Abstract
In the donor versus no donor analysis of the UKALL12/E2993 trial in adult acute lymphoblastic leukaemia (ALL), there was a significant improvement in overall survival (OS) and reduction in relapse in the donor arm. However the OS benefit did not extend to older patients in whom myeloablative allogeneic hematopoietic cell transplant (alloHCT) related mortality was 35% at 2 years and outweighed the reduction in relapse risk. The overall outcome of older patients in this trial was poor, with patients >40 and 50 years having 23% and 15% 5 year survival respectively. In our current UK National Cancer Research Institute UKALL14 study all patients >40 years in CR, regardless of Philadelphia (Ph+) status and other high risk factors, are considered "high risk" and recommended a reduced intensity conditioned (RIC) alloHCT with a matched sibling (sib) or 8/8 matched unrelated donor (MUD) after a 2 course induction and high dose methotrexate. The primary endpoint is event free survival (EFS). We report here the early outcome of 88 patients with at least day 100 follow up (FU) (median FU 18 months) who received RICalloHCT on trial.
Conditioning was with fludarabine 30mg/m2 d -6 to -2, melphalan 140 mg/m2 d -2 and alemtuzumab 30mg d -2 to-1 (MUD) or d-1 (sib). Graft versus host disease (GVHD) prophylaxis was ciclosporin A only. Nine patients did not receive alemtuzumab as per protocol. Multilineage chimerism (MC) and minimal residual disease (MRD) were assessed 3 monthly post alloHSCT. Escalating doses of donor lymphocyte infusions (DLI) were given for T-cell mixed chimerism or MRD, starting dose 1 x 106 CD3 cells/kg, escalating by half log increments every 3 months.
Of 511 registered patients, 306 were >40 years old; 127 of those have completed RIC allograft, 88 of whom have sufficient FU to report. Median age was 51.5 years (range 41 to 64). Donor was sib in 24 and MUD in 64 patients, respectively. Median WBC at diagnosis was 7 x 109/l (0.6-557). Forty one of 77 (53%) evaluable patients had high risk cytogenetics, 22 (25%) were Ph+. Twelve of 56 (21%) with MRD data were MRD +ve pre-alloHCT.
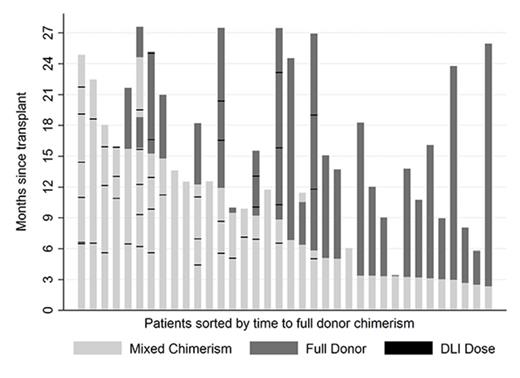
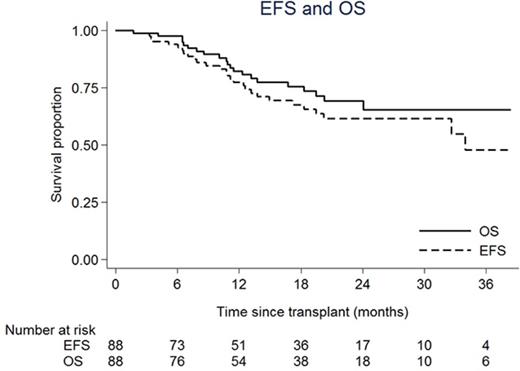
Post-alloHCT, myeloid engraftment occurred in 86/88 patients at a median of 13 days, 2 had missing data. Acute graft versus host disease (GVHD) occurred at grade 1 in 30 patients (34%) and grade 2-3 in 8 patients (9%). Thirty patients developed chronic GVHD (34% of 87/88 patients surviving to D100), 13 limited and 17 extensive. Of 12 patients who suffered transplant-related mortality (TRM), 6 died of infection (one post-transplant lymphoproliferative disease), 4 of organ toxicity and 2 of GVHD. TRM was not associated with age or donor type. Sixteen patients relapsed at a median time of 317 days (range 110-1034). Of those, 9 (of 15 with data) had high risk cytogenetics (EFS, p=0.38) and 6 (of 11 with data) were MRD +ve pre-alloHCT (EFS, HR 3.35, p=0.047). Twenty seven patients in total received 67 DLI (median maximum dose 3 x 106 CD3 cells/kg): 15 for mixed chimerism, 4 for rising MRD and 8 for both. Five patients (19%) developed post-DLI GVHD at grade 1 (n=4) and grade 2 (n=1). MC data is available for 36 patients receiving alemtuzumab and is shown in figure 1 where dark grey bars represent full and light grey bars mixed chimerism, respectively. DLI are denoted by black dashes. Mixed chimerism was seen in 22 of 36 patients (61%) who received alemtuzumab and so far 26 of 36 have achieved full donor chimerism (median 9 months), 10 after DLI (38%). Figure 2 shows Kaplan Meier curves of OS 76% (64-84, 95% CI) and EFS 68% (55-77)at 18 months.
This is the first prospective study of RIC alloHCT in older adults with ALL; these early results demonstrate its feasibility within a large, multicentre trial. T-cell mixed chimerism was common at 1st MC assessment but early data indicate that conversion to full donor chimerism with DLI is a safe and realistic possibility. The impact of mixed chimerism (and pre-emptive DLI) on relapse remain to be evaluated. Severe GVHD and TRM were relatively low, such that the EFS of chemotherapy then RIC alloHCT, albeit with short-term FU, may be higher than expected for chemotherapy alone in this age group.
Copland:Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Ariad: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees. Fielding:Amgen: Consultancy, Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.