Abstract
Background
Activating mutations in FLT3 - and in particular FLT3-ITD (internal tandem duplication) - are common in younger patients with AML, and are associated with relapse and with inferior overall survival (OS), however their prevalence and impact in older adults remains uncertain. We performed an analysis of FLT3-ITD mutations in patients age ≥60 yrs in the large prospective multicenter ECOG-ACRIN (E-A) E2906 Phase III trial.
Methods
Eligible patients (pts) age ≥60 yrs (n=727) were randomized to 'Standard' therapy with 7&3 (Dauno 60mg/m2) induction, and 2 cycles of intermediate dose Ara-C (1.5g/m2 x 12 doses; 6 doses if age ≥70yrs) consolidation (Arm A); or single agent clofarabine (CLO) induction and consolidation (2 cycles) (Arm B). As previously reported (Foran et al, ASH #217a, 2015), there was superior overall survival (OS) with standard therapy. AML diagnostic samples collected prospectively in the central E-A Leukemia Translational Research Lab (LTL) were used to detect mutations in the FLT3 gene by PCR using cDNA from total RNA. LTL investigators were blinded to treatment assignment. Patients with no RNA available or blast count <10% (threshold of sensitivity for the PCR assay) on the submitted sample were excluded. Statistical analysis was performed using X2 (categories) and Wilcoxon rank sum (continuous) tests to compare baseline patient and disease characteristics. Log-rank tests and multivariate Cox models stratified by treatment arm and adjusted for patient and disease variable (including WBC, cytogenetics, sex, performance status, secondary AML) were used to examine FLT3-ITD effect on OS and disease-free survival [DFS; relapse or death after complete remission (CR)/CRi (CR with incomplete CBC recovery)].
Results
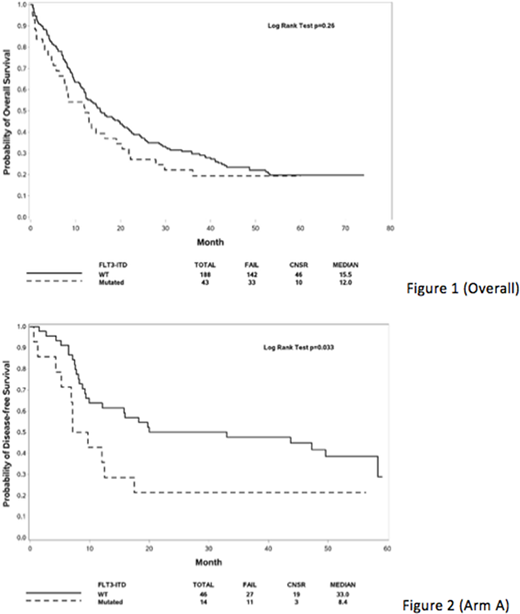
In the first 231 pts tested, FLT3-ITD mutations were identified in 43 (18.6%) pts, and the remainder were FLT3-ITD-negative (i.e. non-ITD). The proportion with FLT3-ITD was the same for patients age 60-69 yrs (18.8%) vs. ≥70 yrs (18.3%). In comparison to non-ITD, FLT3-ITD+ pts had significantly higher WBC (median 18.1 vs. 6.3, p=0.002) and BM blasts (80% vs. 51.5%, p=0.0004) at AML diagnosis, and were more likely to have intermediate risk (79.1% vs. 59%) vs. unfavorable risk (7.0 % vs. 31.4%) cytogenetics (p=0.002). There was no difference in CR/CRi rate overall (p=0.40), however standard (Arm A) pts with FLT3-ITD had a significantly higher CR/CRi rate (78.9% vs. non-ITD 51.5%, p=0.04). With median follow-up 53.5 months, FLT3-ITD patients tended to have worse OS (HR 1.24, 95%CI 0.85-1.81) but this was not statistically significant (Figure 1, p=0.26), and results were similar for both Arms A/B. DFS similarly tended to be worse overall for FLT3-ITD (HR 1.44, 95%CI 0.65-2.43) (p=0.17), and DFS was significantly worse for FLT3-ITD+ Arm A/Standard pts (Figure 2, p=0.033), while Arm B/CLO pts had a worse DFS regardless of FLT3 mutation status (Arm B, FLT3-ITD vs. non-ITD, p=0.93). More patients with FLT3-ITD underwent allogeneic transplantation (25.6% vs. non-ITD 19.7%), although this was not significant (p=0.41).
Conclusions
FLT3-ITD mutations are prevalent in older and elderly (age ≥70 yrs) patients with AML, and while they occur at somewhat lower rates than reported for younger adults, FLT3-ITD+ AML in this population has a leukemia phenotype similar to that reported in younger patients. Despite significantly higher CR/CRi rates with Standard therapy, older pts with FLT3-ITD also have significantly worse DFS following intensified Ara-C consolidation therapy than non-ITD pts. These results support routine assessment of FLT3-ITD status in older AML patients, and the incorporation of novel post-remission treatment strategies to improve outcome in FLT3-ITD+ pts ≥60 yrs.
Foran:Agios: Research Funding; Xencor, Inc.: Research Funding. Lazarus:Pluristem Ltd.: Consultancy. Tallman:BioSight: Other: Advisory board; AbbVie: Research Funding; Daiichi-Sankyo: Other: Advisory board; Cellerant: Research Funding; Orsenix: Other: Advisory board; ADC Therapeutics: Research Funding; AROG: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.