Abstract
Background: Current prognostication in primary myelofibrosis (PMF) utilizes international prognostic scoring systems that rely on clinical parameters that are sensitive to day-to-day variations and subjective interpretation. Recent studies in PMF have disclosed important prognostic information attached to additional cytogenetic details (Blood. 2011;118:4595) and somatic mutations, including CALR and ASXL1 (NEJM. 2013;369:2379; Leukemia. 2013;27:1861).
Methods: PMF diagnosis and definition of blast transformation (BT) were according to World Health Organization criteria (Blood. 2009;114:937). Cytogenetic analysis and reporting was done according to the International System for Human Cytogenetic Nomenclature (Cytogenetic and genome research. 2013. Prepublished on 2013/07/03 as DOI 10.1159/000353118). Previously published methods were used for analyses of CALR, JAK2, MPL and other prognostically-relevant mutations, including ASXL1, SRSF2, EZH2 and IDH(Leukemia. 2014;28:1472).
Results: The training set included 964 Mayo Clinic patients (median age 65 years; 62% males) in whom informative karyotype or mutation information was available; cytogenetic information was available in 903 (94%) cases, JAK2/CALR/MPL mutational status in 532 (55%), ASXL1 in 425 (44%), SRSF2 in 434 (45%), IDH1/2 in 376 (39%) and EZH2 in 268 (28%). DIPSS-plus (JCO. 2011;29:392) risk distribution was high in 37% of patients, intermediate-2 in 37%, intermediate-1 in 15% and low in 11%.
We used a revised risk stratification for cytogenetics (see accompanying ASH 2014 abstract) to distinguish four distinct cytogenetic risk categories: very high (monosomal karyotype, inv(3), i(17q), -7/7q-, 11q or 12p abnormalities; n=67), high (complex non-monosomal, two abnormalities not included in very high risk category, 5q-, +8, other autosomal trisomies except +9, and other sole abnormalities not included in other risk categories; n=164), intermediate (sole abnormalities of 20q-, 1q+ or any other sole translocation, and -Y or other sex chromosome abnormality; n=133) and low (normal or sole abnormalities of 13q- or +9; n=539).
Mutational frequencies were 58% for JAK2, 25% CALR, 7% MPL, 36% ASXL1, 11% SRSF2, 5% IDH1/2 and 6% EZH2. The 131 cases with CALR mutations were further subclassified into two prognostically different groups: type 1/type 1-like (n=110) and type 2/type 2-like (n=21) (see accompanying ASH 2014 abstract).
At a median follow-up time of 4.2 years for patients who are alive, 664 (69%) deaths and 70 BT (7%) were recorded. Age-adjusted multivariable analysis that included cytogenetic and mutational risk groups disclosed the following as independent predictors of shortened survival: very high risk karyotype (HR 4.2; 3 points), high risk karyotype (HR 1.9; 1 point), triple-negative (HR 2.8; 2 points), JAK2 (HR 3.1; 2 points), MPL (HR 3.1; 2 points), type 2/type 2-like CALR (HR 3.6; 2 points), ASXL1 (HR 1.9; 1 points) and SRSF2 (HR 1.9; 1 point); EZH2 (p=0.24) and IDH1/2 (p=0.68) and intermediate risk karyotype (p=0.87) were not significant.
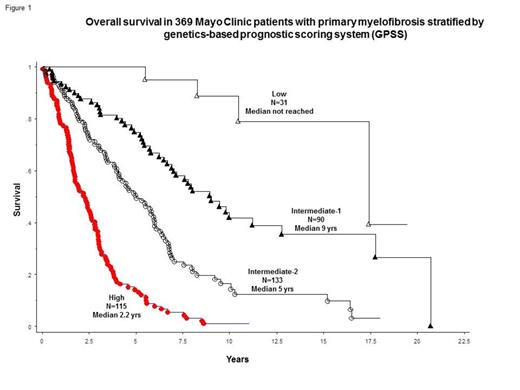
The above-mentioned significant variables and age demarcated at 60 years (2 points), were subsequently used to develop an HR-derived, genetics-based prognostic scoring system (GPSS) for 369 patients who were fully informative for both karyotype and all significant mutations: low risk (0 points; n=31), intermediate-1 (1 or 2 points; n=90), intermediate-2 (3 or 4 points; n=133) and high (5 or more points; n=115); the corresponding median survivals were >17, 9 (HR 4.7, 95% CI 1.7-13.0), 5 (HR 10.7, 95% CI 3.9-29.3) and 2.2 (HR 29.2, 95% CI 10.6-80.0) years (Figure 1). High risk GPSS was also associated with higher BT rate (HR 7.4, 95% CI 2.1-26.3).
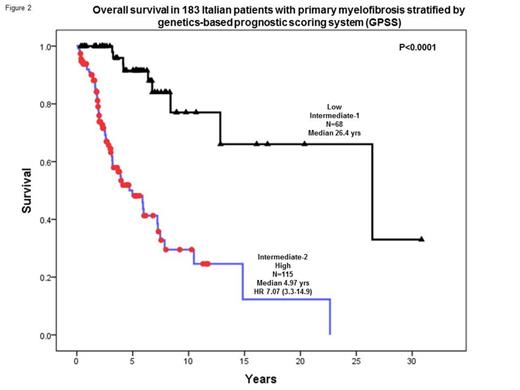
The prognostic distinction between high/intermediate-2 and low/intermediate-1 risk GPSS, in terms of both overall (median 5 vs 26.4 years; HR 7.1, 95% CI 3.3-14.9) and leukemia-free survival (median 11.6 years vs not reached; HR 9.4, 95% CI 2.2-41.0) was validated in an independent cohort of 183 patients from the University of Florence (Figure 2).
Conclusions: The current study demonstrates the feasibility of genetics-based prognostic models in PMF that rely on objective parameters that are amenable to further refinement as new genetic information becomes available.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.