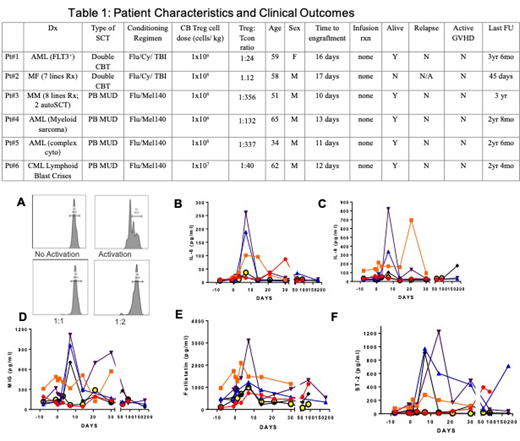
Previously, we presented the results of a phase I trial of using cord blood (CB) derived regulatory T cells (Tregs) at a dose of 1x106 cells/kg in the prevention of graft vs. host disease (GVHD) in five patients undergoing allogeneic stem cell transplant (SCT). We now present an update of those patients and also the outcome of a single patient treated with CB Tregs at higher dose of 1x107 cells/kg. At the last follow up of 3.5 years, 5 of 6 patients are alive, in complete remission, without GVHD and off immune-suppression (table 1). One patient died of head injury at day 45 post-transplant without GVHD. None of the patients relapsed in spite of having high risk disease including Flt3+ acute myeloid leukemia (AML) (pt#1); refractory mycosis fungoides/ sezary syndrome (pt#2); relapsed refractory multiple myeloma including two autologous SCT (pt#3); myeloid sarcoma (pt#4); AML with complex cytogenetics (pt#5). Specifically, the sixth patient (treated at CB Tregs cell dose of 1x107 cells/kg) had a diagnosis of lymphoid blast crisis of chronic myelogenous leukemia (CML) and underwent allogeneic peripheral blood (PB) matched unrelated donor (MUD) SCT in second chronic phase with the conditioning regimen of fludarabine (40 mg/m2/d on day -5 to -2) and melphalan (140mg/m2 on day -2) and received CB Tregs at a dose of 1x107 cells/kg on day -1. The CB Tregs were manufactured at the MD Anderson GMP facility. Tregs were isolated from 4 out of 6 HLA matched CB unit with a post-thaw total nucleated cell (TNC) count of 1295 x 106 cells with 90% recovery. Enrichment of CD25+ Treg cells was accomplished with directly conjugated anti-CD25 magnetic microbeads and the LS column (Miltenyl) based selection. After selection, the total no. of isolated CB Tregs was 17.6 x 106 cells. These cells were ex-vivo expanded in culture for a total of 14 days in the continued presence of CD3/28 microbeads and interleukin-2. On the day of harvest, a total of 1359 x 106 Treg cells were generated with 96% viability. Based on patient's weight of 67 kg, a total of 67x107 CB Treg cells were infused. The expanded CB Tregs were also evaluated for functionality by using an in vitro suppression assay, where the stimulated CD4+ Tcon cells stained with CellTrace Violet and Tregs were added into a 96-well plate at a 1:1 and 1:2 ratio and incubated for three days at 37oC. As shown in figure 1A, the clinically manufactured CB Tregs were functional and exerted 98% suppression of the proliferating Tcon cells. Subsequently, the patient received PB MUD graft on day 0. The graft had a TNC count of 1900 x106 cells where the CD3+ T cells were at a dose of 400 x106 cells/kg resulting in a Treg: Tcon ratio of 1:40, a significantly lower number of Tregs compared to other published clinical trials using Treg prophylaxis for GVHD. There was no infusion reaction related to the CB Treg infusion, the patient engrafted on day+12 with 100% donor chimerism on day +30. The patient did not develop any grade II-IV GVHD and was off immune suppression by +6 months. At the time of last follow up of 2 years and 4 months, the patient remains alive, in complete remission and without GVHD. We compared the inflammatory cytokine profile as well as GVHD biomarkers for pt #6 to the rest of the five patients who received the lower dose of CB Tregs at 1x106 cells/kg (with up to 400 times Tcon cells in the graft), where 4 patients had developed acute GVHD. The inflammatory biomarkers of IL-6 (fig 1B) and IL-8 (fig 1C) for pt#6 were low to undetectable compared to the other patients. Similarly, the GVHD biomarkers of ST2 (fig 1D); MIG/ CXCL9 (fig 1E) and follistatin (fig 1F) were also low to undetectable in pt#6 compared to the rest. At the time of last follow up, all patients had resolved acute GVHD and were off-immune suppression and in complete remission. Based on these data, we conclude that CB Treg dose of 1x107 cells/kg may be able to prevent GVHD without alleviating the graft vs leukemia effect.
Popat:Jazz: Consultancy; Incyte: Research Funding; Bayer: Research Funding. Nieto:Affimed: Consultancy; Astra-Zeneca: Research Funding; Novartis: Research Funding; Affimed: Research Funding. Ciurea:Kiadis Pharma: Membership on an entity's Board of Directors or advisory committees, Other: stock holder; MolMed: Membership on an entity's Board of Directors or advisory committees; Spectrum: Membership on an entity's Board of Directors or advisory committees; Miltenyi: Research Funding. Qazilbash:Amgen: Consultancy, Other: Advisory Board; Genzyme: Other: Speaker; Bioclinical: Consultancy; Autolus: Consultancy. Champlin:Actinium: Consultancy; Johnson and Johnson: Consultancy; Sanofi-Genzyme: Research Funding. Parmar:Cellenkos Inc.: Equity Ownership, Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.