Chronic GVHD is a major cause of morbidity and mortality in allogeneic stem cell transplantation recipients and typically develops from antecedent acute GVHD. In contrast to acute GVHD, chronic GVHD has much broader tissue involvement and clinical manifestations that bear striking similarity to what is observed in autoimmune diseases. How autoimmunity arises out of alloimmunity has been a longstanding unresolved issue. To address this question, in the present study, we performed a comprehensive analysis of the clonotypic T-cell response using complementary murine models that simulate what occurs during the transition from acute to chronic GVHD. These studies revealed repertoire skewing and the presence of high-frequency clonotypes that had undergone significant in vivo expansion, indicating that GVHD-associated autoimmunity was characterized by antigen-driven expansion of a limited number of T-cell clones. Furthermore, we observed that T cells with identical TCRβ CDR3 nucleotide sequences were capable of recognizing donor and host antigens, providing evidence that the loss of self-tolerance during acute GVHD leads to the emergence of self-reactive donor T cells that are capable of recognizing nonpolymorphic tissue or commensally derived antigens. These data provide a mechanistic framework for how autoimmunity develops within the context of preexisting GVHD and provide additional insight into the pathophysiology of chronic GVHD.
Introduction
GVHD is the primary complication associated with allogeneic stem cell transplantation and is the major cause of morbidity and mortality associated with this therapy.1,–3 GVHD has been classified into 2 phases that have been termed acute and chronic.4 Acute GVHD is a proinflammatory syndrome that is initiated by donor T-cell recognition of host antigens presented by host APCs5,–7 and generally targets a restricted set of organs (ie, the skin, liver, and gastrointestinal tract). Chronic GVHD is propagated by donor T-cell recognition of host peptides presented by donor APCs8 and is distinguished from acute GVHD in part by more widespread organ involvement and clinical manifestations that bear strong similarities to autoimmune disorders.9,10 In fact, the reclassification of chronic GVHD has brought into focus the similarities that this disease has with other autoimmune disorders.11 The major diagnostic criteria (ie, bronchiolitis obliterans, lichen planus, lichen sclerosus, and morphea) all have presumed autoimmune etiologies when encountered in non–stem cell–transplanted individuals.12,,,–16 Their corresponding appearance in GVHD recipients therefore supports the premise that autoimmunity is an integral component of chronic GVHD. However, how autoimmunity evolves from antecedent alloimmunity remains an unresolved issue.17
Studies in humans and in murine models have demonstrated that, during acute GVHD, there is a decrease in the relative and absolute number of regulatory T cells (Tregs).18,,–21 In animal models, this has been associated with the expansion of proinflammatory donor-derived Th1 and Th17 cells that are able to infiltrate GVHD target organs and lead to the development of autoimmune-mediated pathologic damage.22 Th1 cells have also been implicated as major effectors in human chronic GVHD based on data demonstrating the presence of T cells with expression of cytotoxic molecules in the oral lesions of patients with this disease.23 Recent studies have shown that CD4+ T-cell hybridomas generated from spleen cells obtained from mice with chronic GVHD can proliferate in vitro against donor and host cells,24 suggesting that these cells might be capable of responding to shared antigens in the donor and host. Prior work in humans has supported this premise by demonstrating that donor-derived CD4+ T cells that proliferate when exposed to donor antigens in vitro can be cloned from the peripheral blood of patients with chronic GVHD.25,26 These results have suggested that the loss of effective T-cell regulation that occurs during GVHD leads to the emergence of donor T cells that respond to shared antigens. However, there is no direct in vivo evidence that T cells can recognize nonpolymorphic antigens during GVHD. Moreover, whether GVHD-associated autoimmunity is characterized by repertoire skewing similar to what has been observed in acute GVHD is not known.27 To address these issues, in the present study, we used a murine BM transplantation model in which GVHD progression is characterized by the development of autoimmunity to perform an extensive analysis of the clonotypic T-cell response that occurs during the transition from acute to chronic GVHD.
Methods
Mice
C57BL/6 (B6; H-2b), Balb/cJ (H-2d), B6.129S7-Rag 1 (B6 Rag), and C.129S7-Rag 1 (Balb/c Rag) mice were bred in the Biomedical Resource Center at the Medical College of Wisconsin or purchased from The Jackson Laboratory. All animals were housed in the American Association for Laboratory Animal Care–accredited Biomedical Resource Center of the Medical College of Wisconsin. Experiments were all carried out under protocols approved by the Medical College of Wisconsin Institutional Animal Care and Use Committee. Mice received regular mouse chow and acidified tap water ad libitum.
Cell isolation
CD4+ T cells were isolated from the spleens of GVHD mice by positive selection using the MACS magnetic bead cell separation system (Miltenyi Biotec). CD4+CD25+ T cells were isolated using the MACS magnetic bead regulatory T-cell isolation kit according to the manufacturer's instructions. To isolate CD11c+ cells, pooled colons were incubated in HBSS buffer (Gibco-BRL) supplemented with 2% FBS, EDTA (0.05mM) and 15 μg/mL of DTT (Invitrogen) at 37°C for 30 minutes and subsequently digested in a solution of 1 mg/mL of collagenase D (Roche Diagnostics) in DMEM with 2% FBS for 75 minutes at 37°C. The resulting cell suspension was then layered on a 44%/67% Percoll gradient (Sigma-Aldrich). CD11c+ cells from liver and lung were isolated by collagenase D digestion, followed by layering on a Percoll gradient.
BM transplantation and adoptive transfer experiments
BM was flushed from donor femurs and tibias with DMEM and passed through sterile mesh filters to obtain single-cell suspensions. RBCs were removed from spleen cell suspensions by hypotonic lysis using distilled water. Host mice were conditioned with total body irradiation administered as a single exposure at a dose rate of 86 cGy using a Mark I cesium irradiator (Shepherd). Irradiated recipients received a single IV injection in the lateral tail vein (0.4 mL) of BM plus spleen or purified CD4+ T cells. Spleen cells or purified CD4+ T cells from GVHD mice 19-26 days after transplantation were injected intravenously into nonirradiated B6 Rag mice.
Generation of BM chimeras
Balb/c Rag mice were lethally irradiated (900 cGy) and transplanted with 10 × 106 B6 Rag BM cells on day 0. Mice were bled 2-3 months after transplantation to confirm complete donor cell engraftment before use in adoptive transfer experiments.
Histologic analysis
Representative samples of colon were obtained from mice and fixed in 10% neutral-buffered formalin. Samples were then embedded in paraffin, cut into 5 micron-thick sections, and stained with H&E. A semiquantitative scoring system was used to account for histologic changes in the colon, as described previously.22 All slides were coded and read in a blinded fashion. Images were visualized using a Nikon Eclipse E400 microscope and a Nikon Plan APO 10×/0.45 objective lens. Image acquisition was performed with a Zeiss Axiom camera and Axiovision Version 3.0.6 SP2 software (Zeiss).
TCRβ spectratype and CDR3 sequence analysis
TCRβ spectratyping was performed as described previously.28,,–31 Briefly, total RNA was prepared from colon samples by homogenizing the piece of colon (0.5 cm) in TRIzol reagent (Gibco-BRL). Then, 20 μg of total RNA was converted to cDNA using MMLV reverse transcriptase and poly-T primer. Rearrangement analysis was performed by PCR amplification of the CDR3 region for 21 Vβ families using Vβ and Cβ region-specific primers. The C-region primer was labeled with 5′ fluorescent 6-carboxyfluorescein or VIC. The labeled PCR products were run on an ABI 3100 genetic analyzer (Applied Biosystems). The fluorescence intensity of each fragment and the relative frequency of the fragments were obtained using Gene Mapper IDX Version 4.1 software (Applied Biosystems). The spectratype from 21 Vβ families were examined in all mice that received adoptive transfer of primary GVHD spleen cells or purified T-cell subsets. A Vβ family was defined as a family of interest if there was a focused immune response characterized by the presence of a dominant peak or set of peaks that was shared by the majority of animals within a given experiment. Peaks corresponding to a CDR3 length of less than 6 amino acids were considered nonfunctional.
Clonotyping was performed using the colony-counting technique. In summary, Vβ to Cβ PCR products of the family of interest were run on a 5% denaturing acrylamide gel and the fragment of interest was then cut from the gel. The DNA was recovered using an extraction kit (QIAGEN). The DNA was subjected to 8-10 rounds of Taq-mediated replication to generate double-stranded DNA that was cloned into Escherichia coli using the TOPO TA cloning kit (Invitrogen). Bacterial colonies were grown overnight and the picked colonies were sent to Agencourt Bioscience (Beckman Coulter) for sequencing. Sequences were analyzed using Finch TV and the clonotypes were designated using the nomenclature established by Yassai et al.32 The nomenclature of Wilson et al was used to define specific Vβ families.33
High-throughput 454 sequence analysis
High-throughput 454 sequencing was carried out using the Genome Sequencer FLX (GS-FLX) system (Roche Diagnostics) and the Amplicon sequencing method. Briefly, fusion primers were constructed by incorporating sequence-specific primers fused with 19 nucleotide GS-FLX primers for subsequent use in a clonal amplification and sequencing reaction. Fusion primers were tagged with unique 10-nucleotide ligation multiplex identifier (MID) tags to allow for discrimination of samples which shared the same Vβ family. cDNA was amplified using these fusion primers using the FastStart High Fidelity PCR System (Roche Applied Science). The PCR products obtained were then purified using AMPure beads (Beckman Coulter) and quantitated using the Quant IT PicoGreen dsDNA Assay kit (Invitrogen). Aliquots of the sample of interest were diluted to the appropriate DNA concentration in 1× TE buffer. Emulsion-based clonal amplification was then carried out using the GS-FLX titanium emPCR kit. After enrichment for amplicon double-stranded DNA, the multiplexed samples were sequenced using the Roche GS-FLX pyrosequencing system. Roche GS-FLX Version 2.3 analysis software was then used to demultiplex the samples based on the MID tags for each distinct sample and to produce quality filtered fasta-format sequence files for each sample. Two mismatches in the MID tag sequence were allowed. A tool was developed using Strawberry Perl to read and sort by Vβ family the large number of sequences. This program was validated and checked for any errors before reading the samples.
Statistics
Group comparisons of pathology scores were performed using the Mann- Whitney U test. P ≤ .05 was deemed to be significant in all experiments.
Results
Analysis of T-cell clonotypes in the colons of mice with GVHD-associated autoimmunity
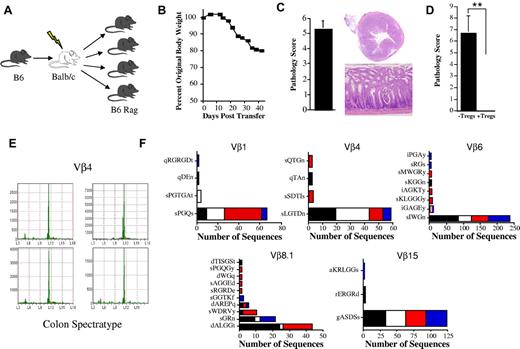
To determine the underlying mechanism by which autoimmunity emerges from antecedent alloreactivity, we used an established murine model of GVHD-associated autoimmunity22,34 (Figure 1A) to define the clonotypic T-cell response during the transition from acute to chronic GVHD. In this model, GVHD-associated autoimmunity is mediated by CD4+ T cells and is characterized by weight loss (Figure 1B) predominantly due to pathologic damage in the colon (Figure 1C). The cotransfer of CD4+ CD25+ Tregs completely abrogates the development of colitis (Figure 1D), indicating that autoimmunity is attributable to a loss of effective T-cell regulation that occurs during the evolution of acute GVHD. Similarly, the transfer of T cells from wild-type mice fails to result in autoimmunity, further supporting a break in tolerance during GVHD.34 TCRβ CDR3 spectratype analysis was performed on all 21 Vβ families to identify specific families in which focused high-intensity peaks were present within inflamed colons of replicate animals within a given experiment. Dominant focused peaks within a given Vβ family (Figure 1E and supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article) were then sequenced at the nucleotide level and the amino acid sequence was determined. For the purposes of this analysis, results are reported in terms of clonotypes that define a T-cell clone. Each clonotype represents the result of a productive VDJ rearrangement resulting in a unique CDR3 DNA sequence encompassing VDJ trimming and N-nucleotide addition.
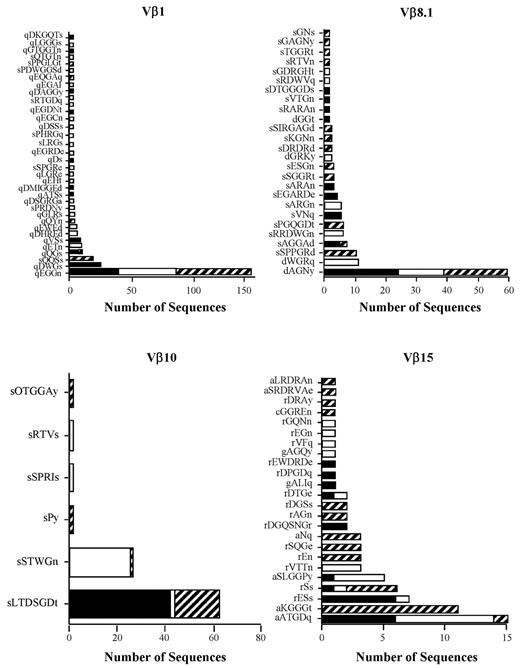
High-frequency clonotypes in mice with GVHD-associated autoimmune colitis. (A) Experimental approach used to examine the development of GVHD-associated autoimmunity. Lethally irradiated (900 cGy) Balb/c mice were transplanted with C57BL/6 (B6) BM (10 × 106) and 0.4 × 106 spleen cells to induce moderate GVHD. Animals were then euthanized 19-21 days after transplantation and pooled spleen cell suspensions from fully donor-engrafted mice adjusted to yield a T-cell dose of 0.5 × 106 were injected intravenously into nonirradiated B6 Rag animals. Mice were typically analyzed 60-70 days after transfer. (B) Serial weight curve after transfer of spleen cells from primary GVHD mice (n = 7). (C) Pathologic damage in the colon using a semiquantitative scoring system as detailed in the “Methods” section. The maximal pathologic score was 12. Histology of colon from a representative animal that received adoptive transfer of primary GVHD spleen cells showing extensive inflammation in the lamina propria, goblet cell depletion, and crypt cell destruction. Magnification is 20× for the upper panel and 100× for the lower panel. (D) Pathologic score in the colons of B6 Rag mice (n = 6-7 group) 60-70 days after transplantation that received spleen cells from primary B6 → Balb/c GVHD mice alone or together with purified CD4+ CD25+ Tregs from normal B6 animals. Data are presented as the means ± SEM. **P < .01. (E) Colons from replicate B6 Rag mice (n = 4) that received the same pooled spleen cells from GVHD animals were harvested and RNA was isolated for spectratype analysis. A representative spectratype showing dominant skewed peaks within the Vβ4 family is depicted. Arbitrary units reflecting peak intensity are on the y-axis, and CDR3 region nucleotide sequence length are on the x-axis. (F) Nonirradiated B6 Rag mice were adoptively transferred with pooled spleen cells from primary GVHD B6 → Balb/c chimeras that had undergone transplantation 19-21 days previously. Animals were euthanized 60-70 days after transfer and TCRβ spectratype and clonotype analysis was performed on processed colon tissue. The name and number of CDR3 region amino acid sequences derived from dominant peaks in specified Vβ families is depicted. Colors in bar graphs represent individual mice.
High-frequency clonotypes in mice with GVHD-associated autoimmune colitis. (A) Experimental approach used to examine the development of GVHD-associated autoimmunity. Lethally irradiated (900 cGy) Balb/c mice were transplanted with C57BL/6 (B6) BM (10 × 106) and 0.4 × 106 spleen cells to induce moderate GVHD. Animals were then euthanized 19-21 days after transplantation and pooled spleen cell suspensions from fully donor-engrafted mice adjusted to yield a T-cell dose of 0.5 × 106 were injected intravenously into nonirradiated B6 Rag animals. Mice were typically analyzed 60-70 days after transfer. (B) Serial weight curve after transfer of spleen cells from primary GVHD mice (n = 7). (C) Pathologic damage in the colon using a semiquantitative scoring system as detailed in the “Methods” section. The maximal pathologic score was 12. Histology of colon from a representative animal that received adoptive transfer of primary GVHD spleen cells showing extensive inflammation in the lamina propria, goblet cell depletion, and crypt cell destruction. Magnification is 20× for the upper panel and 100× for the lower panel. (D) Pathologic score in the colons of B6 Rag mice (n = 6-7 group) 60-70 days after transplantation that received spleen cells from primary B6 → Balb/c GVHD mice alone or together with purified CD4+ CD25+ Tregs from normal B6 animals. Data are presented as the means ± SEM. **P < .01. (E) Colons from replicate B6 Rag mice (n = 4) that received the same pooled spleen cells from GVHD animals were harvested and RNA was isolated for spectratype analysis. A representative spectratype showing dominant skewed peaks within the Vβ4 family is depicted. Arbitrary units reflecting peak intensity are on the y-axis, and CDR3 region nucleotide sequence length are on the x-axis. (F) Nonirradiated B6 Rag mice were adoptively transferred with pooled spleen cells from primary GVHD B6 → Balb/c chimeras that had undergone transplantation 19-21 days previously. Animals were euthanized 60-70 days after transfer and TCRβ spectratype and clonotype analysis was performed on processed colon tissue. The name and number of CDR3 region amino acid sequences derived from dominant peaks in specified Vβ families is depicted. Colors in bar graphs represent individual mice.
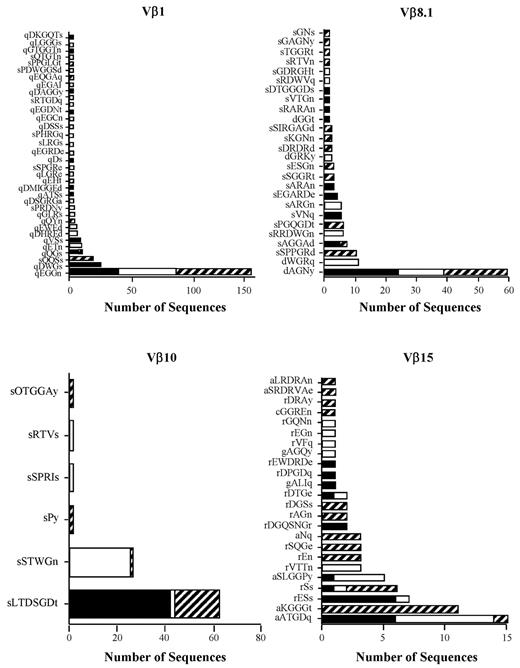
The primary observation was the presence of a dominant clonotype or set of high-ranking clonotypes that were present in all mice within a given experiment except Vβ8.1, in which 2 clonotypes were each present in 3 of 4 animals (Figure 1F). A second, less common pattern was the presence of occasional higher-frequency clonotypes that were not shared by individual mice within the same experiment (supplemental Figure 1 Vβ5.2 and Vβ6), although the majority of dominant clonotypes were still shared by multiple animals. Transfer of purified CD4+ T cells, which are the primary effector T-cell population in this model,22 yielded the same results as observed with unseparated T cells with respect to the presence of dominant clonotypes shared by multiple animals (Figure 2). When all experiments were examined in aggregate, there were a total of 2036 sequences that yielded 174 unique clonotypes in 13 Vβ families (Table 1). A relatively small number of high-frequency clonotypes comprised 50%-90% of the total identified in pathologically involved tissues. Because of the presence of these clonotypes, the percentage of overlapping sequences (range, 52%-94%) was much greater that the percentage of overlapping clonotypes (range, 11%-31%). None of the sequences present in replicate animals within a given experiment was detected in any other experiment (Table 2), with the exception of one sequence (sIWGn, Expt. 1) that was found twice in 1 mouse in a separate experiment. These results showed that GVHD-associated autoimmunity was characterized by repertoire skewing attributable to high-frequency clonotypes.
Dominant high-frequency clonotypes are present in CD4+ T cell–mediated autoimmunity. Lethally irradiated Balb/c mice (n = 10) were transplanted with B6 BM and 0.4 × 106 spleen cells. Animals were then euthanized 22 days after transplantation. CD4+ T cells (0.75 × 106) were purified from pooled spleen cell suspensions and then injected intravenously into nonirradiated B6 Rag animals (n = 3). CDR3 region amino acid sequences derived from specified Vβ families are depicted for individual mice. Colors/patterns in stacked bar graphs denote individual animals.
Dominant high-frequency clonotypes are present in CD4+ T cell–mediated autoimmunity. Lethally irradiated Balb/c mice (n = 10) were transplanted with B6 BM and 0.4 × 106 spleen cells. Animals were then euthanized 22 days after transplantation. CD4+ T cells (0.75 × 106) were purified from pooled spleen cell suspensions and then injected intravenously into nonirradiated B6 Rag animals (n = 3). CDR3 region amino acid sequences derived from specified Vβ families are depicted for individual mice. Colors/patterns in stacked bar graphs denote individual animals.
Expansion of low-frequency clonotypes during autoimmunity
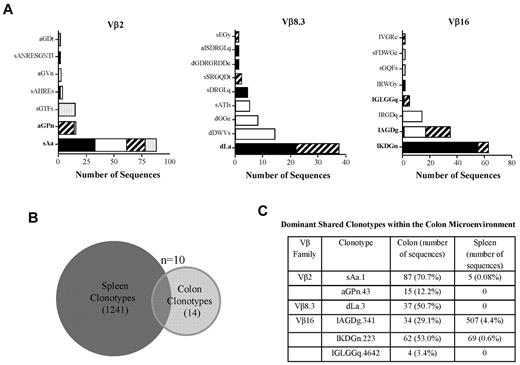
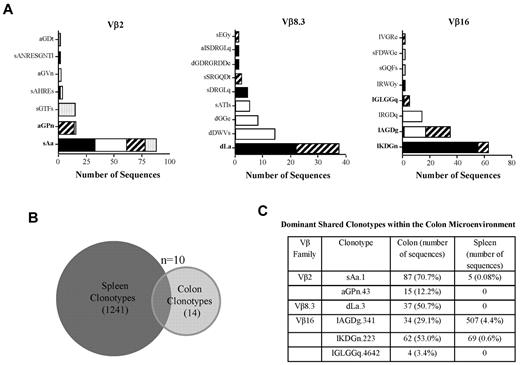
The presence of dominant clonotypes in selected families from the colons of replicate animals could have been either a reflection of existing frequency distributions present in the input splenic T-cell inoculum or the result of the enrichment and expansion of selected clonotypes after transfer into immunodeficient animals. To distinguish between these 2 possibilities, we compared the clonotypes present in the input splenic T-cell population with those present in dominant focused bands from selected Vβ families in the colons of mice with GVHD-associated autoimmunity. As shown in Figure 1, the most abundant clonotypes from colitic mice were shared by multiple animals within this cohort (Figure 3A). A total of 313 sequences representing 24 clonotypes were recovered from the colons of these animals (Table 3). We then performed high-throughput sequencing of an aliquot of pooled spleen cells that had been transferred into these mice to provide a more comprehensive analysis of the TCRβ CDR3 clonotype frequency distribution of these cells. A total of 18 312 sequences were obtained from 3 Vβ families (ie, Vβ2, Vβ8.3, and Vβ16), which constituted the families in which focused peaks were observed in the colons from replicate animals (Figure 3A). There were a total of 1251 unique clonotypes as defined by CDR3 region nucleotide sequence derived from these 3 families. Ten of these (0.8%) were identified within the colon (Figure 3B). We examined the 6 most abundant colon clonotypes that were present in more than one animal to determine the frequency of these clonotypes in the transferred spleen cell population (Figure 3C and Table 3). These 6 clonotypes represented 76% (239 of 313) of all recovered sequences from the colon. Three of these 6 clonotypes were not detected at all in the spleen, but represented 18% of total sequences (56 of 313) in the colon. The remaining 3 that were present in the spleen represented 0.08-4.4% of total clonotypes in the respective Vβ families. Within the colon, however, these T cells were present in frequencies that were 8- to > 800-fold higher than in the spleen and represented 29%-71% of all clonotypes recovered within the specific Vβ families. Conversely, there were high-frequency clonotypes present in the spleen, some of which represented more than > 15% of all sequences, which were not detected at all in the colons of mice (Table 4). In fact, only 1 of the 30 most abundant clonotypes from the 3 Vβ families that were found within the spleen was present within the colon. These data were consistent with the interpretation that there had been retention and subsequent expansion of low-frequency clonotypes within the colon microenvironment.
GVHD-associated autoimmunity is due to antigen-driven expansion. (A) Lethally irradiated Balb/c mice (n = 7) were transplanted with B6 BM and spleen cells to induce GVHD. Animals were then euthanized 18 days after transplantation and pooled spleen cell suspensions adjusted to yield a T-cell dose of 0.5 × 106 were injected intravenously into nonirradiated B6 Rag animals. After 60 days, animals were euthanized and TCRβ spectratype and clonotype analysis was performed on processed colon tissue from each individual mouse. The name and number of CDR3 region amino acid sequences derived from dominant peaks in specified Vβ families is depicted. Colors/patterns in bar graphs represent individual mice. Clonotypes in bold denote shared clonotypes between multiple animals. (B) Venn diagram showing the distribution of unique and overlapping clonotypes derived from the input splenic T-cell population and the colons of animals that received the transferred T cells. (C) Table denoting the 6 most frequent clonotypes in the colon that were shared between multiple mice. The number of sequences in the spleen and colon and the percentage of total sequences within a given Vβ family that these sequences represented are depicted.
GVHD-associated autoimmunity is due to antigen-driven expansion. (A) Lethally irradiated Balb/c mice (n = 7) were transplanted with B6 BM and spleen cells to induce GVHD. Animals were then euthanized 18 days after transplantation and pooled spleen cell suspensions adjusted to yield a T-cell dose of 0.5 × 106 were injected intravenously into nonirradiated B6 Rag animals. After 60 days, animals were euthanized and TCRβ spectratype and clonotype analysis was performed on processed colon tissue from each individual mouse. The name and number of CDR3 region amino acid sequences derived from dominant peaks in specified Vβ families is depicted. Colors/patterns in bar graphs represent individual mice. Clonotypes in bold denote shared clonotypes between multiple animals. (B) Venn diagram showing the distribution of unique and overlapping clonotypes derived from the input splenic T-cell population and the colons of animals that received the transferred T cells. (C) Table denoting the 6 most frequent clonotypes in the colon that were shared between multiple mice. The number of sequences in the spleen and colon and the percentage of total sequences within a given Vβ family that these sequences represented are depicted.
Dominant clonotypes are present in colitis mediated through the indirect alloreactive pathway
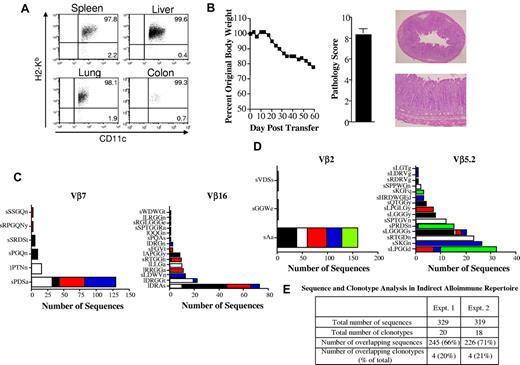
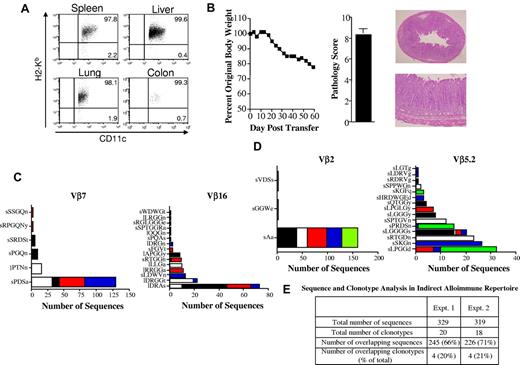
Within the context of autoimmunity that develops as a consequence of GVHD in humans, donor T cells respond to host antigens presented in the context of donor APCs or what has been termed the indirect pathway of allorecognition. This is because of the fact that autoimmune manifestations occur later in the course of GVHD after host APCs have been eliminated and donor APCs have reconstituted the hematopoietic compartment.35,36 Therefore, we performed a similar analysis to examine the T-cell clonotypic response within the colon under conditions in which allorecognition was through the indirect pathway. B6 Rag BM → Balb Rag chimeric mice were created so that recognition of Balb host antigens would occur in the context of B6 donor APCs. To verify that APCs from chimeric mice were of donor origin, animals were killed 80 days after transplantation and CD11c+ cells from the spleen, liver, colon, and lung were phenotyped. These studies revealed that > 95% of CD11c+ cells from these tissue sites were of donor (B6) origin (Figure 4A). More than 99% of CD11c+ cells in the colon (which was the major site of pathologic damage) were donor derived. Transfer of spleen cells from primary GVHD animals resulted in weight loss and pathologic damage in the colon (Figure 4B), similar to what was observed in the autoimmune model. In subsequent experiments, spleen cells from mice with acute GVHD were transferred adoptively into chimeric (B6 Rag BM → Balb/c Rag) mice and clonotyping of identified Vβ families was performed within replicate mice. These studies revealed that the pattern observed in autoimmune animals was also seen in mice in which pathologic damage was mediated by indirect allorecognition (Figure 4C-D). Specifically, we observed that there was a limited number of clonotypes in each of the specific Vβ families and that 1 or 2 constituted the majority of identified sequences within replicate animals. Therefore, the percentage of overlapping sequences (66%-70%) was much higher than the percentage of overlapping clonotypes (20%-21%) because of the presence of these high-frequency clonotypes (Figure 4E).
Dominant clonotypes are present in colitis mediated through the indirect alloreactive pathway. (A) Lethally irradiated (900 cGy) Balb/c Rag mice (n = 4) were transplanted with 10 × 106 BM cells from B6 Rag animals. Representative dot plot showing the percentage of CD11c+ cells that were donor-derived (H-2Kb+) in specified tissue sites 80 days after transplantation is shown. (B) Lethally irradiated Balb/c mice were transplanted with B6 BM and 0.4 × 106 spleen cells. Animals were then euthanized 24-25 days after transplantation. Spleen cells (adjusted to yield a T-cell dose of 0.5 × 106) were pooled from mice and infused intravenously into nonirradiated B6 Rag BM → Balb Rag chimeric mice that had been reconstituted 60-70 days earlier. Serial weight curve and pathologic damage in the colon (n = 9) using a semiquantitative scoring system is depicted. Data represent cumulative results from 2 experiments. Histology of colon from a representative animal shows extensive inflammation in the lamina propria and loss of mucin. Magnification is 20× for the top panel and 100× for the bottom panel. (C-D) Chimeric B6 Rag BM → Balb/c Rag mice were adoptively transferred with pooled spleen cells from primary GVHD B6 → Balb/c animals (n = 10). Mice were killed 60-70 days after transfer and TCRβ spectratype and clonotype analysis were performed on processed colon tissue. The name and number of CDR3 region amino acid sequences derived from dominant peaks in specified Vβ families is depicted. Colors in bar graphs represent individual mice. Panels C and D are results from 2 independent experiments with 4-5 animals per experiment. (E) Table denoting the total number of sequences and clonotypes derived from colons of mice in experiments detailed in panels C and D.
Dominant clonotypes are present in colitis mediated through the indirect alloreactive pathway. (A) Lethally irradiated (900 cGy) Balb/c Rag mice (n = 4) were transplanted with 10 × 106 BM cells from B6 Rag animals. Representative dot plot showing the percentage of CD11c+ cells that were donor-derived (H-2Kb+) in specified tissue sites 80 days after transplantation is shown. (B) Lethally irradiated Balb/c mice were transplanted with B6 BM and 0.4 × 106 spleen cells. Animals were then euthanized 24-25 days after transplantation. Spleen cells (adjusted to yield a T-cell dose of 0.5 × 106) were pooled from mice and infused intravenously into nonirradiated B6 Rag BM → Balb Rag chimeric mice that had been reconstituted 60-70 days earlier. Serial weight curve and pathologic damage in the colon (n = 9) using a semiquantitative scoring system is depicted. Data represent cumulative results from 2 experiments. Histology of colon from a representative animal shows extensive inflammation in the lamina propria and loss of mucin. Magnification is 20× for the top panel and 100× for the bottom panel. (C-D) Chimeric B6 Rag BM → Balb/c Rag mice were adoptively transferred with pooled spleen cells from primary GVHD B6 → Balb/c animals (n = 10). Mice were killed 60-70 days after transfer and TCRβ spectratype and clonotype analysis were performed on processed colon tissue. The name and number of CDR3 region amino acid sequences derived from dominant peaks in specified Vβ families is depicted. Colors in bar graphs represent individual mice. Panels C and D are results from 2 independent experiments with 4-5 animals per experiment. (E) Table denoting the total number of sequences and clonotypes derived from colons of mice in experiments detailed in panels C and D.
Identical TCRβ CDR3 clonotypes can be identified in autoimmune and indirect alloimmune repertoires
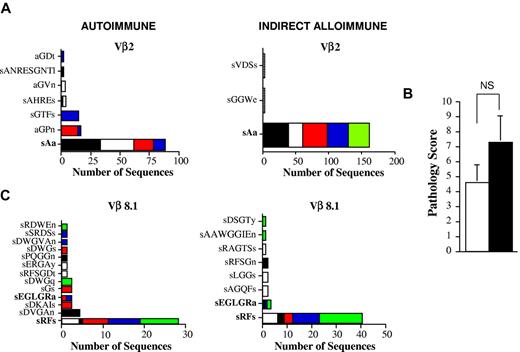
The observation that high-frequency clonotypes predominated under conditions of both autoimmunity and indirect alloimmunity led us to consider whether T cells with identical clonotypes could be identified in these 2 models. To address this issue, we first performed a comparative analysis of all of the clonotypes that we had detected in the autoimmune and indirect alloimmune repertoires from multiple prior independent experiments. We observed that there was one dominant clonotype that was present in both autoimmune mice and in chimeric animals in which alloreactivity was mediated through the indirect pathway. This clonotype, which was present in a total of 9 mice (5 in the autoimmune and 4 in the alloimmune cohorts), was from the Vβ2 family, and the CDR3 region encoded the amino acid sequence sAa (Figure 5A). Within the autoimmune and indirect alloimmune groups, this clonotype constituted 70% and 99% of all identified sequences in the Vβ2 family, respectively. In the autoimmune cohort, this clonotype was detected at very low frequency in the input spleen cell population (0.08%; Table 3), indicating that this T-cell clone had undergone significant in vivo expansion after transfer into B6 Rag animals.
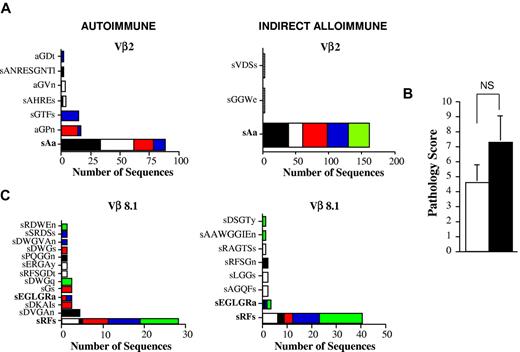
Donor T cells with identical TCRβ CDR3 nucleotide sequences can be identified in both autoimmune and indirect alloimmune repertoires. (A) Lethally irradiated Balb/c mice were transplanted with B6 BM and B6 spleen cells, and pooled spleen cells were then transferred by IV injection into B6 Rag animals. In a separate experiment, pooled spleen cells from acute GVHD mice were transferred into chimeric B6 Rag BM → Balb/c Rag mice. Vβ2 amino acid sequences derived from the colons of mice from both experiments are depicted. The amino acid sequence that was found in both cohorts of mice is shown in bold. (B) Lethally irradiated Balb/c mice (n = 10) were transplanted with B6 BM and B6 spleen cells. Pooled spleen cells from euthanized GVHD animals 26 days after transplantation were transferred by IV injection into either nonirradiated B6 Rag animals or B6 Rag BM → Balb/c Rag chimeric mice. Pathology scores for colon damage in B6 Rag and chimeric B6 Rag BM → Balb Rag mice (n = 5/group) are shown. NS indicates not significant. (C) Vβ8.1 amino acid sequences derived from the colons of mice. Amino acid sequences found in both cohorts of mice that received the same splenic T-cell inoculum are shown in bold.
Donor T cells with identical TCRβ CDR3 nucleotide sequences can be identified in both autoimmune and indirect alloimmune repertoires. (A) Lethally irradiated Balb/c mice were transplanted with B6 BM and B6 spleen cells, and pooled spleen cells were then transferred by IV injection into B6 Rag animals. In a separate experiment, pooled spleen cells from acute GVHD mice were transferred into chimeric B6 Rag BM → Balb/c Rag mice. Vβ2 amino acid sequences derived from the colons of mice from both experiments are depicted. The amino acid sequence that was found in both cohorts of mice is shown in bold. (B) Lethally irradiated Balb/c mice (n = 10) were transplanted with B6 BM and B6 spleen cells. Pooled spleen cells from euthanized GVHD animals 26 days after transplantation were transferred by IV injection into either nonirradiated B6 Rag animals or B6 Rag BM → Balb/c Rag chimeric mice. Pathology scores for colon damage in B6 Rag and chimeric B6 Rag BM → Balb Rag mice (n = 5/group) are shown. NS indicates not significant. (C) Vβ8.1 amino acid sequences derived from the colons of mice. Amino acid sequences found in both cohorts of mice that received the same splenic T-cell inoculum are shown in bold.
Based on these data, we sought additional confirmation as to whether identical clonotypes could emerge under conditions in which transferred T cells were exposed to different antigenic repertoires (ie, B6 vs Balb/c). We therefore performed a similar analysis with the exception that animals in both models received the same input splenic T-cell population. In these studies, spleen cells from primary B6 → Balb/c GVHD mice 3 weeks after transplantation were transferred into either B6 Rag or B6 Rag BM → Balb Rag chimeric mice. Animals in both cohorts were killed 2 months after transfer, and pathologic examination of the colons confirmed that there was no significant difference in the colitis scores between groups (Figure 5B). Spectratype analysis revealed that Vβ8.1 was similarly skewed in replicate animals from both cohorts, so this family was selected for further analysis. Sequencing of the CDR3 TCRβ region demonstrated the presence of 2 clonotypes that were present in both cohorts of animals (Figure 5C). The first, encoding the amino acid sRFs and the nucleotide sequence AGTAGGTTTTCT, was present in all mice and constituted 60% and 77% of all recovered sequences in the autoimmune and alloimmune Vβ8.1 repertoires, respectively. A second sequence encoding the amino acid sEGLGRa was also observed, but was present in only a subset of animals and constituted a smaller percentage (4%-6%) of overall sequences. These data indicated that T cells with identical TCRβ CDR3 sequences were present in 2 different cohorts of mice in which immune reactivity was directed against both donor and host antigenic repertoires, respectively.
Discussion
Chronic GVHD is an evolving entity that has undergone reclassification in recent years in an effort to more clearly distinguish it from the clinical manifestations associated with acute GVHD. What has developed during this process is the awareness that autoimmune manifestations are an integral component of the syndrome.9,–11 Moreover, a link between autoimmunity and a relative absence of effective Treg reconstitution has been strengthened by recent studies in humans showing that a decline in the frequency of Tregs is associated with a higher incidence of extensive chronic GVHD.21 Furthermore, the observation that the infusion of low-dose IL-2 to patients with refractory chronic GVHD is able to both increase Treg numbers and simultaneously ameliorate clinical manifestations has provided compelling support for the loss of self-tolerance as a primary mechanism for the induction and progression of this disease.37 However, how the breaking of self-tolerance and the emergence of autoimmunity arises from antecedent alloimmunity has remained elusive.
To address this issue, we reasoned that examination of the clonotypic T-cell response using an experimental BM transplantation model that simulated the transition from acute to chronic GVHD would allow us to critically dissect the autoimmune and alloimmune repertoires. We first analyzed T-cell clonotypes derived from mice with autoimmune-mediated colitis that received adoptively transferred donor-engrafted cells from animals with acute GVHD. We reproducibly observed a high frequency of the same T-cell clonotypes in replicate animals from individual experiments. This was noteworthy because it differs from what has been reported in murine models of inflammatory bowel disease in which naive CD4+ CD45RBhi T cells are transferred into immunodeficient animals. In these studies, there has either been no evidence of oligoclonal expansion38 or oligoclonal expansion has been characterized by very few shared nucleotide sequences39 in mice that received the same donor pool of transferred CD4+ T cells. A notable difference, however, is that transferred T cells in the present study were obtained from the spleens of mice with acute GVHD. Acute GVHD is known to induce repertoire skewing in the spleen27,31 and therefore results in a restriction of overall diversity compared with a wild-type TCRβ repertoire, which is very heterogeneous and characterized by a low frequency of T cells bearing the same TCR.40 Therefore, this preexisting limitation in overall diversity may have been a predisposing factor for the subsequent outgrowth of high-frequency dominant clonotypes. A similar observation has been made in mice in which TCR diversity is restricted because of genetic modification,41 raising the question of whether T-cell repertoire skewing during acute GVHD is a contributing factor in the subsequent development of autoreactive pathogenic T cells.
Whereas we observed that replicate mice often had the same clonotypes detected in the colon microenvironment, there was essentially no clonotypic overlap observed between experiments. The fact that clonotypes that expanded in replicate mice tended to be unique within a given experiment supports the premise that autoimmunity during chronic GVHD is a stochastic process whereby T cells with different specificities are capable of responding to self-antigens in different tissue sites. This interpretation is consistent with previous data showing that autoimmunity is attributable to the progressive loss of Tregs during acute GVHD, which leads to the emergence of proinflammatory, donor-derived T cells.18,,,–22 The absence of Tregs would be predicted to unleash autoreactive T cells in a random fashion, which is what has been observed in Foxp3-deficient animals in which the absence of Tregs results in widespread, nonselective T-cell activation and subsequent organ pathology.42,43 When extrapolated to a clinical scenario, replicate mice within a given experiment are synonymous with a single patient. Therefore, one can predict that T-cell clonotypes in involved organs would differ between patients because they receive stem cell grafts from different donors and possess different antigenic repertoires. This would be a potential explanation for the variability of clinical manifestations observed in patients with chronic GVHD, in whom different tissue sites can be involved in different patients.
We considered that the observation that dominant high-frequency clonotypes were present in replicate animals was either a reflection of underlying frequency distributions in the input T-cell population or was attributable to the in vivo expansion of lower-frequency clonotypes as a consequence of antigenic exposure. To distinguish between these 2 possibilities, we performed high-throughput sequence analysis of clonotype frequencies in the input spleen cell population. This analysis revealed that some clonotypes that constituted a majority of those found in the colon were not detected at all within the spleen, whereas, in other cases, they were present only at very low frequencies. In the latter instance, there was a marked disparity between the frequency in the spleen and that within the colon microenvironment (Table 3). In fact, we observed a greater than 800-fold increased frequency for at least one clonotype after transfer into recipient animals. This marked disparity was evidence that significant in vivo expansion had occurred in pathologically involved colon tissue. Furthermore, these data provided strong support for the conclusion that GVHD-associated autoimmunity was attributable to the antigen-driven expansion of a limited number of T-cell clones and was not merely the consequence of a nonselective, lymphopenia-induced homeostatic expansion.
In autologous GVHD in humans induced by the administration of cyclosporine, autoreactive T cells have been identified that have restricted diversity and are able to respond to a nonpolymorphic self-antigen.44,45 However, during allogeneic chronic GVHD, it is not clear whether the observed autoimmune manifestations are because of ongoing recognition of polymorphic antigens that are unique to the host or if nonpolymorphic antigens expressed by both donor and host can emerge under these conditions. Because this distinction is difficult to make in human patients, we used animal models to investigate the clonotypic T-cell response against self-antigens (autoimmune repertoire) compared with those directed against alloantigens (indirect alloimmune repertoire) to determine whether clonotypes that recognize both self- and alloantigens could be identified in recipient animals. In both cases, the APC compartment was donor derived, which is typically what is observed in patients with chronic GVHD.35,36 We observed that the same T-cell clonotypes could be detected in both experimental cohorts under conditions of autoreactivity and indirect alloreactivity. Two lines of evidence supported this conclusion. The first was the somewhat unexpected observation that an identical T-cell clone was present in separate experiments in which mice received different T-cell inputs. This T-cell clone, which derived from the Vβ2 family, had the CDR3 TCRβ region sequence sAa and constituted the dominant clonotype in both cohorts of mice. It has been estimated that there are 5-8 × 106 unique TCRβ nucleotide sequences per spleen.46 Therefore, the probability of this observation occurring by chance alone in multiple animals is low and supports a common antigen as the underlying stimulus for their presence. However, because mice received different T-cell inputs, we performed a more definitive analysis in which animals in both cohorts received the same splenic T-cell population from acute GVHD mice. In these studies, we observed 2 separate T-cell clonotypes that were derived from the Vβ8.1 family and were present in B6 Rag and B6 Rag BM → Balb Rag chimeric mice. The sequence sRFs in particular was not only present in all mice, but constituted the dominant clonotype in this analysis. From these results, we conclude that the presence of identical T-cell clones emerging in recipients differing only in the origin of their antigenic repertoire supports the premise that these T cells were responding to a nonpolymorphic antigen.
These studies do not define the nature of the antigen recognized by autoreactive T cells. Because the colon was the site of pathology in these studies, one possibility is that T cells responded to a commensally derived antigen that in effect functions as a self-antigen within the mouse colon microenvironment.47,–49 Models of inflammatory bowel disease in mice have shown that colitis occurs at much higher frequency and penetrance when mice are housed in conventional as opposed to germ-free conditions,50,51 implicating a role for the microbiome in the pathogenesis of mucosal inflammation. Given that animals that are housed together can have similar microbiomes in the gastrointestinal tract,52 it is formally possible that a commensally derived antigen could serve as the stimulus for T-cell reactivity in both experimental cohorts. A second possibility is that T cells could respond to a tissue-specific antigen to which tolerance is broken in the absence of effective T-cell regulation. Under the current experimental setting, this would be an epithelial-derived antigen that is resident in the colon. This would be perhaps a more plausible explanation for the autoimmunity that typically involves some of the other tissue sites (eg, mucous membranes, eyes, salivary glands, lung, skin, and fascia) in human patients with chronic GVHD, particularly because not all of these sites are directly exposed to the microbiome. However, given the varied clinical manifestations associated with chronic GVHD that encompass multiple tissues, it is reasonable to assume that there may be scenarios in which both mechanisms are operative.
The origin of autoreactive T cells in the present study was likely mature T cells that were transferred in the BM graft, given that transferred T cells were obtained from primary GVHD animals 19-21 days after transplantation, when thymic production of new T cells was negligible. Our studies therefore do not address directly the role of the thymus in the development of autoreactive T cells. However, several studies in mice have demonstrated that altered thymic function is another mechanism by which autoreactive T cells may emerge during GVHD.53,–55 This is thought to occur by the escape of autoreactive T cells from negative selection in a thymus that has been damaged because of chemoradiotherapy-based conditioning regimens, GVHD, and/or age-related involution. The relative importance of either pathway has not been resolved, but the fact that autoimmunity can arise in athymic animals56 indicates that there does not appear to be an obligate requirement for the thymus. In the clinical setting, one can make a plausible case that mature graft-derived T cells play some role in inducing autoimmunity given that thymic contributions to the T-cell repertoire are more limited in older subjects, who constitute the majority of patients undergoing allogeneic stem transplantation and who have the highest incidence of chronic GVHD.
In summary, the results of the present study demonstrate that the autoimmunity that occurs as a consequence of GVHD appears to be an antigen-driven response that results in repertoire skewing and the appearance of dominant high-frequency clonotypes that emerge because of the loss of effective T-cell regulation. The loss of self-tolerance also facilitates the emergence of donor T cells capable of recognizing nonpolymorphic tissue or commensally derived antigenic epitopes shared by both donor and host. Finally, these studies provide a mechanistic framework for how autoimmunity can arise within the context of antecedent alloimmunity and contribute additional insight into the pathophysiology of chronic GVHD, which may help in the development of new therapeutic strategies for this debilitating complication.
There is an Inside Blood commentary on this article in this issue.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Dr Calvin Williams for thoughtful comments on this manuscript.
This research was supported by grants from the National Institutes of Health (HL64603, HL081650, and DK083358) and by awards from the Midwest Athletes Against Childhood Cancer Fund.
National Institutes of Health
Authorship
Contribution: H.R. designed and performed research, analyzed data, and wrote the manuscript; M.Y., H.S., and M.W. performed research; R.K. performed pathologic analysis of all tissue samples; J.G. helped to design the experiments and interpret data; and W.R.D. designed the experiments, analyzed data, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: William R. Drobyski, MD, Bone Marrow Transplant Program, 9200 W Wisconsin Ave, Milwaukee, WI 53226; e-mail: wdrobysk@mcw.edu.