Abstract
BACKGROUND
Chimeric antigen receptor (CAR) T cell therapy has revolutionized the treatment of relapsed/refractory B-cell malignancies, as highlighted by high complete remission rates and FDA approval of CD19-specific CAR T cell products. However, depth and duration of remission are limited by antigen loss/downregulation on tumors, as observed in clinical trials using CAR T cells targeting the CD19 or CD22 in leukemia and lymphoma, BCMA in multiple myeloma, and EGFRvIII in glioblastoma. This observation forms the basis of current efforts to develop multi-targeting CAR T cells to prevent antigen-negative escape. Antigen density is an important factor modulating CAR T cell response, since antigen expression below a certain threshold fails to trigger the full range of T cell functions. Given that signal strength induced upon antigen encounter determines CAR T cell activity, we hypothesized that simultaneous targeting of two dimly-expressed antigens will result in enhanced CAR T cell signaling and anti-tumor function, approaching that seen in response to one highly-expressed antigen. This is important given the heterogeneity of antigen expression in various cancers. Therefore, the bi-specific CAR T cells currently being developed to prevent antigen-negative escape could also be used to enhance efficacy against low antigen density (LAD) tumors. Results from this study will provide a novel rationale for using multi-specific CAR T cells and illuminate the mechanisms of successful CAR T cell therapy.
METHODS
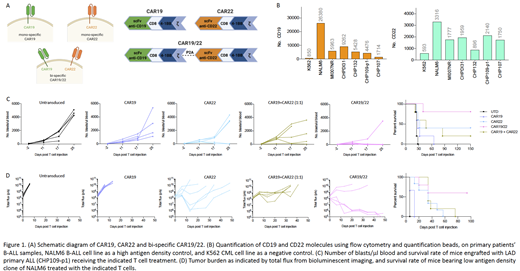
Lentivirus transduction was performed to generate CAR T cells from healthy human T cells, using second generation 4-1BBz CARs specific for either human CD19 or CD22, or both in cis, herein referred to as CAR19, CAR22, or CAR19/22, respectively (Figure 1A). For in vitro functional characterization, we performed co-culture assay of T cells and B cell leukemia cell line NALM6, which is known to express high levels of both CD19 and CD22. To assess T cell function against LAD tumor cells, primary patients' B-ALL samples expressing low antigen density in comparison to the NALM6 cell line were used (Figure 1B). CAR T cell anti-tumor potency was determined by assessing CAR T cell cytotoxicity and cytokine production. For in vivo therapeutic study, primary patients' B-ALL samples with dimly expressed CD19 and CD22 were used to evaluate and compare the therapeutic efficacy of mono- versus bi-specific CAR T cells. Additionally, we generated a LAD tumor model by deleting the highly expressed CD19 and CD22 from the ALL cell line NALM6 using CRISPR/Cas9, transducing the now antigen-negative cell line with CD19 and CD22, followed by single cell cloning to generate a cell line expressing low antigen density for both the CD19 and CD22. We engrafted tumor cells in NSG mice, followed by administration of CAR19, CAR22, CAR19/22 or untransduced T cells. Therapeutic efficacy was assessed by measuring tumor burden using either flow cytometry or bioluminescent imaging.
RESULTS
Cytotoxicity assay revealed that the bi-specific CAR19/22 T cells killed tumor cells more rapidly than CAR19 or CAR22 T cells. Further, compared to mono-specific CAR T cells, the bi-specific CAR19/22 T cells produced significantly more pro-inflammatory cytokines including IL-2 and IFNg, in response to stimulation with LAD primary samples or NALM6 cells. This increased cytokine-producing capacity compared to mono-specific CAR T cells was maintained following repeated antigen stimulation when in vitro exhaustion assay was performed. In vivo, enhanced tumor elimination was observed in mice receiving bi-specific CAR19/22 T cells compared to either of the mono-specific CAR T cells, in both low antigen density primary ALL and NALM6 tumor models. This translated to increased survival rates seen in mice treated with the bi-specific CAR19/22 T cells (Figure 1C-D).
CONCLUSIONS
Here we showed that bi-specific CAR19/22 T cells are superior to mono-specific CAR19 or CAR22 T cells, not only against LAD tumors but also tumor cells expressing high antigen density, NALM6. This was demonstrated by their enhanced cytokine-producing function, cytotoxic capacity, and therapeutic efficacy in vivo. Results from this study provide a novel rationale for repurposing multi-specific CAR T cells as a strategy to improve efficacy against LAD tumors, in addition to the recognized benefit of reducing antigen-negative escape.
Shestova: Hemogenyx Pharmaceuticals LLC: Research Funding. Grupp: Novartis, Roche, GSK, Humanigen, CBMG, Eureka, and Janssen/JnJ: Consultancy; Novartis, Kite, Vertex, and Servier: Research Funding; Novartis, Adaptimmune, TCR2, Cellectis, Juno, Vertex, Allogene and Cabaletta: Other: Study steering committees or scientific advisory boards; Jazz Pharmaceuticals: Consultancy, Other: Steering committee, Research Funding. Ruella: viTToria biotherapeutics: Research Funding; Novartis: Patents & Royalties; BMS, BAYER, GSK: Consultancy; AbClon: Consultancy, Research Funding; Tmunity: Patents & Royalties. Gill: Novartis: Other: licensed intellectual property, Research Funding; Interius Biotherapeutics: Current holder of stock options in a privately-held company, Research Funding; Carisma Therapeutics: Current holder of stock options in a privately-held company, Research Funding.