Abstract
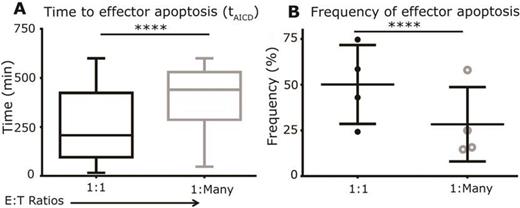
T cells genetically modified to express a CD19-specific chimeric antigen receptor (CAR) for the investigational treatment of B-cell malignancies comprise a heterogeneous population, and their ability to persist and participate in serial killing of tumor cells is a predictor of therapeutic success. We developed Timelapse Imaging Microscopy In Nanowell Grids (TIMING) to dynamically analyze thousands of interactions between T cells and tumor cells. We showed that the ability of killer CAR+ T cells to adaptively modify both cell-intrinsic (polarization and motility) and extrinsic (conjugation and kinetics of tumor-cell death) behavior was modulated by number of tumor cell encounters. Our results further demonstrate that while CAR+ T cells launch fully-competent anti-tumor responses as defined by polarized motility, serial-killing and IFN-γ secretion, the cytolytic functionality of CAR+ T cells must be quantified in the context of their ability to resist apoptosis. By comparing both the serial killer and single killer CAR+ T cells it appears that the propensity and kinetics of CAR+ T cells apoptosis was markedly different: single target killer effectors underwent apoptosis at higher frequencies (50%), than serial killers (28%) and with faster kinetics (time to apoptosis: 256±12 minutes, single target; vs. 396±25 minutes, multiple targets; p-value < 0.0001) (Figure 1 ). These data indicate that activation for lysis through multiple targets as opposed to prolonged contact with a single target reduces the propensity for effector apoptosis. Although the mechanistic basis for the responsiveness of these T cells to antigen/target density is not known, it is conceivable that the continuous propagation of these cells on irradiated aAPC at defined ratios, allows for balanced activation and minimized AICD. These data could provide mechanistic insights into observations that infused CAR+ T cells swell in number and produce high levels of cytokine in response to addressing large numbers of CD19+ tumor cells, but then decline in number as the tumor burden is lowered due to the serial killing by effector T cells. Our findings emphasize the notion that motility and persistence (resistance to apoptosis), in addition to direct cytolytic activity, may serve as criteria to rapidly evaluate CAR+ T-cell potency.
In order to further define functional CAR+ T cells at the single-cell level, we identified individual killer CAR+ T cells based on their interaction with CD19-expressing EL4 tumor cells. These killer CAR+ T cells were retrieved and subjected to single-cell gene expression profiling. The expression profiles were then fitted using a logistic regression model with an implicit gene selector function to identify sets of candidate genes that uniquely distinguish (non-apoptotic) killers and non-killers. We anticipate that our findings and the TIMING methodology may provide a rapid approach for assessing the impact of CAR designs and sub-populations of cells modified to redirect specificity.
Frequency and kinetics of effector apoptosis are dependent on functional activation provided by varying numbers of tumor cells. (A) Box and whisker plots (extremities indicate range) of the distribution of time to AICD of single CAR+ T cells after target cell killing. (B) Donor variation in the average frequencies of apoptotic killer CAR+ T cells. P-values were computed using an unpaired two-tailed t-test (n=4).
Frequency and kinetics of effector apoptosis are dependent on functional activation provided by varying numbers of tumor cells. (A) Box and whisker plots (extremities indicate range) of the distribution of time to AICD of single CAR+ T cells after target cell killing. (B) Donor variation in the average frequencies of apoptotic killer CAR+ T cells. P-values were computed using an unpaired two-tailed t-test (n=4).
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.