Case presentations
Case 1
A 48-year-old man presents with epistaxis, fatigue, and pancytopenia, and is diagnosed with acute myeloid leukemia (AML) with a t(9;11)(p22;q23) translocation in 16 of 20 metaphases. He has an excellent performance status and no comorbidities. Curative-intent chemotherapy with cytarabine and daunorubicin (“7 + 3”) is initiated. What is the most appropriate strategy to prevent fungal infections in this patient?
Case 2
The patient has achieved a morphologic complete remission with 2 cycles of induction chemotherapy. An HLA-matched unrelated donor has been identified, and he is planned to undergo myeloablative allogeneic hematopoietic cell transplantation (HCT). What strategy should be pursued to prevent fungal infections before and after engraftment?
Introduction
Invasive fungal infections (IFIs) occur in 5% to 40% of patients with hematologic malignancies and are most common in AML.1 Aspergillus and Candida species (spp) currently account for ∼95% of all cases, but the epidemiological characteristics of IFIs evolve under the selection pressure of antimicrobials and other factors.2,3 With increasing use of intensively immunosuppressive cancer therapies, IFIs have become more frequent and now constitute a leading cause of morbidity and mortality. An important reason for delays and reductions of antileukemia therapies, they can also reduce AML cure rates.1,4-6 High mortality from IFIs is attributed to diagnostic difficulties and protracted treatment initiation, limited activity of antifungal agents, drug side effects, and increasing use of high-dose corticosteroids.7 Primary prevention of fungal infections, repeatedly demonstrated to reduce IFIs as well as infection-attributable and all-cause mortality, therefore remains essential.8,9
The ideal prophylactic antifungal agent is safe and well tolerated with long-term use, effective against a wide spectrum of organisms, and manufactured as IV and oral formulations with good bioavailability.10 With multiple polyenes, echinocandins, and triazoles now available, several antifungal agents fulfill some of these requirements. In developing a rationale for antifungal prophylaxis, the potential risks need to be balanced against the benefits. Among the risks of antifungal prophylaxis are drug toxicities, selection for resistant pathogens, adverse drug-drug interactions, and costs. Among the key benefits of prevention of invasive fungal infections during neutropenia in AML induction therapy are the reduction of morbidity and mortality, and shortening of hospital stay. One must appreciate that there is no single agent that will prevent all mycoses; thus, careful monitoring throughout the risk period is essential with treatment of emergent breakthrough invasive fungal infections. Herein, we examine the evidence guiding the choices of antifungal prophylaxis in adults undergoing curative-intent AML therapy.
Methods
Literature search strategy and study selection criteria
A systematic literature search restricted to English language articles published since 1990 was conducted using MEDLINE (May 18, 2015; see supplemental Table, available on the Blood Web site). Two authors independently reviewed all abstracts for eligibility assessment, with a third mediating discordant results. The full article was reviewed if eligibility was clearly met or if there was uncertainty regarding a priori–defined eligibility criteria based on the abstract. We included all randomized controlled trials (RCTs) and meta-analyses assessing a systemic antimycotic relative to no intervention, placebo, or another antifungal agent as prophylaxis in afebrile adults age >18 years with AML undergoing intensive chemotherapy or allogeneic HCT.
Outcome measures and data extraction
The primary outcome of interest was the incidence of probable and/or proven IFIs as defined by the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG).11 Secondary outcomes of interest included frequency of IFIs by organism, rates of adverse drug effects and toxicity-related drug discontinuation, overall survival, and IFI-attributable as well as all-cause mortality. A database of abstracted variables was created that included the above outcomes as well as years of enrollment, study design, number and age of study subjects, inclusion/exclusion criteria, type of malignancies and transplants, adverse events, and duration of follow-up. The strength of recommendations and the quality of evidence were evaluated on the basis of the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) system (Table 1).12
Results
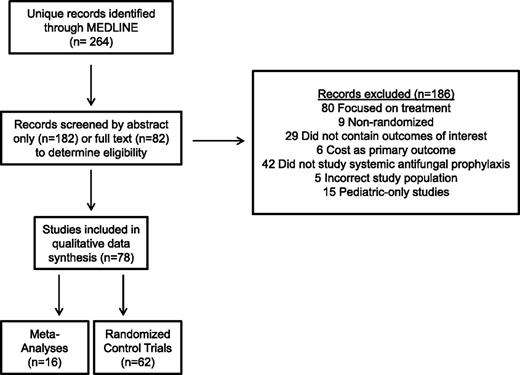
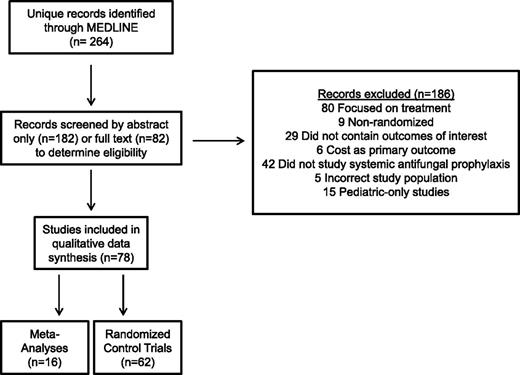
Our literature search yielded 264 records (Figure 1). Seventy-eight of these (62 RCTs [including 6 blinded, 20 double-blinded/placebo-controlled], 16 meta-analyses) were eligible for qualitative synthesis. We prioritized trials that exclusively or primarily involved patients with AML or high-risk myelodysplastic syndrome (MDS) receiving intensive chemotherapy to enhance the applicability of our findings. We excluded studies solely examining patients undergoing autologous HCT but included trials reporting on autologous and allogeneic (or allogeneic only) HCTs.
Case 1: Antifungal prophylaxis in adults undergoing AML induction chemotherapy
Polyenes.
Regardless of the systemic formulation (deoxycholate or lipid formulation), amphotericin B is usually too toxic for antifungal prophylaxis in AML induction therapy.13-15 The oral formulation of amphotericin B is not widely available and has no activity against inhaled molds, as this drug is not absorbed systematically. Inhalations of aerosolized amphotericin B are better tolerated but are not well established as preventative measures in this setting and are not standardized for delivery to the alveoli.16,17
Fluconazole and itraconazole.
Fluconazole, available in IV and oral formulations, is well tolerated and has activity against many yeast (particularly Candida) species. However, the incidence of non-albicans Candida spp with intrinsic resistance or limited susceptibility to fluconazole is increasing,3 and the drug lacks activity against filamentous fungi such as Aspergillus spp. Fluconazole has been widely studied in RCTs and found to reduce the incidence and mortality of IFIs compared with placebo18 and to be equally effective, but better tolerated than, amphotericin B formulations.13,15,19 Itraconazole’s activity spectrum is wider than that of fluconazole and includes Aspergillus spp. Itraconazole is available in oral and IV formulations (in some countries) but has poor gastrointestinal (GI) tolerability when given orally as solution.20 Four independent meta-analyses of RCTs comparing these 2 triazoles in neutropenic patients with hematologic malignancies showed that itraconazole significantly reduced IFIs (but not IFI-attributable or all-cause mortality) compared with fluconazole at the expense of greater drug toxicity.8,9,21,22
Echinocandins.
Caspofungin, micafungin, and anidulafungin are available in IV form only. They are well tolerated and have activity against Candida and Aspergillus spp but not Mucorales or Fusarium spp. RCTs have only been conducted with caspofungin as prophylaxis in acute leukemia patients undergoing chemotherapy. Two studies in 175 patients with acute lymphoblastic leukemia or AML and 192 patients with AML or MDS found no differences in the incidence of probable/proven IFIs, IFI-attributable and all-cause mortality, and drug toxicities compared with itraconazole in 1 study or physician’s choice of either itraconazole (82%), fluconazole (12%), posaconazole (1%) or no prophylaxis (5%) in the other study.23,24
Voriconazole.
Available as IV and oral formulations, voriconazole is active against a broad range of fungi including Candida spp, Aspergillus spp, Scedosporium spp, and Fusarium spp.25 Its toxicity and safety profile is limited by visual hallucinations, cutaneous solar hypersensitivity, and hepatic transaminase elevation, whereas resistance of Aspergillus spp to voriconazole is increasingly recognized.26 RCT-level evidence supporting voriconazole prophylaxis in the nontransplant AML population is limited to 2 published studies, 1 of which was terminated after enrollment of only 25 patients because the use of placebo in the control arm was no longer deemed ethical.27 In the second study, 123 patients with AML/high-risk MDS receiving induction or salvage chemotherapy were randomized to oral voriconazole or IV itraconazole.28 This study failed to reach its target accrual and found no statistically significant differences in probable/proven IFIs, all-cause mortality, or toxicity-related drug discontinuation rates, suggesting equal value for prophylaxis in this patient population.
Posaconazole.
Posaconazole has activity against a diverse array of fungi including Candida spp, Aspergillus spp, Scedosporium spp, Fusarium spp, and several species of the Mucorales. Posaconazole has been compared with fluconazole and itraconazole (2 RCTs) but not to voriconazole or echinocandins in non-HCT AML patients. In a landmark study of AML/MDS patients undergoing induction chemotherapy, patients who received oral suspension posaconazole (n = 304) were less likely than those receiving oral fluconazole (n = 240) or oral itraconazole (n = 58) to develop probable/proven IFIs (total IFIs: 2% vs 8%, P < .001; invasive aspergillosis [IA]: 1% vs 7%, P < .001), and had lower 100-day mortality (14% vs 21%, P = .04) and lower IFI-attributable deaths (2% vs 5%, P = .01).20 Serious triazole-related adverse events were more frequent in the posaconazole arm (6% vs 2%, P = .01). An estimated 16 and 14 patients needed to be treated with posaconazole to prevent 1 IFI and 1 death, respectively. Partly consistent with these findings, a second RCT of 252 AML patients found that oral suspension posaconazole was associated with a lower rate of probable/proven IFIs than oral fluconazole (4% vs 9%, P = .026) and insignificantly lower all-cause mortality (2.6% vs 6%, P = .22), whereas drug-related adverse events were similar.29
A major limitation of oral suspension posaconazole is variable bioavailability, including in patients who develop diarrhea (eg, due to gut graft-versus-host disease [GVHD]), with subtherapeutic drug levels linked to increased risk of breakthrough IFIs in AML patients.30-33 Thus, monitoring plasma drug levels is recommended in patients with risk factors for poor absorption who are receiving the suspension.34 This limitation has been largely overcome by the recently introduced extended-release tablet form.35 Unlike the oral suspension formulation, absorption of the tablet is minimally affected by food, mucositis, and GI pH, resulting in increased serum drug levels in leukemia patients.34,36,37 The recommendation for monitoring of plasma drug levels therefore does not pertain to patients receiving the tablet form of posaconazole. Other important limitations of posaconazole include interactions with cytochrome P450 enzymes and P-glycoprotein,38 complicating its use in patients requiring multiple concomitant transplant-related medications or newer targeted antileukemic agents such as isocitrate dehydrogenase inhibitors. New-generation, broad-spectrum triazoles such as isavuconazole may overcome some of these limitations,39,40 but testing as prophylaxis in AML patients has just begun.41
Recommendation.
Fluconazole is better than placebo in preventing invasive candidiasis during AML remission induction therapy. However, fluconazole lacks activity against molds. Although itraconazole and voriconazole are used for prophylaxis against infections caused by Candida spp and molds, posaconazole is the only agent that has demonstrated a significant survival and outcome benefit in prophylaxis against these mycoses during AML induction therapy (GRADE 1A). Despite the lack of RCT data in AML, use of the tablet, if tolerated, rather than the oral suspension of posaconazole is recommended based on phase 1 findings in hematologic malignancies,35 retrospective studies,36,42 and early results from phase 3 studies43 demonstrating superior bioavailability of the tablet without worsening of adverse effects (GRADE 1C). As there is a good relationship between serum concentrations of posaconazole and therapeutic outcome of IA, use of the tablet formulation in lieu of the suspension is tenable. For patients who are not able to swallow the tablet, the oral suspension is recommended. Coadministration of posaconazole and drugs that strongly interfere with the 3A4 isoform of cytochrome P450 should be avoided. For patients who are not able to tolerate oral posaconazole and in cases where the IV formulation is not available, administration of an echinocandin is a reasonable substitute for antifungal prophylaxis (GRADE 1B). If there is a high risk for IA, IV voriconazole should be considered (GRADE 2B).
Case 2: Antifungal prophylaxis in adults with AML undergoing HCT
The incidence of IFIs after allogeneic HCT is as high as 10% to 20%, and associated mortality ranges from 30% to 80% depending on the organism.4,5 Although antifungal prophylaxis after HCT has long been considered standard,44 issues specific to this setting include concerns for drug-drug interactions with conditioning and immunosuppressive agents, poor oral absorption secondary to treatment-related gut toxicity, as well as GVHD and associated use of corticosteroids.45 Several factors, including donor source, history of IFIs, active hematologic cancer, and GVHD can identify patients at particularly high risk of IFIs throughout the post-HCT period and could help tailor prevention strategies.46
Polyenes.
Fluconazole and itraconazole.
Fluconazole is well suited for posttransplant use due to good tolerability and minimal drug-drug interactions, and is as effective as amphotericin B with regard to incidence of IFIs and mortality.49 In 2 placebo-controlled RCTs, conducted at a time when the majority of IFIs were due to Candida spp, fluconazole was associated with lower incidence of IFIs and reduced IFI-related mortality.51,52 In 1 of the studies, fluconazole given until day 75 after allogeneic HCT also led to improved overall survival.52,53 These findings established this use of fluconazole as standard prophylaxis at that time54 and as benchmark for novel antifungal agents. Several RCTs have exclusively compared itraconazole with fluconazole in patients undergoing HCT.55-58 In a study of 300 patients undergoing allogeneic HCT who received either itraconazole or fluconazole for 120 to 180 days posttransplant, rates of probable/proven IFIs or survival were similar but fewer mold infections were noted with itraconazole (5% vs 12%, P = .03).56 In a similar trial of 140 patients, oral itraconazole was associated with a lower rate of IFIs (9% vs 25%, P = .01), but no statistically significant difference in IFI-related or all-cause mortality.57 In contrast, in a third RCT of 195 patients with acute leukemia undergoing HCT who received either itraconazole or fluconazole until engraftment, there was no difference in IFIs, IA, or mortality.58 In all of these studies, there was significantly more GI toxicity with itraconazole. Although these studies largely supported the superiority of itraconazole over fluconazole in prevention of IFIs in allogeneic HCT, the toxicity profile of itraconazole has precluded its wide acceptance for prophylaxis in this setting. Increased renal and hepatic toxicities were observed when itraconazole was administered concurrently with cyclophosphamide and busulfan due to inhibition of their metabolism,59,60 highlighting the need for avoiding simultaneous exposures of antineoplastic agents and triazoles that inhibit hepatic microsomal enzymes. This toxicity may also increase the risk of sinusoidal obstruction syndrome and, consequently, nonrelapse mortality.61 Because of poor tolerance, itraconazole is therefore not recommended over fluconazole in HCT patients despite potentially higher efficacy.62
Echinocandins.
Because of their tolerability, echinocandins are good candidates for prophylaxis in HCT. Three RCTs have compared micafungin with fluconazole63,64 or itraconazole65 in this setting. In a study of 882 patients randomized to either micafungin or IV fluconazole in the neutropenic phase of HCT, micafungin was associated with a lower incidence of suspected or probable/proven IFIs (P = .03) and a trend toward lower IA (0.2% vs 1.5%, P = .07) but similar IFI-related and all-cause mortality and adverse events.63 On the other hand, in a similarly designed but underpowered trial of 106 patients, no differences in probable/proven IFIs or adverse events were found between micafungin and fluconazole.64 As an important limitation for our purpose, these 2 trials included a large proportion of non-AML patients and individuals undergoing autologous HCT. In the third RCT, there was no difference in probable/proven IFIs and IFI-related or all-cause mortality among 287 HCT patients (83% allogeneic) assigned to micafungin or itraconazole, but significantly more patients discontinued itraconazole early due to toxicity (19.7% vs 0.7%, P < .01).65 Thus, micafungin is noninferior to fluconazole and itraconazole and is less toxic than itraconazole.
Voriconazole.
Two large RCTs of 600 and 489 patients (>50% with AML or MDS) undergoing allogeneic HCT have compared voriconazole with fluconazole or itraconazole given until day 100 posttransplant.25,66 There were no differences in IFIs or survival between the treatment arms in either study, but in the study comparing voriconazole with fluconazole, a protocol-driven, structured empirical antifungal therapy and diagnostic screening program was used and evaluations for IFIs were mandated if a positive serum galactomannan assay or suggestive signs of fungal infection were noted.66 In the second study, better tolerance of voriconazole than itraconazole was demonstrated, with a higher proportion of patients completing the study (54% vs 39%, P < .01) although hepatotoxicity occurred more commonly with voriconazole (13% vs 5%, P < .01), requiring frequent adjustment of calcineurin inhibitors.25 Indeed, unpredictable interactions between voriconazole and commonly used immunosuppressants (tacrolimus, cyclosporine A) are well documented.67,68 Thus, voriconazole has no clear benefit over fluconazole or itraconazole in terms of efficacy, but is preferred over itraconazole due to better tolerability.
Posaconazole.
Two RCTs have evaluated posaconazole as prophylaxis in AML patients undergoing HCT. In a study of 40 patients randomized to either posaconazole oral solution or weekly IV amphotericin B lipid complex, rates of IFIs were similar (0% vs 5%, P = .48) but the median number of days on fungal prophylaxis was longer in the posaconazole group (42 vs 20 days, P = .01) due to amphotericin intolerance, particularly renal toxicity, which led to early trial termination.50 In the second RCT, 600 patients (30% with AML or MDS) with active GVHD requiring immunosuppressive therapy after allogeneic HCT were allocated to either posaconazole or fluconazole. Posaconazole was associated with a trend toward lower incidence of probable/proven IFIs (5% vs 9%, P = .07), a lower incidence of probable/proven IA (2.3% vs 7%, P = .006), and lower IFI-attributable mortality (1% vs 4%, P = .046), whereas all-cause mortality and rates of treatment-related adverse events were similar.69
Recommendations.
Following allogeneic HCT, fluconazole, voriconazole, posaconazole, and micafungin are all equally recommended over no antifungal prophylaxis in low-risk patients (GRADE 1B). For the quality of evidence, the distinction is made between use of posaconazole for prophylaxis in AML during induction therapy and for preengraftment and postengraftment prophylaxis after HCT, where matters of study design and interpretation influence the recommendations. Additional high-quality studies are needed to refine the recommendations regarding the optimal antifungal agent for the pre- and post-engraftment period following allogeneic HCT. For high-risk patients, especially those with GVHD, posaconazole is recommended over other antifungal strategies (GRADE 1A); if poor oral absorption of posaconazole is anticipated, voriconazole is an acceptable substitute (GRADE 2B).
Discussion
We found high-quality evidence to recommend posaconazole for IFI prophylaxis in patients with AML undergoing induction chemotherapy and those with GVHD following allogeneic HCT, whereas there is no strong evidence to guide the selection of antifungal prophylaxis following allogeneic HCT in patients who have no GVHD or other high-risk factors. Although costly, several studies have demonstrated cost-effectiveness of posaconazole in patients at high risk for fungal infections compared with other triazoles due to the reduction of IFIs and shortened hospital stays.70-74
As an important caveat for recommendations based on RCTs, studied subjects are typically highly selected based on stringent inclusion/exclusion criteria. Therefore, RCT findings may not be generalizable to all patients with AML.75 However, retrospective and prospective observational studies support the use of posaconazole rather than itraconazole or fluconazole in AML patients undergoing intensive chemotherapy76,77 or allogeneic HCT78 and complement the RCT data. Still, strategies for antifungal prophylaxis in a broader range of patients with AML are needed such as those with end-organ toxicity for whom individual assessments should be made. For example, in patients with preexisting hepatic transaminase elevation, an echinocandin may be the best choice for antifungal prophylaxis as it provides protection from most Candida spp and possibly Aspergillus spp. Furthermore, few studies of antifungal prophylaxis have been conducted in pediatric AML. Antifungal prophylaxis in pediatric patients has largely focused on high-risk acute lymphoblastic leukemia. In the absence of large robust randomized trials in the pediatric population, extrapolation with appropriate pharmacokinetic/pharmacodynamic bridging provides for rational dosing of antifungal agents in pediatric patients during chemotherapy-induced neutropenia.79,80
A number of questions remain unanswered, including the efficacy of the many novel antifungal agents that are under preclinical and/or clinical development (eg, isavuconazole, ravuconazole, albaconazole, aminocandin, among others), the efficacy and tolerability of newer formulations of established drugs, and the role and reliability of monitoring strategies for early detection of IA.81 Serum galactomannan testing is useful in the early detection of IA in neutropenic patients who are not receiving antimold prophylaxis. The presence of circulating levels of antimold agents diminishes the galactomannan index signal and utility of this assay as most positive galactomannan tests during surveillance in the setting of mold-active prophylaxis represent false-positive results.81 Open questions also relate to the best strategy for the treatment of breakthrough fungal infections in patients receiving prophylaxis with broad-spectrum antifungal drugs, the role of therapeutic drug monitoring, and resistance development.82-85 As each new antifungal agent has been introduced and accepted into wider use, the pattern of IFIs has shifted from susceptible invasive Candida spp to resistant Candida spp and IA. Now, with increased use of mold-active triazoles, breakthrough IFIs with resistant Aspergillus spp and non-Aspergillus molds such as Fusarium spp are being seen. Additionally, although the recommendations throughout this article are broad and population based, there are many subtleties in decision-making when choosing prophylaxis for individual patients, and other factors beyond type of therapy, engraftment stage, and GVHD (Table 2)86-104 are increasingly recognized as being important for risk stratification and choice of antifungal agent. As we move toward personalized medicine and targeted therapy in oncology, efforts to allow for more individualized choices in fungal prophylactic therapy are ongoing, with work being done on risk prediction scores for IFI development, immunogenetic profiling to determine host-specific risk factors for IFI infection, and individual mycobiome monitoring.86,87,100,105
The online version of this article contains a data supplement.
Acknowledgments
A.B.H. is supported by a fellowship training grant from the National Heart, Lung, and Blood Institute/National Institutes of Health (NHLBI/NIH: T32-HL007093).
T.J.W. is a Scholar of the Sharp Family Foundation in Emerging Pediatric Infectious Diseases and a Scholar of the Henry Schueler Foundation in Mucormycosis.
D.P.K. acknowledges the Francis King Black Endowment for Cancer Research.
R.B.W. is a Leukemia & Lymphoma Society Clinical Scholar in Clinical Research.
Authorship
Contribution: A.B.H. and R.B.W. were responsible for the concept of this review and contributed to the literature search and data collection quality assessment, analyzed and interpreted data, and wrote the manuscript; G.H.L. was responsible for the concept of the review, contributed to the literature search, and critically revised the manuscript; and T.J.W. and D.P.K. contributed to the concept of the review, assessed the literature, and critically revised the manuscript.
Conflict-of-interest disclosure: T.J.W. has received research grants for experimental and clinical antimicrobial pharmacotherapeutics from Astellas, Novartis, Merck, ContraFect, Pfizer, Cubist, and Theravance, and has served as consultant to Astellas, ContraFect, Cubist, Drais, iCo, Novartis, Pfizer, Methylgene, SigmaTau, and Trius. D.P.K. has received research support from Merck, Pfizer, Astellas, and T2 Biosystems, and has received honoraria from Merck, Astellas, Gilead, Mylan, F2G, iNc, and T2 Biosystems. The remaining authors declare no competing financial interests.
Correspondence: Anna B. Halpern, Clinical Research Division, Fred Hutchinson Cancer Research Center, 1100 Fairview Ave N, D5-100, PO Box 19024, Seattle, WA 98109-1024; e-mail: halpern2@uw.edu.