Abstract
An often-cited defining property of hematopoietic stem cells (HSCs) is their extensive or unlimited in vivo self-renewal capacity. We have recently described a novel mouse disease model forFanconi anemia, in which serial challenge with pro-inflammatory agonists that mimic infection, such aspolyinosinic:polycytidylic acid (pI:C), results in HSC attrition followed by a highly penetrant severe aplastic anemia, closely recapitulating the disease in patients (Walter et al., 2015, Nature).
In order to explore the broader implications of these findings in the context of HSC self-renewal, we conducted apI:Cdose escalation regimen using standard C57BL6 mice. A single injection withpI:Cprovoked transient peripheral blood (PB)cytopenias, with the recovery of mature blood cell numbers correlating with HSCs being forced into active cell cycle. Injection with 1-3 rounds ofpI:C(1-3 x 8 injections) led to no discernable sustained impact on blood production as, at 5 weeks post-treatment, PB frequencies were in the normal range, as were the absolute numbers of HSCs and all progenitor compartments in the bone marrow (BM), as determined by flowcytometry.
However, in vitro analysis of the proliferation and differentiation potential of 411 individual sorted long-term (LT)-HSCs 5 weeks after 3 rounds of pI:C challenge, revealed a decrease in the frequency of LT-HSCs able to generate progeny in vitro (1.6-fold reduction, p<0.05), and a 2-fold reduction in the total number of progeny produced per HSC, which was even more pronounced inmultilineage potential clones (2.6-fold decrease, p<0.0001) compared touni- or bi-lineage clones. In line with this data, competitive repopulation assays demonstrated a progressive depletion of functional HSC numbers with increasing rounds ofpI:C treatment, with a 1.8, 3.4 and 15.3-fold decrease in donorchimerism across all lineages at 6 months post-transplantation (p<0.01) following 1, 2 or 3 rounds ofpI:C treatment, respectively. Notably, robust engraftment (up to 65% donorchimerism, 6 months post-transplantation, p<0.01) was achieved when mice exposed to 3 rounds ofpI:C treatment were used as a recipient for non-treated BM cells in the absence of any irradiation conditioning, while engraftment was always <1% when non-treated controls were used as recipients. This excludes the possibility that the observed progressive depletion of functional HSCs was the result of artifacts associated with a compromised niche or the non-physiologic stress imposed on donor cells during transplantation.
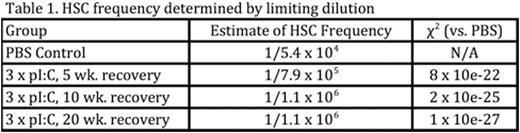
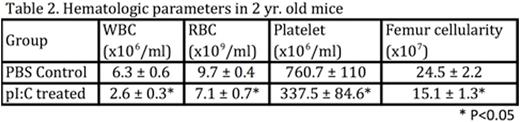
In order to test the kinetics of HSC recovery following HSC challenge, BM was harvested from mice at either 5, 10 or 20 weeks after treatment with 3 rounds of pI:C, and both competitive and limiting dilution transplantation assays (Table 1) were used to quantify HSC frequencies. Surprisingly, both assays demonstrated that HSCs failed to regenerate at all following pI:Cchallenge, directly contradicting the canonical view that HSCs possess extensive self-renewal capacity in vivo. The physiologic relevance of this observation was illustrated when we measured the hematologic parameters of aged mice that had been exposed to chronicpI:C treatment in early to mid-life. Although these mice had normal PB counts at 4 weeks post-treatment, at 2 years of age, peripheral bloodcytopenias and bone marrow aplasia became evident (Table 2), recapitulating clinically relevant features of non-malignant aged human hematopoiesis that are never seen in standard laboratory mice. Together, these data suggest that functional HSCs can be progressively and irreversibly depleted in response to environmental agonists, such as infection and inflammation, which force HSCs to reconstitute mature blood cells consumed by such stimuli.
This model has clear implications relating to the role of adult stem cells in tissue maintenance and regeneration during ageing, and how stress agonists that are absent in most laboratory animal models, but would be ubiquitous in the wild, are likely key mediators of age-associated disease pathologies.
Frenette:PHD Biosciences: Research Funding; Pfizer: Consultancy; GSK: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.