Key Points
CD34+c-Kit+Flt3+ myeloid progenitors from BM and blood of high-risk LCH patients give rise in vitro to BRAF-mutated LC-like cells.
BRAF mutation–carrying cells found in blood and lesions of low-risk LCH patients are likely derived from oligopotent progenitors.
Abstract
Langerhans cell histiocytosis (LCH) is a myeloid neoplasia, driven by sporadic activating mutations in the MAPK pathway. The misguided myeloid dendritic cell (DC) model proposes that high-risk, multisystem, risk-organ–positive (MS-RO+) LCH results from driver mutation in a bone marrow (BM)-resident multipotent hematopoietic progenitor, while low-risk, MS-RO− and single-system LCH would result from driver mutation in a circulating or tissue-resident, DC-committed precursor. We have examined the CD34+c-Kit+Flt3+ myeloid progenitor population as potential mutation carrier in all LCH disease manifestations. This population contains oligopotent progenitors of monocytes (Mo’s)/macrophages (MΦs), osteoclasts (OCs), and DCs. CD34+c-Kit+Flt3+ cells from BM of MS-RO+ LCH patients produced Langerhans cell (LC)-like cells in vitro. Both LC-like and DC offspring from this progenitor carried the BRAF mutation, confirming their common origin. In both high- and low-risk LCH patients, CD34+c-Kit+Flt3+ progenitor frequency in blood was higher than in healthy donors. In one MS-RO+ LCH patient, CD34+c-Kit+Flt3+ cell frequency in blood and its BRAF-mutated offspring reported response to chemotherapy. CD34+c-Kit+Flt3+ progenitors from blood of both high- and low-risk LCH patients gave rise to DCs and LC-like cells in vitro, but the driver mutation was not easily detectable, likely due to low frequency of mutated progenitors. Mutant BRAF alleles were found in Mo’s /MΦs, DCs, LC-like cells, and/or OC-like cells in lesions and/or Mo and DCs in blood of multiple low-risk patients. We therefore hypothesize that in both high- and low-risk LCH, the driver mutation is present in a BM-resident myeloid progenitor that can be mobilized to the blood.
Introduction
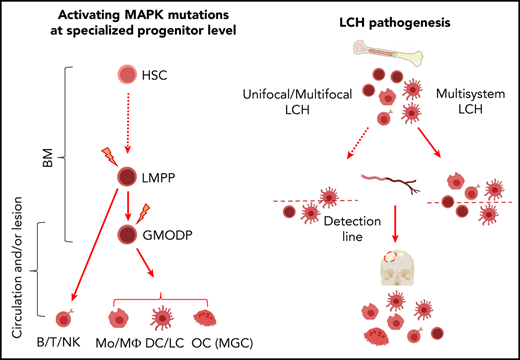
Langerhans cell histiocytosis (LCH) is a myeloid cell neoplasm affecting children and adults, with manifestations ranging from easy-to-treat solitary lesions to life-threatening disseminated disease.1,2 Single-system (SS) LCH is characterized by 1 or multiple LCH lesions confined to 1 tissue or organ and discerned into unifocal (UF) or multifocal (MF) LCH when occurring in bone or skin.3 Multisystem (MS) LCH is characterized by concurrent lesions in multiple organ systems, including skin, bone, lung, lymph node, gastrointestinal tract, or central nervous system. It is called MS-RO+ LCH when liver, spleen, and/or bone marrow (BM) are involved, and this disease entity has a considerable mortality rate.4,5 Treatments range from wait-and-watch or topical application of anti-inflammatory drugs in UF LCH to systemic treatment with conventional chemotherapeutic drugs for MF bone and MS LCH. Current treatments induce disease remission in most pediatric patients,5 but prognostic markers are needed that identify patients at risk of first-line treatment failure, or patients at risk of disease recurrence after response to initial therapy. Ideally, such markers also guide treatment decisions.3
LCH lesions are hallmarked by Langerhans cell (LC)-like cells (LCH cells) expressing CD1a and variable levels of Langerin (CD207). These cells are clonal6-8 but lack gross chromosomal alterations.9 LCH-affected tissues furthermore display a myeloid cell–dominated infiltrate of activated immune cells, irrespective of disease severity.1,8 The myeloid cell population in LCH lesions includes monocytes (Mo’s), macrophages (MΦs), dendritic cells (DCs), and osteoclast (OC)-like cells that are termed multinucleated giant cells (MGCs). Apart from myeloid cells, lesions also contain T, B, and natural killer (T) cells.1,10 LCH presents as an inflammatory disease,11-14 but the discovery of somatic driver mutations in genes encoding MAPK pathway components identified it as a neoplasm.15 Whole-exome DNA sequencing confirmed that LCH lesions contain CD1a+ cells with mutation in BRAF, MAP2K1, or, incidentally, other genes encoding proteins in the MAPK pathway,16 which generally occur in an otherwise wild-type genome.17,18 These mutations constitutively activate the MAPK pathway, leading to enhanced cell proliferation and survival, which classify LCH as a myeloid neoplastic disease.
Detection of mutant BRAF alleles in BM-derived CD34+ cells and in blood-derived myeloid cells, B cells, and (sporadically) T cells19,20 of patients with MS-RO+ LCH provided the first evidence that LCH may be prompted by somatic BRAF driver mutation in a multipotent hematopoietic progenitor. Mutant BRAF alleles could not be detected in unfractionated peripheral blood mononuclear cells (PBMCs) of patients with SS LCH.19 Expression of BRAFV600E under the CD11c promotor in transgenic mice gave rise to inflammatory lesions in lung and liver, characterized by accumulation of myeloid cell types, including LCH-like CD11c+CD207+ cells.19 Based on information from LCH patients and transgenic mice, the “misguided myeloid DC differentiation” model of LCH pathogenesis was proposed, wherein disease severity is determined by the specific stages of hematopoietic development when somatic mutation occurs.21-24 The model proposes that neoplastic LCH cells in MS-RO+ patients originate from a BM-resident hematopoietic progenitor, while LCH cells in low-risk LCH would originate from a DC-committed precursor cell present in the circulation or in the lesion. However, the cell of origin that first acquires the driver mutation in low-risk LCH remains to be clarified.
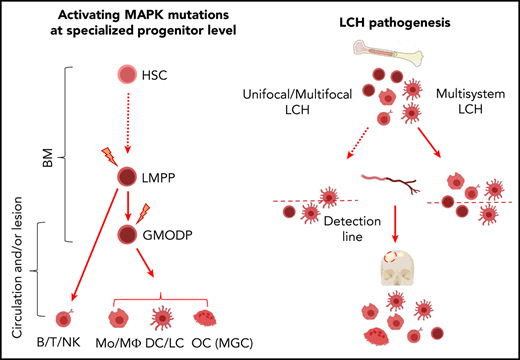
We recently reported that the Lin−CD34+c-Kit+Flt3+ cell population in human BM and blood that is commonly termed the granulocyte/MΦ progenitor (GMP),25,26 contains the granulocyte/macrophage/osteoclast/dendritic cell progenitor (GMODP), a progenitor oligopotent for formation of neutrophilic granulocytes, MΦs, OCs, and DCs, as well as the downstream MODP, which is devoid of granulocyte potential but oligopotent for the other 3 lineages.27 The (G)MODP gives rise to multiple myeloid cell types that are commonly found in LCH lesions. Therefore, we tested in the current study the hypothesis that both in high-risk and low-risk LCH, the (G)MODP population may contain cells that carry the driver mutation and give rise to mutation-carrying Mo’s/MΦs, DCs, LCs, and OCs.
Materials and methods
Patient samples
This study (P10.163.A1) was approved by the Leiden University Medical Center Institutional Review Board and the Dutch Childhood Oncology Group (OC2011-011). The use of LCH biopsy specimens was also approved under University of Pittsburgh Institutional Review Board protocol PRO12110055. Blood, BM, and/or LCH lesion samples were obtained at first disease onset (n = 22) or LCH relapse (n = 6) (supplemental Table 1, available on the Blood Web site). Cryopreserved blood (n = 39) and BM (n = 4) from healthy hematopoietic stem cell donors were retrieved from the biobank of the Willem Alexander Children’s Hospital. In compliance with the Declaration of Helsinki, patients (or their parents/legal guardians) and healthy donors (HDs) consented to use of their biological materials for research. For isolation and preservation of cell samples, see supplemental Materials and methods.
IHC and cell isolation by laser-guided microdissection
Tissue sections (5 µm) were cut from LCH-containing formalin-fixed paraffin-embedded (FFPE) tissue and deposited on high adhesive glass slides (Waldemar Knittel). Presence of myeloid cell types or cytoplasmic expression of BRAFV600E protein by these cells was analyzed by manual or automated28 immunohistochemistry (IHC) staining protocols. To prepare LCH tissue sections for laser-guided microdissection, 5-µm tissue sections were deposited on UV-activated polyethylene naphthalate membrane slides (Zeiss). Sections were treated and stained as explained in supplemental Materials and methods using antibodies listed in supplemental Table 2.
Flow cytometry
For flow cytometric isolation of cells, cryopreserved cells were thawed and washed in RPMI-1640 medium with 2% fetal calf serum (FCS) and incubated with a sorting panel of antibodies (supplemental Table 2) for 45 minutes in the dark at 4°C. LIVE/DEAD Fixable near-infrared fluorescent reactive dye (Invitrogen) was added to exclude dead cells. Prior to sorting, DNAse (Sigma-Aldrich) was added to prevent cell aggregation. Cell sorting was performed on a FACSAria or FACSAria Fusion (BD Biosciences). In vitro–cultured cells were stained with a panel of diagnostic antibodies (supplemental Table 2) in phosphate-buffered saline with 1% FCS for 45 minutes in the dark on ice and analyzed on an LSR Fortessa (BD Biosciences). Data were analyzed using FlowJo software (TreeStar).
In vitro differentiation of progenitor cells into DCs or LCs
Lin−CD34+c-Kit+Flt3+ cells were sorted flow cytometrically, seeded at 1000 to 2000 cells per well in round-bottom 96-well plates (BD Falcon), and cultured at 5% CO2 and 37°C in Iscove modified Dulbecco medium containing 10% FCS and relevant cytokines. To induce DC differentiation, cells were cultured for 2 to 3 weeks in medium with 100 ng/mL recombinant human (rh) Flt3 ligand (Flt3L), 0.1 ng/mL rh macrophage colony-stimulating factor (M-CSF), 5 ng/mL rh thrombopoietin (TPO), 10 ng/mL rh interleukin-3 (IL-3), and 5 ng/mL rh stem cell factor (SCF). To induce LC differentiation, cells were cultured for 2 to 3 weeks in medium with 5 ng/mL rh SCF, 5 ng/mL rh TPO, 10 ng/mL rh granulocyte macrophage colony-stimulating factor (GM-CSF), 5 ng/mL rh transforming growth factor β (TGF-β), 2.5 ng/mL rh tumor necrosis factor α (TNF-α), and 0.25 ng/mL rh M-CSF. Growth factors were from R&D Systems or Peprotech. Irradiated mesenchymal stromal cells were used as feeder layers.
BRAF mutation detection by next-generation sequencing or ddPCR
An automated method29 was used to extract nucleic acid from FFPE tissue fragments obtained by manual scraping or laser-guided microdissection or from cells flow-cytometrically sorted from thawed BM, blood, or tissue biopsy specimens. FFPE tissue–derived DNA samples were screened for the presence of BRAFV600E or BRAFV600D by targeted sequencing using custom-designed next-generation sequencing panels implemented in routine procedures performed by the pathology departments of the University Medical Centers of Leiden, Amsterdam, or Utrecht. For DNA analysis by droplet-digital polymerase chain reaction (ddPCR),30 simultaneous duplex amplification of BRAFV600E or BRAFV600D and BRAFWT sequences was performed on a QX200TM system using ddPCR Supermix for probes and ddPCR Mutation Assay for BRAFV600E (dHsaMDV2010027) and BRAFV600D (dHsaMDS526433948), respectively, according to the manufacturer’s instructions (Bio-Rad Laboratories) (supplemental Figure 1). For data analysis, see supplemental Materials and methods.
Statistical analysis
Nonparametric (Mann-Whitney) U testing using GraphPad Prism 8.0.1 software was used for comparing precursor cell frequencies analyzed in healthy control- and LCH patient-derived samples, respectively.
Results
DC and LC offspring from BM-derived Lin−CD34+c-Kit+Flt3+ progenitors of MS-RO+ LCH patients carry BRAFV600E
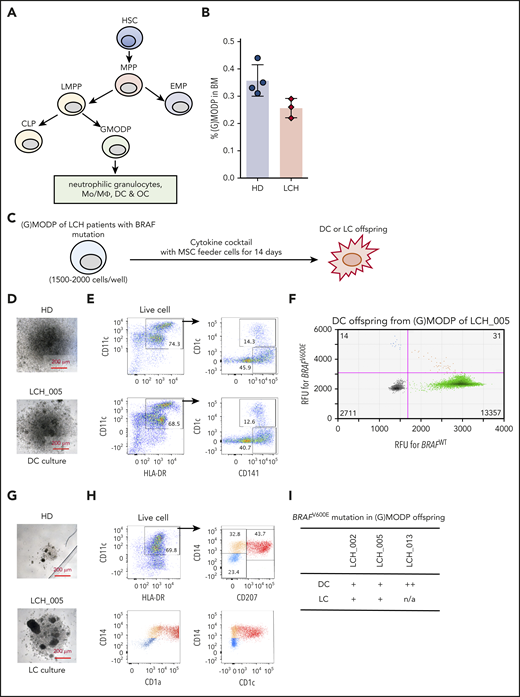
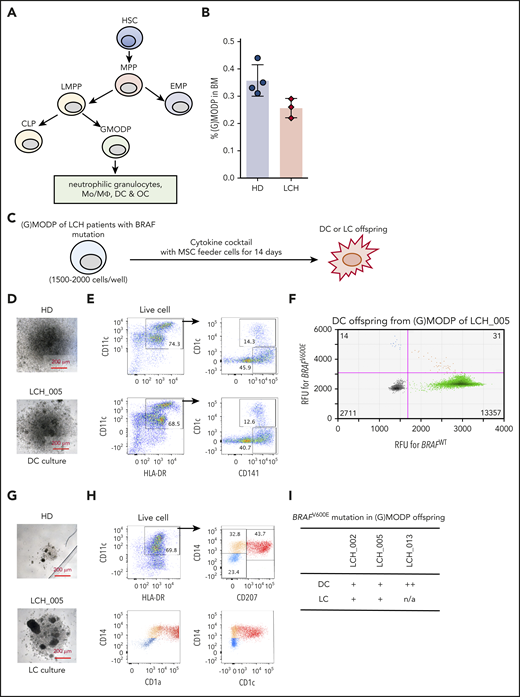
The position of the GMODP in the hematopoietic tree is schematically indicated in Figure 1A.26,27 We have shown that the Lin−CD34+c-Kit+Flt3+ population (supplemental Figure 2A) harbors GMODP cells and downstream, more committed macrophage/osteoclast/dedritic cell progenitor (MODP) cells that can be discerned on basis of interleukin-3 receptor α expression.27 In the current study, we have examined the total Lin−CD34+c-Kit+Flt3+ population that we call the granulocyte (G)MODP population. Our general hypothesis was that both in high-risk and in low-risk LCH patients, the driver mutation may be present in the (G)MODP population and thereby give rise to Mo’s/MΦs, DCs, OCs, and possibly LCs that all carry the driver mutation. To address this hypothesis, we first examined 3 high-risk MS-RO+ patients with a confirmed BRAFV600E mutation in their biopsy specimens (supplemental Table 1). MS-RO+ LCH patients reportedly acquire the driver mutation in CD34+ progenitor cells.19,31 We therefore expected mutated cells to be present within the (G)MODP population and its offspring. The frequency of (G)MODP cells in BM of the 3 patients examined and 4 age-matched HDs was not significantly different (Figure 1B).
Detection of BRAF mutation-bearing GMODP in the BM of high-risk LCH patients. (A) Scheme depicting the position of GMODP in the hematopoietic tree. The GMODP is a redefinition of the GMP, based on defined oligopotency, as determined by dedicated in vitro differentiation cultures.26 CLP, common lymphoid progenitor; EMP, erythroid-myeloid progenitor; HSC, hematopoietic stem cell; LMPP, lymphoid-primed myeloid progenitor; MPP, multipotent progenitor. (B) The frequencies (%) of (G)MODP (Lin−CD34+c-Kit+Flt3+) cells among total live cells in BM samples of MS-RO+ LCH patients (LCH_002, 005, and 013) and 4 age-matched HDs, as determined by flow cytometry. For the gating strategy used, see supplemental Figure 2A. (C) Culturing scheme. (D-I) (G)MODP cells were sorted from BM of LCH_005 and cultured under DC-differentiation conditions (Flt3L; D-F) or LC-differentiation conditions (TGF-β and TNF-α; G-I) and analyzed for appearance by light microscopy (D,G), cell-surface phenotype by flow cytometry (E,H), and BRAFV600E mutation status by ddPCR (F,I). (F) Droplets containing BRAFWT or BRAFV600E PCR products are depicted by the green and blue dots, respectively. Orange dots represent droplets containing both PCR products. The numbers of droplets analyzed are indicated in the quadrants. (I) BRAF mutation scoring method (−, ±, +, ++, and +++) is explained in supplemental Materials and methods and supplemental Figure 1B. RFU, relative fluorescence units.
Detection of BRAF mutation-bearing GMODP in the BM of high-risk LCH patients. (A) Scheme depicting the position of GMODP in the hematopoietic tree. The GMODP is a redefinition of the GMP, based on defined oligopotency, as determined by dedicated in vitro differentiation cultures.26 CLP, common lymphoid progenitor; EMP, erythroid-myeloid progenitor; HSC, hematopoietic stem cell; LMPP, lymphoid-primed myeloid progenitor; MPP, multipotent progenitor. (B) The frequencies (%) of (G)MODP (Lin−CD34+c-Kit+Flt3+) cells among total live cells in BM samples of MS-RO+ LCH patients (LCH_002, 005, and 013) and 4 age-matched HDs, as determined by flow cytometry. For the gating strategy used, see supplemental Figure 2A. (C) Culturing scheme. (D-I) (G)MODP cells were sorted from BM of LCH_005 and cultured under DC-differentiation conditions (Flt3L; D-F) or LC-differentiation conditions (TGF-β and TNF-α; G-I) and analyzed for appearance by light microscopy (D,G), cell-surface phenotype by flow cytometry (E,H), and BRAFV600E mutation status by ddPCR (F,I). (F) Droplets containing BRAFWT or BRAFV600E PCR products are depicted by the green and blue dots, respectively. Orange dots represent droplets containing both PCR products. The numbers of droplets analyzed are indicated in the quadrants. (I) BRAF mutation scoring method (−, ±, +, ++, and +++) is explained in supplemental Materials and methods and supplemental Figure 1B. RFU, relative fluorescence units.
We have previously shown that (G)MODP cells cultured with Flt3L give rise to DCs, which represents the homeostatic, noninflammatory pathway.27 To test whether the (G)MODP population can also generate LC-like cells, we replaced Flt3L with TGF-β and TNF-α, which can drive differentiation of LC-like cells from CD34+ progenitors32,33 (Figure 1C). (G)MODP cells from patient LCH_005 and an age-matched HD tested in the same experiment had similar DC-differentiation potential, according to DC yield and phenotype, as judged by light microscopy and flow cytometry, respectively (Figure 1D-E). DC offspring were defined as HLA-DR+CD11c+ cells that coexpressed CD1c and/or CD141 (Figure 1E). DC offspring contained mutant BRAF alleles, as demonstrated by ddPCR (Figure 1F). Under LC-differentiation conditions, the (G)MODP cells from patient LCH_005 gave more offspring than the (G)MODP from the HD, as judged by light microscopy (Figure 1G). The offspring cells included LC-like cells, defined as HLA-DR+CD11c+CD207+CD1a+ (Figure 1H). A second independent experiment with BM from LCH_005 and the same HD gave similar results (data not shown). For all 3 MS-RO+ LCH patients and 1 HD, the frequencies of DC and LC-like output from BM-derived (G)MODP cells were comparable (supplemental Figure 2B). Cells with LC-like phenotype were detected in (G)MODP offspring generated under LC- but not DC-differentiation conditions (supplemental Figure 2C).
The BRAFV600E driver mutation was detected in the in vitro generated LC-like and/or DC offspring cells from the BM-derived (G)MODP in all 3 LCH patients analyzed (Figure 1I). We could not easily detect the mutation in the (G)MODP population itself. This can be explained by the low frequency of the (G)MODP in BM27 (Figure 1B). Progenitor cell yields after cell sorting were generally low, limiting the number of cells that could be seeded and thereby the likelihood to detect BRAF-mutated cells. Our data show that the DC/LC-differentiation cultures increased the chance to detect the mutation. Differentiation cultures using, side-by-side, flow-cytometrically sorted HSC, LMPP, and (G)MODP cells from BM of LCH_005 (supplemental Figure 3A-B) showed that DC offspring from the LMPP and (G)MODP contained the BRAFV600E mutation (supplemental Figure 3C-D). In agreement with the mutation being present in the LMPP population, it was also detected in B cells of patient LCH_005 (supplemental Figure 3E). The low frequency of HSC limited the number of cells that could be seeded, and therefore, mutation analysis was not robust. Titration indicated that the frequency of BRAF-mutated cells in these progenitor populations was ≤0.1% (supplemental Figure 3D). These data indicate that in MS-RO+ LCH, the BRAFV600E mutation incurs at the level or upstream of the GMODP in the pathway of hematopoiesis.
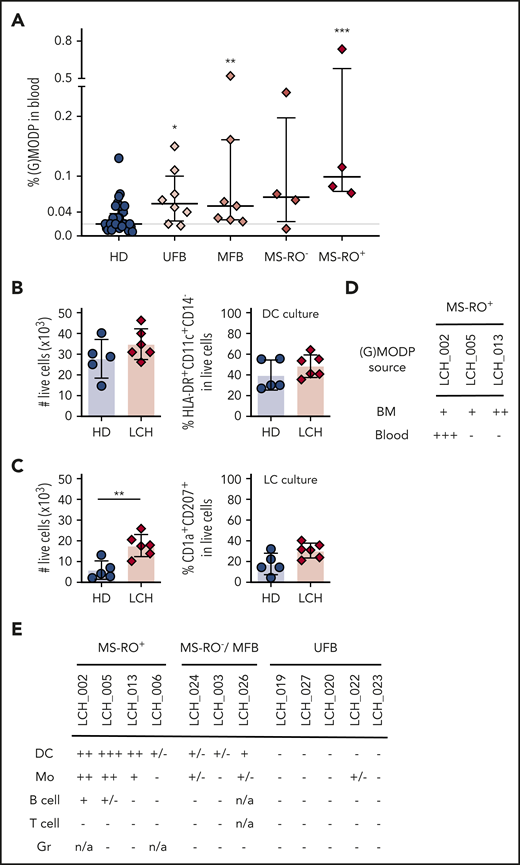
The (G)MODP population in the blood is elevated in LCH patients
Since BM is not routinely sampled from patients suspected of LCH,34 we examined blood as alternative source to isolate mutation-carrying (G)MODP cells. (G)MODP cell frequency in PBMCs was analyzed by flow cytometry in 23 patients diagnosed with LCH according to clinical symptoms (supplemental Table 1). The (G)MODP cell frequency in blood was significantly higher in LCH patients than in age-matched HDs (Figure 2A). MS-RO+ patient LCH_002 had an exceptionally high frequency of (G)MODP cells in the blood before therapy (Figure 2B). This patient was eventually treated with salvage chemotherapy and has remained relapse-free. Remarkably, the (G)MODP cell frequency dropped to HD levels during therapy and remained at this level (Figure 2B).
GMODP frequency in blood reports response to therapy in 1 case study. (A) The frequencies (%) of (G)MODP cells among total live cells in PBMC samples of 23 LCH patients (supplemental Table 1) collected at first disease onset or relapse (prior to (n = 20) or after the start of systemic therapy (n = 3)) and age-matched HDs (n = 36), as determined by flow cytometry. Error bars indicate median and interquartile range. Indicated P value was determined by Mann-Whitney U test (**P < .01). The gray area indicates the calculated progenitor frequency range based on HD data. (B) Frequencies (%) of (G)MODP cells among live cells in the blood of MS-RO+ patient LCH_002 at diagnosis (before therapy [pre Tx]), 1 month after starting chemotherapy (on Tx), and 3 years after finishing salvage chemotherapy (post Tx) and an age-matched HD. (C-H) (G)MODP cells sorted from blood samples of LCH_002 taken before, during, or after therapy and an age-matched HD were seeded at 1000 cells per well and cultured under LC-differentiation conditions. (C) Light microscopic appearance of the cultures. (D) Absolute number of live cells per well at the end of the culture period. (E) Flow cytometry plots depicting the gating strategy for sorting of (G)MODP offspring cells for BRAF mutation analysis. (F) Flow cytometry plots depicting CD207+CD1a+ LC-like cells within HLA-DR+CD11c+CD14+ offspring cells. (G-H) DNA was extracted from the G1, G2, and G3 fractions of the cultured cells, sorted as depicted in panel E and analyzed for the presence of the BRAF mutation by ddPCR. (G) Representative 2-dimensional ddPCR plot depicting BRAFV600E mutation in (G)MODP-derived HLA-DR+CD14-CD11c+ offspring cells from LCH_002. (H) BRAFV600E mutation status as detected by ddPCR of (G)MODP offspring cells of the indicated phenotypes (sorted as indicated in panel E). (I) Flow cytometry plots depicting the gating strategy for sorting of DCs and Mo’s from blood samples of LCH_002. (J) Corresponding BRAFV600E mutation status of DCs and Mo’s identified by the listed markers.
GMODP frequency in blood reports response to therapy in 1 case study. (A) The frequencies (%) of (G)MODP cells among total live cells in PBMC samples of 23 LCH patients (supplemental Table 1) collected at first disease onset or relapse (prior to (n = 20) or after the start of systemic therapy (n = 3)) and age-matched HDs (n = 36), as determined by flow cytometry. Error bars indicate median and interquartile range. Indicated P value was determined by Mann-Whitney U test (**P < .01). The gray area indicates the calculated progenitor frequency range based on HD data. (B) Frequencies (%) of (G)MODP cells among live cells in the blood of MS-RO+ patient LCH_002 at diagnosis (before therapy [pre Tx]), 1 month after starting chemotherapy (on Tx), and 3 years after finishing salvage chemotherapy (post Tx) and an age-matched HD. (C-H) (G)MODP cells sorted from blood samples of LCH_002 taken before, during, or after therapy and an age-matched HD were seeded at 1000 cells per well and cultured under LC-differentiation conditions. (C) Light microscopic appearance of the cultures. (D) Absolute number of live cells per well at the end of the culture period. (E) Flow cytometry plots depicting the gating strategy for sorting of (G)MODP offspring cells for BRAF mutation analysis. (F) Flow cytometry plots depicting CD207+CD1a+ LC-like cells within HLA-DR+CD11c+CD14+ offspring cells. (G-H) DNA was extracted from the G1, G2, and G3 fractions of the cultured cells, sorted as depicted in panel E and analyzed for the presence of the BRAF mutation by ddPCR. (G) Representative 2-dimensional ddPCR plot depicting BRAFV600E mutation in (G)MODP-derived HLA-DR+CD14-CD11c+ offspring cells from LCH_002. (H) BRAFV600E mutation status as detected by ddPCR of (G)MODP offspring cells of the indicated phenotypes (sorted as indicated in panel E). (I) Flow cytometry plots depicting the gating strategy for sorting of DCs and Mo’s from blood samples of LCH_002. (J) Corresponding BRAFV600E mutation status of DCs and Mo’s identified by the listed markers.
In the (G)MODP taken directly from blood of LCH_002, mutant BRAF alleles were represented at the limit of detection of the ddPCR assay (data not shown). We used in vitro differentiation cultures to monitor BRAFV600E-mutated cells in the (G)MODP offspring. (G)MODP cells from blood samples, taken before, on, and after therapy (supplemental Figure 4), were cultured under LC-differentiation conditions. (G)MODP cells isolated before therapy produced the most offspring (Figure 2C-D), with the highest proportion of CD207+ LC-like cells (Figure 2E-F). In LC offspring of the (G)MODP from blood isolated before therapy, the BRAFV600E mutation was detected in 3 distinct HLA-DR+ populations (Figure 2E). Interestingly, the mutation was undetectable in LC offspring from the (G)MODP isolated during or after therapy (Figure 2G-H). In DCs and Mo’s that were isolated directly from blood on the basis of the indicated phenotypes (Figure 2I), the BRAFV600E mutation was detected before and during, but not after, therapy (Figure 2J). These data suggest that the (G)MODP frequency in blood of LCH patients may be correlated with disease status, which will be of interest to examine in a cohort study. They furthermore highlight that generating offspring from blood-derived (G)MODP in vitro can facilitate detection of LCH driver mutations.
BRAF mutation–carrying myeloid cells in blood of both high- and low-risk LCH patients
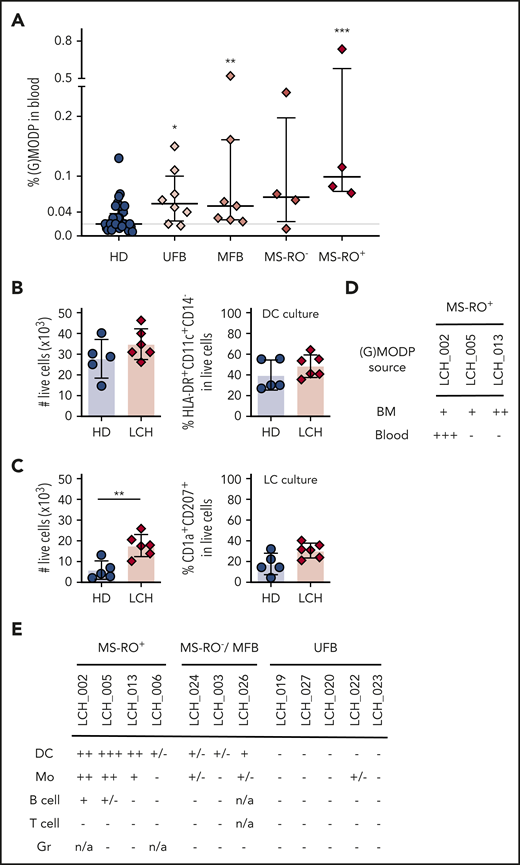
Upon dissection of LCH patients in different disease manifestations, it appeared that both in low-risk and high-risk LCH patients, the (G)MODP cell frequency in blood was significantly elevated as compared with age-matched HD (Figure 3A). Overall, white blood cell and platelet counts were not different between patients and HD, indicating that the increase in (G)MODP frequency in LCH was specific to this progenitor population (supplemental Figure 5A).
Blood GMODP frequency is elevated in low-risk LCH patients and their myeloid cells contain the BRAF mutation. (A) The frequencies (%) of (G)MODP in blood from unifocal bone (UFB), multifocal bone (MFB), MS-RO−, and MS-RO+ LCH patients and age-matched HD. See supplemental Table 1 for clinical details of the patients. Error bars indicate median and interquartile range; P values according to statistical testing by Mann-Whitney U test (*P < .05; **P < .01, ***P < .001). (B-C) (G)MODP cells were sorted from blood, cultured under DC (B) or LC (C) differentiation, and counted and analyzed for cell-surface phenotype by flow cytometry. Live-cell yield from 500 (G)MODP and the frequencies (%) of DC (HLA-DR+CD11c+CD14−) or LC-like cells (HLA-DR+CD11c+CD1a+CD207+) among live (G)MODP-derived offspring cells are represented for HDs (n = 5) and patients LCH_002, 005, 006, 013 (MS-RO+), 014 (MFB), and 019 (UFB) (n = 6) (supplemental Table 1). P values according to statistical testing by Mann-Whitney U test (**P < .01). (D) GMODP from BM and blood of the indicated BRAFV600E genotyped high-risk MS-RO+ LCH patients were seeded at 1500 to 2000 cells per well and cultured under DC-differentiation conditions, and the BRAF mutation was assessed by ddPCR in offspring cells. (E) BRAF mutation status of DCs, Mo’s, B cells, T cells, and granulocytes (Gr) from indicated LCH patients (supplemental Table 1).
Blood GMODP frequency is elevated in low-risk LCH patients and their myeloid cells contain the BRAF mutation. (A) The frequencies (%) of (G)MODP in blood from unifocal bone (UFB), multifocal bone (MFB), MS-RO−, and MS-RO+ LCH patients and age-matched HD. See supplemental Table 1 for clinical details of the patients. Error bars indicate median and interquartile range; P values according to statistical testing by Mann-Whitney U test (*P < .05; **P < .01, ***P < .001). (B-C) (G)MODP cells were sorted from blood, cultured under DC (B) or LC (C) differentiation, and counted and analyzed for cell-surface phenotype by flow cytometry. Live-cell yield from 500 (G)MODP and the frequencies (%) of DC (HLA-DR+CD11c+CD14−) or LC-like cells (HLA-DR+CD11c+CD1a+CD207+) among live (G)MODP-derived offspring cells are represented for HDs (n = 5) and patients LCH_002, 005, 006, 013 (MS-RO+), 014 (MFB), and 019 (UFB) (n = 6) (supplemental Table 1). P values according to statistical testing by Mann-Whitney U test (**P < .01). (D) GMODP from BM and blood of the indicated BRAFV600E genotyped high-risk MS-RO+ LCH patients were seeded at 1500 to 2000 cells per well and cultured under DC-differentiation conditions, and the BRAF mutation was assessed by ddPCR in offspring cells. (E) BRAF mutation status of DCs, Mo’s, B cells, T cells, and granulocytes (Gr) from indicated LCH patients (supplemental Table 1).
We next cultured the (G)MODP from peripheral blood of 9 low-risk LCH patients with MS-RO−, MF, or UF bone disease and a confirmed BRAF mutation in affected tissue(s). We also included the 3 MS-RO+ patients, comparing differentiation cultures with the (G)MODP from either BM or blood. Age-matched HDs were used as controls. Confirming data with BM-derived (G)MODP of MS-RO+ patients (Figure 1), DC yield from blood-derived (G)MODP was higher than LC yield. Moreover, DC yield was similar for LCH patients and HDs (Figure 3B). Under LC-differentiation conditions, blood-derived (G)MODP from LCH patients gave more offspring cells than blood-derived (G)MODP from HDs, but the frequency of LC-like cells in the cultures was similar in both cases (Figure 3C).
For the 3 MS-RO+ LCH patients, we had access to both BM and blood samples, while for low-risk LCH patients, only blood is sampled. We used the material from the MS-RO+ patients to compare side by side whether the likelihood to detect the driver mutation in the DC offspring of BM- or blood-derived (G)MODP was distinct. In the DC offspring of BM-derived (G)MODP cells, the BRAF mutation was detected in all 3 MS-RO+ patients. However, in the DC offspring of blood-derived (G)MODP cells, the BRAF mutation was detected only in LCH_002 (Figure 3D; supplemental Figure 5B). We also performed mutation analysis on the DC offspring of the blood-derived (G)MODP from the 9 low-risk LCH patients. For this purpose, 1200 to 2000 (G)MODP cells were sorted (maximal yield) and cultured under DC-differentiation conditions to get the highest cell yield. In none of these cases was the mutation detected (data not shown).
We next examined whether the mutation could be detected in low-risk patients in blood-derived granulocytes, DCs, Mo’s, B cells, and/or T cells. These cells were isolated by flow cytometry from the blood (supplemental Figure 6A). MS-RO+ patients were included as positive controls. The BRAF mutation was detected in DCs and/or Mo’s in 4 out of 4 MS-RO+ patients, in 1 out of 1 MS-RO− patient, and in 3 out of 7 SS LCH patients, including UF bone patient LCH_022 (Figure 3E; supplemental Figure 6B). The BRAF mutation was also detected in blood B cells in 2 out of 4 MS-RO+ patients but in none of the T-cell or granulocyte samples analyzed (Figure 3E). The presence of the BRAF driver mutation in circulating DCs and/or Mo’s in low-risk LCH patients is consistent with the possibility that the driver mutation is present in a myeloid progenitor.
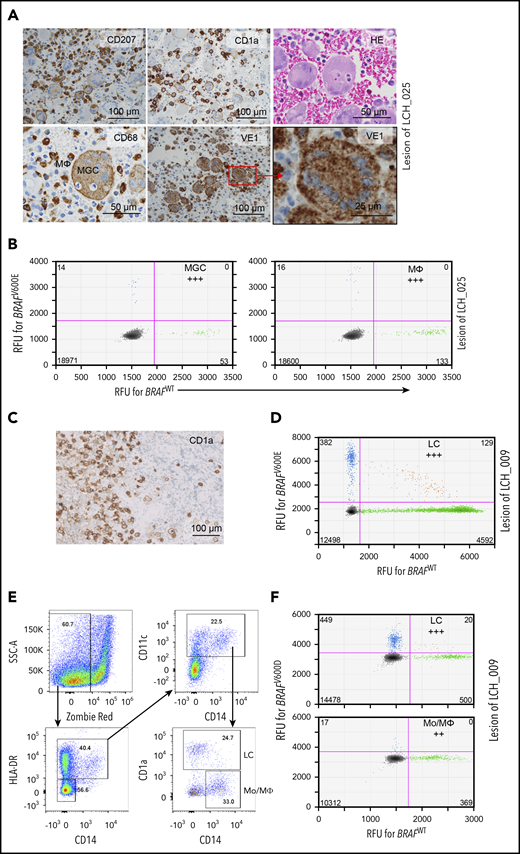
The BRAF driver mutation is present in multiple myeloid cell types in low-risk LCH lesions
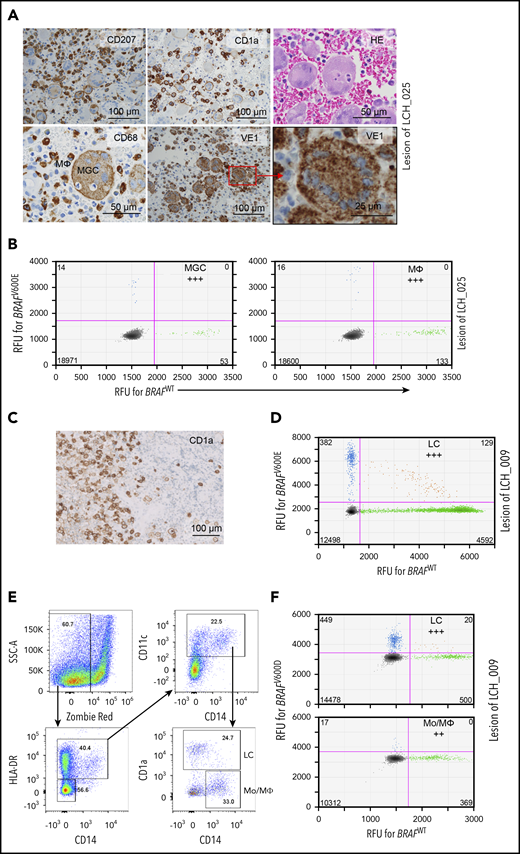
Our hypothesis predicts that in low-risk LCH, a mutation-carrying progenitor cell, possibly the (G)MODP, gives rise to different myeloid cell types present in LCH lesions. To test this possibility, we traced the BRAF driver mutation in MΦs and MGCs from LCH lesions of low-risk patients. Cells in the single osteolytic skull lesion of patient LCH_025 (supplemental Table 1) were variable in size and shape and included the prototypical CD1a+ and CD207+ LCH cells (Figure 4A). CD68+ cells in these lesions could be discerned as LCH cells with a paranuclear dot-like CD68 staining; smaller cells with diffuse, dark CD68 staining; and large cells with diffuse CD68 staining that are described in the LCH field as multinucleated OC-like MGCs.35 The VE1 antibody that is specific for BRAFV600E protein36,37 identified the driver mutation in LCH cells/MΦs and OC-like MGCs in the lesion of patient LCH_025 (Figure 4A). We isolated MGCs and LC/MΦ-like cells from the same FFPE lesion by laser-guided microdissection, based on a CD68+ phenotype and cell morphology (Figure 4A). The BRAFV600Emutation was detected in both LC/ MΦs and OC-like MGCs (Figure 4B). For MF bone patient LCH_009, we traced the BRAFV600D mutation in CD1a+ LC-like cells that had been microdissected from FFPE lesion tissue (Figure 4C-D) and myeloid cells that were flow-cytometrically sorted from cryopreserved cells derived from the other half of the biopsy specimen (Figure 4E-F). The cells were sorted based on an HLA-DR+CD11c+ phenotype and subdivided into CD14−CD1a+ LC-like cells and CD14+CD1a− Mo’s /MΦs. The mutation was found in both LC-like cells and Mo’s /MΦs. Similar results were obtained for UF bone patients LCH_001, LCH_022, and LCH_023 (supplemental Figure 7). These data indicate that in LCH lesions, not only the prototypical LCH cells but also Mo’s /MΦs and OC-like MGCs contain the BRAF driver mutation. MGCs may differentiate from Mo's or DCs,38 but the data are consistent with the idea that in low-risk MF and even in UF LCH, the driver mutation is present in the common progenitor of these cell types, the (G)MODP. Notably, in MFB patient LCH_009, the BRAFV600D mutation was detected in T cells in the lesion, while in UFB patient LCH_022, the mutation was detected in B cells in the lesion (supplemental Figure 8A-B). The fact that lymphoid cells carry the driver mutation in these patients argues that in low-risk patients, as in high-risk patients, the somatic mutation event may take place at, or even upstream of, the level of the LMPP.
Detection of the BRAF mutation in multiple myeloid cell types isolated from LCH lesions of SS LCH patients. (A) FFPE biopsy taken from the osteolytic lesion in the skull of UFB patient LCH_025. IHC was performed to stain LC-like cells (CD207, CD1a), MGCs, and MΦs (CD68 and morphology). Cells expressing mutant BRAFV600E protein were detected with VE-1 antibody. (B) Detection of the BRAFV600E mutation in MGCs and CD68+ LCs/MΦs microdissected from the skull lesion of patient LCH_025 by ddPCR; (C) IHC depicting CD1a+ LCH cells in the lesion of MFB patient LCH_009. (D) ddPCR 2-dimensional plot depicting BRAFV600D mutation detection in microdissected lesional CD1a+ cells of MFB patient LCH_009. (E) Gating strategy for flow cytometric sorting of HLA-DR+CD14+CD11c+CD1a+ LC-like cells and HLA-DR+CD14+CD11c+CD1a− Mo’s/MΦs from cryopreserved lesional cells of patient LCH_009. (F) Two-dimensional ddPCR plot depicting BRAFV600D mutation detection in sorted LC-like cells and Mo’s/MΦs of patient LCH_009.
Detection of the BRAF mutation in multiple myeloid cell types isolated from LCH lesions of SS LCH patients. (A) FFPE biopsy taken from the osteolytic lesion in the skull of UFB patient LCH_025. IHC was performed to stain LC-like cells (CD207, CD1a), MGCs, and MΦs (CD68 and morphology). Cells expressing mutant BRAFV600E protein were detected with VE-1 antibody. (B) Detection of the BRAFV600E mutation in MGCs and CD68+ LCs/MΦs microdissected from the skull lesion of patient LCH_025 by ddPCR; (C) IHC depicting CD1a+ LCH cells in the lesion of MFB patient LCH_009. (D) ddPCR 2-dimensional plot depicting BRAFV600D mutation detection in microdissected lesional CD1a+ cells of MFB patient LCH_009. (E) Gating strategy for flow cytometric sorting of HLA-DR+CD14+CD11c+CD1a+ LC-like cells and HLA-DR+CD14+CD11c+CD1a− Mo’s/MΦs from cryopreserved lesional cells of patient LCH_009. (F) Two-dimensional ddPCR plot depicting BRAFV600D mutation detection in sorted LC-like cells and Mo’s/MΦs of patient LCH_009.
Discussion
LCH derives its name from the presence of Birbeck granules in lesional cells, which are characteristic of epidermal LCs.39 Furthermore, the presence of cells with the LC marker protein Langerin (CD207) in LCH lesions is typical. However, micro-array-based transcriptomics showed that these LC-like cells are distinct from epidermal LCs.21,40 Epidermal LCs are tissue-resident MΦs that derive from mesenchymal cells during fetal development.41 LCH cells may stem from embryonic precursor cells,42 but LCH cells formed after birth likely have a hematopoietic origin.43 The discussion about the cell of origin in LCH is confusing, because pathways of myeloid cell lineage commitment are still being redrawn. Furthermore, mature myeloid cells can be plastic, as exemplified by the Mo that can differentiate into not only MΦs but also DCs, OCs,44 or LC-like cells.45 Also, CD1c+ DCs from the blood can reportedly differentiate into CD207+ cells.46 Classical and nonclassical Mo's, as well as CD1c+ cells in the blood of MS-RO+ LCH patients, have all been shown to express BRAFV600E alleles.31,47
A recent single-cell transcriptomic study of LCH phenotype cells from lesions has been very illuminating.8 LCH cells proved to reflect a spectrum of differentiation from noncommitted hematopoietic progenitors to diverse myeloid lineage commitment stages, specifically DCs, MΦs, and OCs. In LCH cells from lesions of both MS and SS patients, 2 populations with progenitor characteristics were found, including cells with high cell-cycle activity.8 Dividing, Ki67+ cells have been reported in LCH lesions48,49 but were not precisely defined. Halbritter et al8 mapped cell division to progenitor type LCH cells.8 This finding suggests that not only MS LCH but also SS LCH originates from a multipotent progenitor that may be present in the lesion. Our data support this concept.
BRAF-mutated CD34+ cells were previously found in BM of high-risk LCH patients19,31 and specified to LMPP cells in an adult MS-RO+ patient.31 However, we newly demonstrate here that the CD34+c-Kit+Flt3+ (G)MODP population derived from high-risk LCH patients can give rise to BRAF mutation–carrying DCs and LCs in vitro. This is the first time that we demonstrate LC-differentiation potential of the GMODP. The output from LCH patient–derived GMODP cultured under LC conditions was found to be higher than under DC conditions, while this is not the case for the HD-derived GMODP output. These data suggest that the presence of BRAF-mutated cells in the LC culture promotes the proliferation of the GMODP offspring cells. This effect is likely paracrine, because the mutated cells are only present at low frequency in these cultures according to ddPCR analysis. The frequency of BRAF-mutated cells in the BM-derived (G)MODP populations of MS-RO+ LCH patients was ∼1 in 2000. In patient LCH_002, the frequency was likely much higher, as this was the only patient in whom we also found the mutation in the progeny of blood-derived (G)MODP cells. This circulating, mutation-carrying (G)MODP population was not detectable in the blood after curative chemotherapy, indicating that its occurrence is associated with disease manifestation. To detect the mutation in the (G)MODP, sufficient numbers of cells need to be seeded to ensure the presence of rare, somatically mutated LCH cells within the population. In 2 out of 3 MS-RO+ LCH patients, the mutation was detected in offspring from BM-derived, but not blood-derived, (G)MODP cells, possibly reflecting a higher frequency of mutated cells in the BM. As we show here, in vitro culture of (G)MODP cells from BM and/or blood facilitates detection of the sporadic driver mutation and thereby may offer the opportunity to monitor response to therapy, initially in experimental settings.
In MS-RO+ patients, we found mutant BRAF alleles in DC- and Mo-like offspring of BM-derived (G)MODP. BRAF mutation–carrying cells with the same marker profiles were detected in the blood of the same patients, confirming earlier observations.19,31 However, we here identify BRAF driver mutations also in blood DCs and Mo’s of low-risk LCH patients. Presence of BRAF-mutated myeloid cells in the blood of 2 MF and 1 UF bone LCH patients is novel, while it was previously reported for 1 MS-RO− LCH patient.19 Remarkably, we did not detect the driver BRAF mutation in granulocytes isolated from the blood of either high- or low-risk LCH patients. This does not seem consistent with the presence of the mutation in GMODP that are progenitors of neutrophilic granulocyte. Possibly, the driver mutation is unfavorable for granulocytes and therefore selected against in the population. Also, requirements for granulocyte differentiation may not be met in the microenvironment of mutated progenitors.
We did not find BRAF-mutated cells among the offspring of the (G)MODP cells from blood in patients other than LCH_002. The small blood sample size limited the yield of (G)MODP cells, and therefore, mutated cells were likely not represented among the seeded cells. We hypothesize, nonetheless, that presence of BRAF-mutated HLA-DR+CD11c+ cells in the blood of both high-risk and low-risk LCH patients is an indicator of a BRAF-mutated (G)MODP population in the BM. To validate this hypothesis, BM rather than blood sampling may be the best approach to discover whether the (G)MODP is mutation carrier in both SS and MS LCH. Unfortunately, at present, BM is not sampled for routine clinical evaluation of LCH,34 unless there is evidence for cytopenia, a complication displayed only in MS-RO+ disease.
We here identified for the first time the BRAF mutation in small CD68+ myeloid cells that may be LCH cells and/or MΦs and in OC-like MGC cells present in the LCH lesions of low-risk patients. LCH lesion–associated MGC display hallmarks of OC and are suspected to cause the osteolysis that characterizes bone LCH.1,35 These MGC might be derived from Mo’s or DCs38 or the (G)MODP.27 Halbritter et al8 have identified an OC transcriptome signature within CD1a+CD207+ LCH cells from both low- and high-risk patients. Taken together, these new findings support the concept that MGC in LCH lesions are OC-like cells that can originate from driver mutation–carrying progenitor cells, either directly or via Mo’s/MΦs. Furthermore, in 2 low-risk LCH patients, we found the driver mutation in lymphoid cells isolated from biopsied lesions. In patient LCH_028 with isolated disease of the pituitary stalk manifesting as diabetes insipidus,34 we found mutant BRAF alleles in natural killer cells isolated from blood (supplemental Figure 8C). These data indicate that the cell of origin in LCH may in certain low-risk patients lie at or upstream of the level of the LMPP.
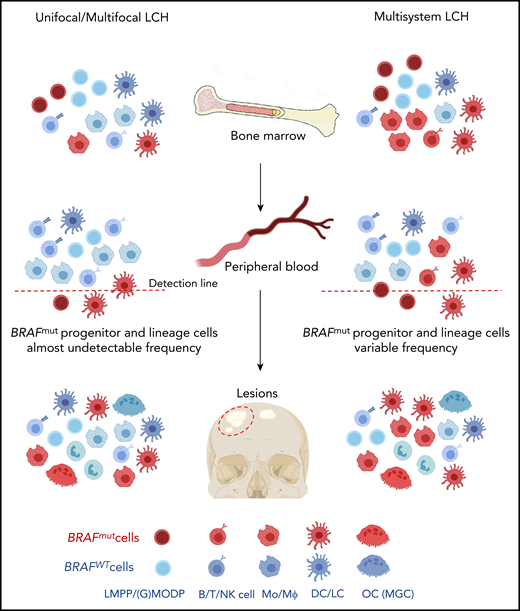
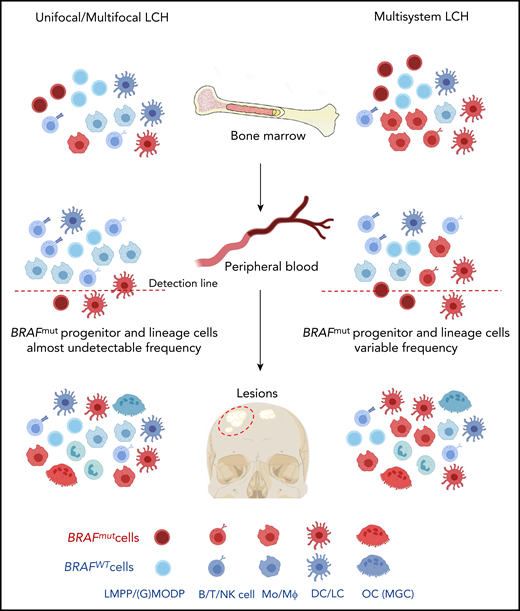
Contrary to the misguided myeloid DC model, we propose that both in high- and low-risk LCH patients, oligopotent progenitor cells are the drivers of LCH pathology (Figure 5). The somatic mutation may occur at the level of the (G)MODP or more upstream at the level of the LMPP, but the result is that myeloid cells differentiating from the progenitor may carry the mutation. Possibly, these progenitors are mobilized from the BM into the blood in response to elevated serum levels of cytokines such as GM-CSF, M-CSF, and Flt3L. Serum levels of these cytokines are reportedly increased in LCH patients.50,51 Such progenitors may be recruited into inflamed tissue sites and thereupon encounter myeloid cell favorite growth factors such as M-CSF, RANKL, TNF, GM-CSF, and TGF-β, which occur in LCH-affected tissues.35,52-56 The features associated with constitutive MAPK pathway activation may then lead to progenitor cell proliferation, increased survival, and in situ differentiation into DCs, LCs, MΦs, and OCs,57,58 as supported by Halbritter et al.8
Hypothesis on the cell of origin in high- and low-risk LCH. Based on the results described in this paper, both low-risk (left part) and high-risk (right part) LCH are caused by somatically mutated oligopotent progenitor cells originating in the BM, such as the LMPP or (G)MODP. Progeny formed in the BM and/or at the tissue site that these progenitor cells are recruited to after mobilization into the blood determines the composition of myeloid cell types in the lesion. As shown here and supported by Halbritter et al,8 both high-risk and low-risk LCH lesions may contain the same diversity of mutation-carrying differentiated myeloid cell types, including OCs and MΦs. The different disease manifestations between high- and low-risk LCH may be due to significant differences in the frequencies of mutated progenitor cells and their place in the hematopoietic tree and may be reflected in the frequency and type of mutated end-differentiated cell types present.
Hypothesis on the cell of origin in high- and low-risk LCH. Based on the results described in this paper, both low-risk (left part) and high-risk (right part) LCH are caused by somatically mutated oligopotent progenitor cells originating in the BM, such as the LMPP or (G)MODP. Progeny formed in the BM and/or at the tissue site that these progenitor cells are recruited to after mobilization into the blood determines the composition of myeloid cell types in the lesion. As shown here and supported by Halbritter et al,8 both high-risk and low-risk LCH lesions may contain the same diversity of mutation-carrying differentiated myeloid cell types, including OCs and MΦs. The different disease manifestations between high- and low-risk LCH may be due to significant differences in the frequencies of mutated progenitor cells and their place in the hematopoietic tree and may be reflected in the frequency and type of mutated end-differentiated cell types present.
The presence of mutated progenitor cells in lesions of SS LCH patients8 and mutated myeloid cells in both blood and lesions of low-risk LCH patients (our data) suggests that the mechanism underlying low-risk and high-risk LCH pathogenesis may be the same (Figure 5). The LCH driver mutation can incur at, or upstream of, the level of the LMPP, but its detection in lymphoid cells is more rare than detection in myeloid cells. Whether the dominant myeloid signature of LCH lesions reflects a differential impact of the mutation on myeloid rather than lymphoid development, or a preferred occurrence at the level of the (G)MODP remains to be addressed. The difference in disease manifestation would lie in the lower frequency of mutation-carrying cells in the circulation of low-risk LCH patients. This hypothesis needs to be validated by determining the presence of BRAF mutation–carrying progenitor cells, facilitated by in vitro DC/LC culture as demonstrated here, in a large number of high- and low-risk LCH patients.
In >10% of patients presenting with a single LCH lesion, the disease progresses to multifocal disease, or it relapses after topical treatment–induced remission.59,60 These observations are in agreement with the idea that the cell of origin is not eliminated by local treatment. We suggest that in these cases, the cell of origin is a BM-derived progenitor that needs to be eliminated by systemic treatment to cure the disease. Sensitive techniques for tracing of pathogenic progenitor cells, ideally in BM samples of patients with low-risk LCH, may support therapy decisions, particularly for UF bone or skin-limited LCH patients who develop new LCH lesions.
For original data, please contact one of the corresponding authors.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank the patients and their parents for permission to use their biological samples and clinical data for this study. The authors also thank R. L. P. Vlierberghe for assisting in the setup of laser-guided microdissection, A. Schmitz, E. Karelioti (GenomeScan, Leiden), and D. van Egmond (Department of Pathology, Leiden University Medical Center) for their help in ddPCR experiments. They thank F. van Diepen, M. van Baalen, E. Mul, and other personnel of the flow cytometry core facilities at the Netherlands Cancer Institute, Sanquin Blood Supply and Leiden University Medical Center for support in cell sorting. C. J. M. van Noesel, (Amsterdam University Medical Center), M. Scheijde-Vermeulen (Princess Máxima Center for Pediatric Oncology/Utrecht University Medical Center), P. Saeed, and J. A. M. Bramer (Princess Máxima Center for Pediatric Oncology, Utrecht/Amsterdam University Medical Center) are acknowledged for providing FFPE blocks, LCH lesion–derived DNA, or fresh tissue samples. They also thank A. C. Lankester (Willem Alexander Children’s Hospital/Leiden University Medical Center) for suppling biobanked samples from healthy hematopoietic stem cell donors, and A. von Deimling (Institut für Pathologie Heidelberg) and L. Schmitt (UPMC Children’s Hospital of Pathology) for performing automated VE1 immunostaining. BioRender.com was used to create the visual abstract and Figure 5.
This work was supported by Stichting Kinderen Kankervrij (grant 285) (A.G.S.v.H., Y.X., C.v.d.B., and Jannie Borst), the Landsteiner Foundation for Blood Research (grant 1355) (Y.X. and Jannie Borst), Stichting Histiocytose Nederland (A.G.S.v.H.), and Stichting 1000 Kaarsjes voor Juultje (A.G.S.v.H.).
Authorship
Contribution: Y.X. and A.G.S.v.H. designed the research, performed experiments, interpreted/analyzed data, prepared figures, and wrote the manuscript; Jannie Borst designed research, interpreted data, and wrote the manuscript. E.S., Jelske Borst, X.L., T.d.W., J.G., R.V., A.S., P.K., and J.P. performed experiments, analyzed data, and helped prepare figures; and C.v.d.B. organized informed consent, contributed patient material, and provided clinical data.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Astrid G. S. van Halteren, Leiden University Medical Center, PO Box 9600, 2300 RC Leiden, The Netherlands; e-mail: a.g.s.van_Halteren@lumc.nl; Yanling Xiao, Leiden University Medical Center, PO Box 9600, 2300 RC Leiden, The Netherlands; e-mail: y.xiao@lumc.nl; and Jannie Borst, Leiden University Medical Center, PO Box 9600, 2300 RC Leiden, The Netherlands; e-mail: j.g.borst@lumc.nl.
REFERENCES
Author notes
Y.X. and A.G.S.v.H. contributed equally to this study.
X.L. and Jelske Borst contributed equally to this study.

![GMODP frequency in blood reports response to therapy in 1 case study. (A) The frequencies (%) of (G)MODP cells among total live cells in PBMC samples of 23 LCH patients (supplemental Table 1) collected at first disease onset or relapse (prior to (n = 20) or after the start of systemic therapy (n = 3)) and age-matched HDs (n = 36), as determined by flow cytometry. Error bars indicate median and interquartile range. Indicated P value was determined by Mann-Whitney U test (**P < .01). The gray area indicates the calculated progenitor frequency range based on HD data. (B) Frequencies (%) of (G)MODP cells among live cells in the blood of MS-RO+ patient LCH_002 at diagnosis (before therapy [pre Tx]), 1 month after starting chemotherapy (on Tx), and 3 years after finishing salvage chemotherapy (post Tx) and an age-matched HD. (C-H) (G)MODP cells sorted from blood samples of LCH_002 taken before, during, or after therapy and an age-matched HD were seeded at 1000 cells per well and cultured under LC-differentiation conditions. (C) Light microscopic appearance of the cultures. (D) Absolute number of live cells per well at the end of the culture period. (E) Flow cytometry plots depicting the gating strategy for sorting of (G)MODP offspring cells for BRAF mutation analysis. (F) Flow cytometry plots depicting CD207+CD1a+ LC-like cells within HLA-DR+CD11c+CD14+ offspring cells. (G-H) DNA was extracted from the G1, G2, and G3 fractions of the cultured cells, sorted as depicted in panel E and analyzed for the presence of the BRAF mutation by ddPCR. (G) Representative 2-dimensional ddPCR plot depicting BRAFV600E mutation in (G)MODP-derived HLA-DR+CD14-CD11c+ offspring cells from LCH_002. (H) BRAFV600E mutation status as detected by ddPCR of (G)MODP offspring cells of the indicated phenotypes (sorted as indicated in panel E). (I) Flow cytometry plots depicting the gating strategy for sorting of DCs and Mo’s from blood samples of LCH_002. (J) Corresponding BRAFV600E mutation status of DCs and Mo’s identified by the listed markers.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/136/19/10.1182_blood.2020005209/2/m_bloodbld2020005209f2.png?Expires=1769824873&Signature=0CpQYUxBT0KvCKyfJF8fgS326YK7YPMZiC0Y83tmATYrfd8hGu5J7hsR3P8BiF6gYvy-PrDDxVeBlQDhK6TXNSA-JR2iKHYsDm74R94akLT8ALSWKvpYDJ3w16nyRa-Z4yjNd7BMha7RnO9JDPjOzrIszUb7Rmokb17d7w5qoc6VwWtEZbGs5F8pL9MBog~qV2mjygBr8hsY9wzNR~EzQmJzhncgTgYOfmTb3dV4hdLHlNuLaRsp1PajF87MsnL6bcIJBfaR2b9Asiz1e7soot0-x81Smw0Ojs8xlBU6ZXYHQdDDilwvbq-gfUK6iYm90GaMiCQF9j-RnwpnAeBoEA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![GMODP frequency in blood reports response to therapy in 1 case study. (A) The frequencies (%) of (G)MODP cells among total live cells in PBMC samples of 23 LCH patients (supplemental Table 1) collected at first disease onset or relapse (prior to (n = 20) or after the start of systemic therapy (n = 3)) and age-matched HDs (n = 36), as determined by flow cytometry. Error bars indicate median and interquartile range. Indicated P value was determined by Mann-Whitney U test (**P < .01). The gray area indicates the calculated progenitor frequency range based on HD data. (B) Frequencies (%) of (G)MODP cells among live cells in the blood of MS-RO+ patient LCH_002 at diagnosis (before therapy [pre Tx]), 1 month after starting chemotherapy (on Tx), and 3 years after finishing salvage chemotherapy (post Tx) and an age-matched HD. (C-H) (G)MODP cells sorted from blood samples of LCH_002 taken before, during, or after therapy and an age-matched HD were seeded at 1000 cells per well and cultured under LC-differentiation conditions. (C) Light microscopic appearance of the cultures. (D) Absolute number of live cells per well at the end of the culture period. (E) Flow cytometry plots depicting the gating strategy for sorting of (G)MODP offspring cells for BRAF mutation analysis. (F) Flow cytometry plots depicting CD207+CD1a+ LC-like cells within HLA-DR+CD11c+CD14+ offspring cells. (G-H) DNA was extracted from the G1, G2, and G3 fractions of the cultured cells, sorted as depicted in panel E and analyzed for the presence of the BRAF mutation by ddPCR. (G) Representative 2-dimensional ddPCR plot depicting BRAFV600E mutation in (G)MODP-derived HLA-DR+CD14-CD11c+ offspring cells from LCH_002. (H) BRAFV600E mutation status as detected by ddPCR of (G)MODP offspring cells of the indicated phenotypes (sorted as indicated in panel E). (I) Flow cytometry plots depicting the gating strategy for sorting of DCs and Mo’s from blood samples of LCH_002. (J) Corresponding BRAFV600E mutation status of DCs and Mo’s identified by the listed markers.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/136/19/10.1182_blood.2020005209/2/m_bloodbld2020005209f2.png?Expires=1769824874&Signature=R~21Z3GfWpBy1kN2cGzXkdlUbupfwPXPHlnnzrJ-U-mroj1W1lYKEbVyFaytJnd54lUWqqTQsnD8pOuWHUg8KWx2cmcxHpgXGZPWJbUkmy7H0xD4GuPT22gxSO55W3uaFzf56W~Xe1k1rW7zptHXWK4idij4MefT43YGJFnQl555UUofvg-rmSkxSA4fSNP8bQM4uriTRq-yQMqvmQlHgx93u4vWOksN86w~mO6y~n85bEhhJ0-U8ktbF9g1GcrehVN80eVr7Fo6L2TT8EnF4Vu~msjpFyypm8oJ6wnb9tHZ5N4Cgh3sv2s31S5bpeDwFzYPg7pqwtYlhb-1ToIknw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)