Abstract
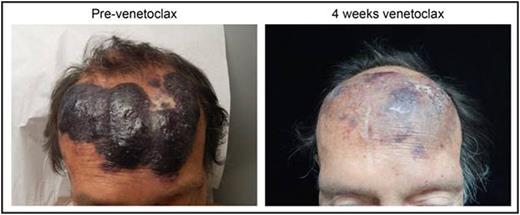
Blastic plasmacytoid dendritic cell neoplasm (BPDCN) is a particularly aggressive hematologic malignancy with median survival of <12 months and no standard therapy. BPDCN involves the skin in nearly all patients, and frequently infiltrates bone marrow and lymph nodes. Its normal counterpart may be the plasmacytoid dendritic cell (pDC), leading to the name BPDCN. Outcomes are poor with cytotoxic chemotherapy. An interleukin 3-diphtheria toxin fusion (SL-401) has activity in BPDCN (Frankel, Blood 2014), but additional novel agents are urgently needed. BPDCN over-expresses the anti-apoptotic protein BCL-2 compared to normal pDCs (Sapienza, Leukemia 2014). We confirmed this by RNA-sequencing 12 BPDCNs and pDCs from 4 normal donors (reads/kb mapped [RPKM] 22.7 vs 1.33, P=0.0005). BPDCN shares some genetic characteristics with myeloid malignancies, and some acute myeloid leukemias (AMLs) are dependent on BCL-2. We performed RNA-seq on 6 BPDCN and 16 AML patient-derived xenografts (PDXs) and found higher BCL-2 expression in BPDCN (RPKM 48.2 vs 11.5, P=0.0005). We analyzed BCL-2 expression by immunohistochemistry in patient skin and bone marrow biopsies and found that all BPDCNs had BCL-2 staining that was equivalent to or stronger than that of any AMLs. We next tested BPDCN cell lines, primary patient samples, and patient-derived xenografts (PDXs) for BCL-2 dependence and sensitivity to the BCL-2 inhibitor venetoclax (previously ABT-199). Using BH3 profiling, we found that BPDCN is markedly dependent on BCL-2 to prevent mitochondrial cytochrome c release in response to apoptotic stimuli. In comparison to AML, BPDCN had significantly more cytochrome c release after BAD peptide stimulation (81.1% vs 11.8%, P<0.0001), suggesting greater dependency on BCL-2 and/or BCL-XL. BPDCN was uniformly sensitive to treatment with venetoclax in vitro, in cell lines and primary cells, as measured by direct cytotoxicity and Annexin V apoptosis assays. We used dynamic BH3 profiling (Montero, Cell 2015) in primary BPDCNs and PDXs (n=7) to measure early apoptotic signals after 4-hr exposure to venetoclax, avoiding the need for prolonged culture. BPDCNs were more likely than AMLs to undergo cytochrome c release in response to BIM peptide stimulation after 4 hours of venetoclax (increase in apoptotic potential, or "delta priming" 63.4% vs 14.5%, P<0.0001). Targeted sequencing of the BPDCNs found various combinations of mutations in TP53, FLT3, JAK2, SRSF2, TET2, ASXL1, IDH2, GNB1, NRAS and/or ZRSR2. All responded equally to venetoclax, suggesting the response was independent of genotype. Next we treated two BPDCN PDXs in vivo in NSG mice with oral venetoclax (100 mg/kg/day x 28 days). PDX genetics were, PDX1: ASXL1 G646fs*, NRAS G13D, JAK2 V617F, TET2 D1017fs*, and TET2 Q1687fs*; PDX2: IDH2 R140Q, TP53 S241F, TP53 C176Y, and ZRSR2 S188*. Venetoclax caused significant reductions in BPDCN burden in peripheral blood, spleen, and bone marrow after 21 days of therapy in both models. Overall survival was improved in venetoclax compared to vehicle treated animals in a leukemia-watch cohort (57 vs 36 days, P=0.0025). On the basis of these findings, we treated a relapsed BPDCN patient with venetoclax. He is an 80 year-old male who had received 3 prior lines of therapy. He had extensive skin disease with multiple cutaneous tumors, lymph node involvement, and >80% bone marrow blasts. His BPDCN carried the mutations ASXL1 Y581fs*, ASXL1 E553fs*, GNB1 K57E, IDH2 R140W, and NRAS G12D, and expressed high levels of BCL-2 protein in bone marrow and skin. BH3 profiling of a skin tumor biopsy revealed marked BCL-2 dependence and dynamic BH3 response to venetoclax (4 hr delta priming 55.6%). We treated him using a regimen recently FDA-approved for chronic lymphocytic leukemia (CLL) consisting of weekly dose escalation (20 -> 50 -> 100 -> 200 mg), to a target dose of 400 mg daily. At the time of this writing, he had reached 200 mg without significant toxicity, including no evidence of tumor lysis syndrome. His skin disease has responded remarkably (Figure), with the first response evident within 10 days. Our data suggests that BPDCN is highly sensitive to BCL-2 inhibition, which could provide an urgently needed new treatment for patients with this disease. We propose that BCL-2 inhibition should undergo expedited clinical evaluation in BPDCN. In addition, this case offers an example of precision cancer medicine by functional rather than genetic means.
Davids:Infinity: Honoraria, Research Funding; Genentech: Consultancy, Honoraria, Research Funding; Gilead: Honoraria; Janssen: Consultancy, Honoraria; Pharmacyclics: Consultancy, Honoraria, Research Funding; TG Therapeutics: Honoraria, Research Funding; Abbvie: Consultancy, Honoraria. Stone:ONO: Consultancy; Novartis: Consultancy; Amgen: Consultancy; Seattle Genetics: Consultancy; Roche: Consultancy; Celator: Consultancy; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Agios: Consultancy; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Karyopharm: Consultancy; Jansen: Consultancy; Pfizer: Consultancy; Juno Therapeutics: Consultancy; Merck: Consultancy; Sunesis Pharmaceuticals: Consultancy; Xenetic Biosciences: Consultancy. Konopleva:Reata Pharmaceuticals: Equity Ownership; Abbvie: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Stemline: Consultancy, Research Funding; Eli Lilly: Research Funding; Cellectis: Research Funding; Calithera: Research Funding. Letai:AbbVie: Consultancy, Research Funding; Astra-Zeneca: Consultancy, Research Funding; Tetralogic: Consultancy, Research Funding. Lane:N-of-1: Consultancy; Stemline Therapeutics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.