Abstract
Introduction: Immune checkpoint inhibitors have limited single agent activity in B-cell non-Hodgkin lymphoma (NHL). Hence, it is important to develop strategies which will thwart immune evasion in this disease. Neddylation is a sequential enzyme-based process which ultimately regulates protein turnover. In the initial step, NAE activates NEDD8 in an ATP-dependent reaction in which a high-energy thioester bond is formed between NEDD8 and the catalytic cysteine of NAE. Active NEDD8 is then transferred to the NEDD8-specific E2 conjugating enzyme (UBE2M) and is conjugated to cullin proteins which are part of the Cullin-RING E3 ubiquitin ligases (CRL). Pevonedistat (pevo) forms a covalent adduct with NEDD8, thereby inhibiting NAE and thus reduces CRL activity and diminishes ubiquitination and proteasomal degradation of CRL substrates (IκB, HIF-1α, etc). We have recently reported that neddylation regulates T cell activation and polarization (Best et al, Leukemia 2021). Here we investigate how pharmacologic targeting of neddylation modulates anti-tumor immunity using NHL models.
Methods: Peripheral blood mononuclear cells were isolated from patients with NHL and T cells were purified using Dynabeads. A20 cells were transplanted into flanks of syngeneic BALB/c mice. When tumors reached 100 mm 3, mice were randomized into groups and treated with pevo 60 mg/kg subcutaneously daily for 10 days or vehicle control. Once moribund, mice were sacrificed, tumors were processed into single-cell suspension and analyzed by flow cytometry. Pevo was provided by Takeda Development Center Americas Inc. (Cambridge, MA).
Results: Primary patient-derived CD3/28-stimulated CD3 + T cells exhibited upregulation of TNFα and IFNγ in vitro in the presence of pevo. Concurrently, we observed increased expression of PD-1 and CTLA-4. Pre-treatment of T cells with pevo enhanced killing of NHL cell lines (JeKo-1, Mino, Maver-1 and VAL) in allogeneic cytotoxicity assays. Expectedly, treatment with pevo resulted in increased expression of HIF-1α in TCR-stimulated T cells. shRNA-mediated knockdown of HIF-1α abrogated the pevo effect, suggesting that NAE inhibition modulates T cell function in HIF-1α-dependent manner.
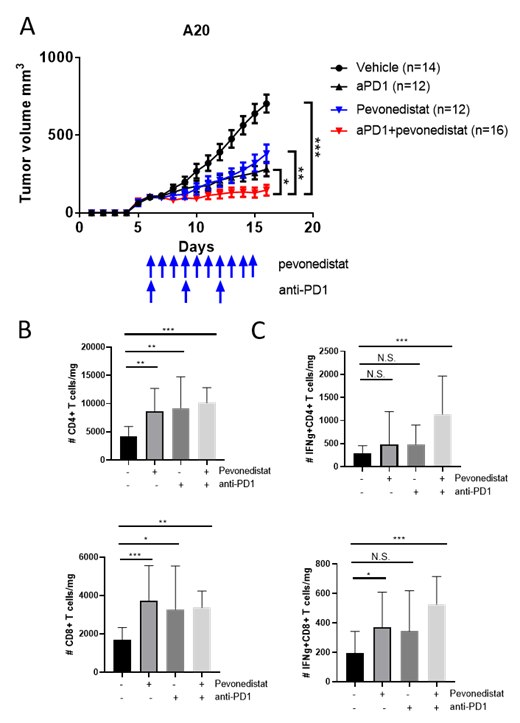
While A20 cells showed resistance in vitro, treatment with pevo delayed lymphoma progression in A20 mice in vivo (Fig 1A). This was accompanied by an increase of tumor-infiltrating lymphocytes (TILs; Fig 1B). CD8 + TILs from pevo-treated mice exhibited activated phenotype as manifested by increased secretion of IFNγ (Fig 1C). Meanwhile, expression of the exhaustion molecules CTLA-4 and PD-1 by CD4/CD8 + TILs remained unchanged. To further investigate the role of T-cell immunity in this setting, we employed 1) CD8 depletion by pre-treatment with 12.5 mg/kg anti-CD8 antibody (IV); or 2) CRISPR/Cas9-mediated knockout of β2-microglobulin (MHC class I protein) in A20 cells. Either approach led to a partial decrease of pevo efficacy in vivo compared with respective controls. To exclude tumor-intrinsic effect of NAE inhibition, we knocked down UBE2M in A20 cells. Loss of UBE2M had no effect on growth of control tumors, or pevo therapeutic effect, implying that the anti-tumor efficacy of NAEi was T cell-mediated in this model.
Since pevo modulates PD-1 on human T cells, we explored its effect on PD-L1 expression. Treatment with pevo upregulated PD-L1 expression in A20 cells in a MYC-dependent manner. Hence, we explored a combination of pevo and αPD-1 blockade in A20 model. Combination treatment significantly increased the CD4 + and CD8 + TILs. A decrease in tumor growth was significantly more pronounced than with either drug alone (Fig 1A). The combination benefit was fully reversed by loss of B2M, again highlighting the importance of immune mechanism . We observed expansion of IL-2, IL-4 and IL-17-secreting CD4+ TILs following the combined treatment, compared with either drug alone. In addition, CD4+ and CD8+ TILs derived from these mice secreted high levels of IFNγ (Fig. 1C).
Conclusions: NAE inhibition enhanced T cell-mediated cytotoxicity in vitro. Treatment with pevo promoted activation of TILs and restricted tumor growth in an A20 mouse lymphoma model. Pevo-treated tumors were sensitized to αPD-1 . Thus, targeting NAE enhances anti-tumor immunity. Our data provide a strong rationale for future studies of pevo in combination with immune checkpoint inhibitors in lymphoma and other tumors.
Siddiqi: Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; BeiGene: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; BMS: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; TG Therapeutics: Research Funding; Pharmacyclics LLC, an AbbVie Company: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Janssen: Speakers Bureau; AstraZeneca: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Membership on an entity's Board of Directors or advisory committees; Kite Pharma: Membership on an entity's Board of Directors or advisory committees, Research Funding; Oncternal: Research Funding. Berger: Takeda Development Center Americas, Inc.: Current Employment. Danilov: Bayer Oncology: Consultancy, Honoraria, Research Funding; SecuraBio: Research Funding; Genentech: Consultancy, Honoraria, Research Funding; TG Therapeutics: Consultancy, Research Funding; Abbvie: Consultancy, Honoraria; Beigene: Consultancy, Honoraria; Pharmacyclics: Consultancy, Honoraria; Gilead Sciences: Research Funding; Takeda Oncology: Research Funding; Astra Zeneca: Consultancy, Honoraria, Research Funding; Bristol-Meyers-Squibb: Honoraria, Research Funding; Rigel Pharm: Honoraria.