Abstract
Acute Lymphoblastic Leukemia (ALL) is the most common malignancy observed in the pediatric population. Recent genomic studies have made considerable strides in improving our understanding of the molecular pathogenesis of pediatric B-cell ALL. This includes the discovery of recurrent somatic alterations targeting B-cell transcription factors, mutations in epigenetic regulators, and oncogenic mutations which activate signaling effectors, including in Ph-like B-ALL. However, the molecular pathogenesis of adult B-cell ALL has been less well studied, and there is a need to identify novel therapeutic targets in adult B-ALL. We therefore performed detailed genomic profiling in adults with B-cell ALL.
We performed targeted DNA/RNA capture and sequencing of 189 patients with B-cell ALL enrolled on the ECOG E2993 clinical trial. All samples were taken at the time of diagnosis before patients were treated with multi-agent chemotherapy and followed for clinical outcomes including overall survival and remission/relapse duration and kinetics. We used the FoundationOne® Heme assay for our genomics study. This platform allows us to identify short events (point substitutions, short insertions/deletions) copy number and fusion events present in DNA and/or RNA. Of the 203 samples profiled, 189 had sufficient quality genomic data with a median coverage of 435 on DNA sequencing and 7.85 million on target distinct reads per case on RNA sequencing.
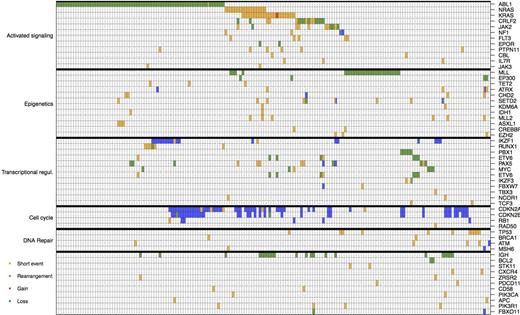
In all, we identified cancer-related mutations in 108 genes, 55 of these genes being mutated in more than one sample. We identified a median of 2.98 genomic alterations per sample (standard deviation of 1.78) in the target gene set, which included all genes known to be altered in B-ALL and in other hematologic malignancies. The most common genomic events were the BCR-ABL1 fusion (69 samples, 36.5%), followed by CDKN2A loss (57 samples, 30%), CDKN2B loss (35 samples, 18%), NRAS and KRAS point mutations (32 samples, 16.9%), and MLL fusion genes with a spectrum of partners (25 samples, 13.2%). Notably, we found that BCR-ABL1 fusion are mutually exclusive with both RAS mutations (p<10^-7) and MLL mutations (p<5*10^-4).
We next investigated the spectrum and frequency of alterations observed in different classes of oncogenes/tumor suppressors. We identified mutations which result in activation of oncogenic signaling pathways in 130 cases or 68.7% of our cohort. This included known mutations/fusions, such as BCR-ABL, RAS-MAPK pathway mutations, and JAK-STAT pathway alterations, and included missense alleles, insertion/deletion events, and fusion genes targeting a spectrum of genes including ABL, PDGFRB, and CRLF2. In aggregate, 33.8% of the cohort had mutations which activate signaling exclusive of BCR-ABL, demonstrating the high prevalence of the Ph-like B-ALL subtype in adult B-ALL. We identified mutations affecting key transcription factors, including PAX5 and IKZF1, in 31.2% of our cohort including missense, nonsense and deletion events. To confirm our ability to detect small deletions of PAX5 and IKZF1, we performed array CGH of 50 cases and compared copy number calls using high-throughput sequencing and CGH. Of note, all deletions targeting PAX5 and IKZF1 identified by NGS were confirmed by CGH. We also identified mutations in epigenetic regulators in 30.6% of cases, including MLL fusions, mutations in the DNA methylation/hydroxymethylation the pathway (DNMT3A, TET1, TET2), and mutations targeted chromatin modulators.
Detailed analyses of the mutational spectra and their relationship to clinical outcome will be presented at the meeting. Taken together, these data show we can use a clinically validated sequencing assay to identify known and novel somatic mutations in adult B-ALL, including sequence alterations, rearrangements and copy number aberrations. These data demonstrate the feasibility of combined DNA and RNA study to identify a complex landscape of fusions and short mutations with prognostic and therapeutic relevance, and inform the use of genomic profiling in B-ALL in the clinical context.
He:Foundation Medicine, Inc.: Employment, Equity Ownership. Brennan:Foundation Medicine, Inc.: Employment, Equity Ownership. Nahas:Foundation Medicine, Inc.: Employment, Equity Ownership. Balassubramanian:Foundation Medicine, Inc.: Employment, Equity Ownership. Otto:Foundation Medicine, Inc.: Employment, Equity Ownership. Lipson:Foundation Medicine, Inc.: Employment, Equity Ownership. Stephens:Foundation Medicine, Inc.: Employment, Equity Ownership. Miller:Foundation Medicine, Inc.: Employment, Equity Ownership. van den Brink:Boehringer Ingelheim: Consultancy, Other: Advisory board attendee; Merck: Honoraria; Regeneron: Honoraria; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Tobira Therapeutics: Other: Advisory board attendee. Rowe:Amgen: Consultancy; BioSight Ltd.: Consultancy, Membership on an entity's Board of Directors or advisory committees; BioLineRx Ltd.: Consultancy. Levine:Foundation Medicine: Consultancy; CTI BioPharma: Membership on an entity's Board of Directors or advisory committees; Loxo Oncology: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.