cluster funded by the German Federal Ministry of Education and Research) for providing study nurses for some of the participating institutions, and all colleagues and patients who participated. We thank Beate Mann for her excellent technical support.
Authorship
Contribution: N.S. was the principal investigator of the MegaCHOEP studies; M.P. was the chairman of the DSHNHL-B1, -B2, and RICOVER-60 studies; L.T. was the principal investigator of the Hi-CHOEP phase II and III studies; N.S., M.P., and M.Z. designed the study; N.S. wrote the manuscript; M.Z. and M.L. performed the statistical analysis; and A.R. is the coordinating reference pathologist of the DSHNHL.
Conflict-of-interest disclosure: M.P. and N.S. are members of Roche advisory boards. N.S., M.L., and M.P. have received research support from Roche. The remaining authors declare no competing financial interests.
Correspondence: Norbert Schmitz, Department of Hematology, Oncology and Stem Cell Transplantation, Asklepios Hospital St Georg, Lohmühlenstrasse 5, D-20099 Hamburg, Germany; e-mail: n.schmitz@asklepios.com.