In this issue of Blood, Gadner et al provides data from the third Histiocyte Society trial (LCH III).1
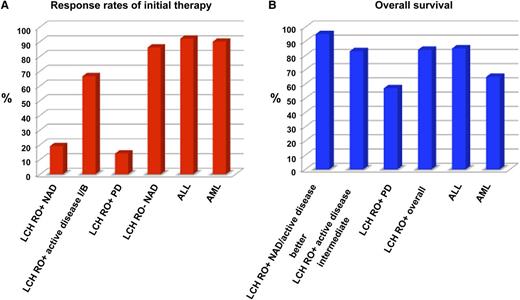
(A) Response rates of initial therapy of LCH compared with childhood ALL and AML. (B) Overall 5-year survival percentages. RO+ is defined as patients with LCH that involves risk organs, whereas RO– defines patients with multisystem disease without risk organ involvement (ie, liver, spleen, hematopoietic system, and lung). NAD, no active disease; Active Disease I/B, active disease is still present but has shown an intermediate response (unchanged disease or regression but with evidence of some new involvement) or is improved (ie, B for better); PD, progressive disease. Response rates to induction therapy and OS rates for ALL and AML for the time period of the LCH III trial were extracted from Pui et al.6
(A) Response rates of initial therapy of LCH compared with childhood ALL and AML. (B) Overall 5-year survival percentages. RO+ is defined as patients with LCH that involves risk organs, whereas RO– defines patients with multisystem disease without risk organ involvement (ie, liver, spleen, hematopoietic system, and lung). NAD, no active disease; Active Disease I/B, active disease is still present but has shown an intermediate response (unchanged disease or regression but with evidence of some new involvement) or is improved (ie, B for better); PD, progressive disease. Response rates to induction therapy and OS rates for ALL and AML for the time period of the LCH III trial were extracted from Pui et al.6
Langerhans cell histiocytosis (LCH), acute myelogenous leukemia (AML), and acute lymphoblastic leukemia (ALL) in children are all considered to be rare diseases based on the National Institutes of Health’s definition of a prevalence of fewer than 200 000 cases in the United States. LCH, AML, and ALL have estimated annual incidences of approximately 5, 7, and 20 cases per million children, respectively, and a combined prevalence estimated to be significantly less than the National Institutes of Health’s upper limit.2-4 The report by Gadner et al1 provides data from the LCH III.1 This trial represents a logical succession of the 2 previous trials over the course of 17 years. Because the results of LCH I (1991-1995) and LCH II (1996-2001) did not achieve the outcomes reported by the more intense DAL HX83/90 studies,5 as well as an increasing concern of the leukemogenic potential of etoposide, LCH III tested whether the addition of methotrexate for patients with multisystem and risk organ involvement improved outcomes. No significant differences in outcome were observed between the 2 treatment groups; 5 year overall survival (OS) for the entire study was 84%, with 27% reactivation. LCH III also randomized patients without risk organ involvement to either 6 or 12 months of maintenance therapy with vinblastine/prednisone, resulting in a significant decrease in disease reactivation (54% vs 37%, respectively).
These results are important for several reasons. First, methotrexate, as given in LCH III, did not improve outcome and was associated with significantly more grade III and IV toxicities. This result is somewhat surprising and disappointing because methotrexate appeared to play a prominent role in the DAL HX83/91 studies, where it was given not during the initial treatment phase but during maintenance at the 500-mg/m2 dose. In addition, OS appeared to show a significant improvement compared with the previous studies. Such comparisons are a flirtation with statistical incredulity. Another potential problem with the analyses is the intent-to-treat analysis used, as some patients were omitted because of inadequate data. Nevertheless, if indeed OS has improved over the 3 studies, 2 probable explanations would include improved treatment of patients who did not respond to up-front therapy as well as possible improvements in supportive care.
Further, several sobering numbers showed that only 19% of patients completing the initial 2 blocks of therapy had “no active disease (NAD),” whereas 66% still had active disease classified as either “better” or “intermediate” in response. Fourteen percent of patients showed progression of their disease during initial therapy. Thus, although OS appears to be improved compared with previous studies, there remains an appallingly high percentage of patients whose disease does not optimally respond to initial therapy with vinblastine plus prednisone with or without methotrexate. In contrast, comparative complete remission rates are approximately 85% to 90% for children with AML and >90% for those with ALL. Further, the OS of patients with LCH with risk organ involvement who achieved NAD or “active disease better” was 95% compared with 83% for those with “active disease intermediate” responses and 57% for those with “progressive disease.” Such percentages are in the range for patients with favorable-risk, intermediate-risk, and high-risk leukemia (see figure).4,6 These data make it clear that LCH is not a disorder that should be considered lightly by physicians or the public.
The demonstration that 12 months of maintenance therapy decreased the reactivation rate in patients with risk organ involvement (compared with historical controls), as well as in the prospective randomized question for patients without risk organ involvement, is a step in the right direction. A more careful look at these data shows a nonsignificant difference in the time to reactivation among those who reactivate, but only in the plateau of the percentage of reactivation episodes. Despite the overall decrease, disease reactivation rates remain high. In addition, no decrease in the incidence of diabetes insipidus was observed in any of the groups studied in LCH III or in comparison with prior studies. This suggests that the addition of methotrexate did not have an impact on this complication.
A reasonable question to ask is why so much effort should be given to reducing the recurrence of LCH when such recurrences are rarely life threatening and may often carry little perceived morbidity. Such reactivations of disease certainly do not have the obligate significance of relapsed leukemia. Notwithstanding, LCH disease recurrence has been shown to convey significant morbidity.7 Further, inconvenience and sometimes life-threatening consequences of diabetes insipidus represent a substantial burden.
The identification of BRAF1 mutations in LCH has provided the potential opportunity to bring the testing of more specifically targeted agents in patients, similar to what is now occurring in the acute leukemias.8,9 In particular, clinical testing of BRAF1 inhibitors, alone or in combination with MEK inhibitors as in melanoma, as well as to cytokine-mediated pathways and immunologically selective targets, should be moved rapidly into clinical trials with the intention of improving both initial and prolonged responses.
In spite of the positive outcomes of the international LCH trials, a significant failure has been the lack of biological banking of tumor and germ line samples, which has been so effectively done for pediatric cancers through the cooperative groups. Future trials have an obligation to patients to assure that such banking is accomplished and samples made available to the international research community. In this era of genomics, a disease being “rare” is not an excuse for not ensuring detailed analysis of samples or not performing definitive clinical trials.
Two final points should be noted. First, it must be acknowledged that no international trial has been in effect since 2008, but LCH IV, just now moving forward, will attempt to address the issues of early mortality and further reduction in disease reactivation. Although sometimes either advisable and/or unavoidable, such gaps may miss important opportunities. Second, the immense progress made by the international trials has focused on children and not adults. Adults have become the true orphans of this orphan disease.
Conflict-of-interest disclosure: The authors declare no competing financial interests.