TO THE EDITOR:
Primary vitreoretinal lymphoma (PVRL) is a rare non-Hodgkin lymphoma, frequently associated with primary central nervous system (CNS) lymphoma (PCNSL).1 It particularly concerns elderly subjects with limitations to receive chemotherapies. The prognosis is poor due to the risk of CNS involvement. Between 60% and 85% of patients will develop CNS localization within 29 months.2 Publications on subsequent treatments are scarce. Temozolomide (TMZ) is a well-tolerated, second-generation alkylating agent with excellent penetration into to the CNS and the cerebrospinal fluid (CSF). Its activity in PCNSL has been demonstrated in small studies,3-6 but never in PVRL. Here, we present the results of a retrospective, multicentric study evaluating the efficacy and safety of TMZ as treatment of relapsed or refractory (R/R) patients or in patients not eligible for intensive treatment in first line.
Adults with R/R PVRL or not eligible for intensive treatments were treated with TMZ orally at a dose of 150 mg or 200 mg/m2, 5 days a month, every month. TMZ was proposed through the French National Lymphome Oculo-Cérébral (LOC) network, which brings together experts on PVRL and gives therapeutic guidelines. In accordance with the Declaration of Helsinki, the study was approved by the LOC network institutional review board. The diagnosis of PVRL was made by cytological examination and staining with anti-CD3, anti-CD20, and anti-CD79b antibodies of vitrectomy material or by histological examination of cerebral biopsy in case of associated brain lesions. Interleukin (IL)-10 and IL-6 assays were performed on the vitreous and/ or the aqueous humor (AH) using the Cytometric Bead Array (CBA) technique.7 The IL Score for intraOcular Lymphoma Diagnosis (ISOLD), with a published sensitivity and specificity of 93% and 95%, respectively, was used.7 According to the literature, an ISOLD score >4.6 in vitreous or AH has a 99% probability of PVRL. Clonality assay was performed on the vitreous by multiplex polymerase chain reaction–based analysis of immunoglobulin heavy and light chain gene rearrangements following the BIOMED-2 protocol.8 Patients underwent gadolinium magnetic resonance imaging (MRI), lumbar puncture, and positron emission tomography or computed tomography scan. CSF was analyzed by cytology, flow cytometry, clonality, and IL-10 and IL-6 assays when possible. Responses were evaluated by ophthalmological examination, supplemented by gadolinium MRI in the case of associated brain lesions, according to International Primary CNS Lymphoma Collaborative Group criteria.9 IL levels were measured periodically in the AH during the follow-up of some patients. Survival functions and median survivals were estimated using the Kaplan-Meier method, and survival rates and 95% confidence intervals (CIs) were calculated using Greenwood variance.
Twenty-one patients were included. Median age was 75 years (range, 35-90 years), 1 patient had associated brain localization, and all patients tested for CSF were negative. Nine patients had received 1 line of treatment before, 7 patients 2 lines, 2 patients 3 lines, and 1 patient 4 lines. Two patients, aged 85 and 90 years, had never been treated before. Anterior lines treatments contained high-dose (HD) methotrexate (MTX) for 14 patients. The remaining 5 patients had received HD cytarabine (n = 3) or intravitreous injections of MTX (n = 2). Two patients had received an autologous stem cell transplant (ASCT), and 1 patient cerebral radiotherapy prior to TMZ. One patient with cerebral localization was refractory to lenalidomide, and 2 patients were refractory to ibrutinib. Median duration of treatment with TMZ was 5.2 months (range, 1-40 months). Patients’ therapeutic history is summarized in Table 1.
B-cell clonality was positive in 14 out of 15 tested patients. In the single patient with a negative search for clonality, the diagnosis was confirmed by cytological test and antibodies, and the IL-10/ IL-6 ratio was 30. Median IL-10 concentration in the vitreous was 1805 pg/mL (range, 346-5000 pg/mL). IL-10/IL-6 ratio was >1 in the 13 tested patients (range, 1.4-29.6).
Median follow-up was 42 months (range, 9-115). Overall response rate was 81%; 15 patients (71%) achieved a CR, including 1 patient with an ocular and cerebral response and 2 patients (10%) with a PR. Four patients had PD. Median duration of response was 10.9 months (range, 3-109 months). At last follow-up, 6 patients are still in CR, including 1 treated in front line, and all but one had stopped TMZ. One patient who had previously received an ASCT is still in CR 115 months after starting treatment. Three patients previously treated with lenalidomide (n = 1) or ibrutinib (n = 2) obtained a CR. Some patients described a visual acuity improvement within a few days. For the patients tested, the course of the IL-10 assays dramatically decreased during the treatment of 9 responder patients and increased for 1 patient who had PD 8 months later.
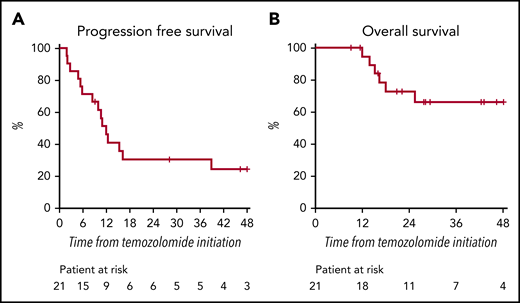
Median PFS was 12 months (95% CI, 8 not reached) with a 2-year PFS of 30% (95% CI, 15-61) (Figure 1A). Median OS has not yet been reached (Figure 1B). Six patients died, 5 due to cerebral progression.
Three patients had grade 3 toxicities (anemia and vomiting), and 1 patient had 2 grade 4 toxicities (neutropenia and thrombocytopenia). One patient requested discontinuation because a feeling of worsening of memory impairment, but neurological dysfunction had begun before TMZ.
We report here a cohort of patients with PVRL treated with TMZ. The results of molecular and cytokine analyses propose strong arguments for the correct diagnosis and follow-up. First-line IV chemotherapy usually consists of a combination of HD MTX and cytarabine.10,11 There have been few publications on R/R PVRL and no consensus exists in this field.12-19 The interest of ASCT in R/R patients has been demonstrated but it concerns only younger patients.20-22 The efficacy of lenalidomide in PVRL and PCNSL was suggested by Rubenstein et al12 and was recently confirmed by the LOC network13,14 . Similarly, ibrutinib has been reported to be effective in the treatment of PVRL and PCNSL.15,16 However, PFS with these 2 new drugs remains short, toxicity is not negligible, and costs are high.
PFS and OS of patients with isolated PVRL treated in first line are 29.6 and 58 months,23 and there are no OS data in R/R patients, but the prognosis is usually poor. In our study, patients had usually been treated several times, and some patients had received 4 lines of treatment or ibrutinib, lenalidomide, or ASCT, thus representing a population with a very poor prognosis and very limited available treatments. All but 1 patient treated for R/R PRVL had received MTX and/or aracytine before, which represents the main drugs for vitreal localization. Few patients received rituximab or local radiation because of lack of proof efficacy.
We report an excellent overall response rate of 81%, a CR of 71%, and long-lasting responses, which represent the best response rates published to date. Median OS was not reached, while median follow-up was 42 months.
No unusual toxicity was observed and, compared with chemotherapy, radiotherapy, lenalidomide, and ibrutinib, TMZ seems to be better tolerated. Comparatively, HD aracytine is associated with 62% grade 3 or 4 myelosuppression.24
In economic terms, the very low ratio of cost/benefits of TMZ compared with other drugs used in the same clinical context and until progression, such as ibrutinib or lenalidomide, appears to be very interesting. Limitations include retrospective analysis and lack of central pathologic confirmation.
In summary, TMZ, with its very high overall response rate, low toxicity, and affordable cost, can be considered as a very good therapeutic option and a new reference for elderly patients or R/R PVRL.
For original data, please e-mail the corresponding author.
Acknowledgment
The authors acknowledge Network LOC for patients' recruitment and treatments' decisions.
Authorship
Contribution: M. Baron and S.C. wrote the manuscript; L.B. analyzed statistical data; N.C., C.F., M. Blaizeau, C.S., C.H., K.H.-X., E.G., M.-L.L.L., A.L., P.S., B.B., V.L., V.T., M.-H.E., and D.R.-W. took care of the patients and completed the data forms for the study; C.S., C.H., K.H.-X., P.S., E.G., and S.C. participated in the LOC network and made treatment decisions; and K.M., M.C., and M.L.G.-T. provided biological diagnosis.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Marine Baron, Groupe Hospitalo-Universitaire Pitié-Salpêtrière, 47-83 Boulevard de l’Hôpital, 75013 Paris, France; e-mail: marine.baron@aphp.fr.