Abstract
The CBFA2T3-GLIS2 (CBF/GLIS) fusion is a product of a cryptic translocation exclusively seen in refractory infant AML. Lack of relevant model systems that accurately recapitulate this infant AML has limited progress. To overcome this barrier, we developed an endothelial cell (EC) co-culture system to support malignant transformation, self-renewal, and propagation of leukemia-initiating cells (LIC) in CBF/GLIS-transduced human cord blood hematopoietic stem/progenitor cells (CB HSPCs) ex vivo.
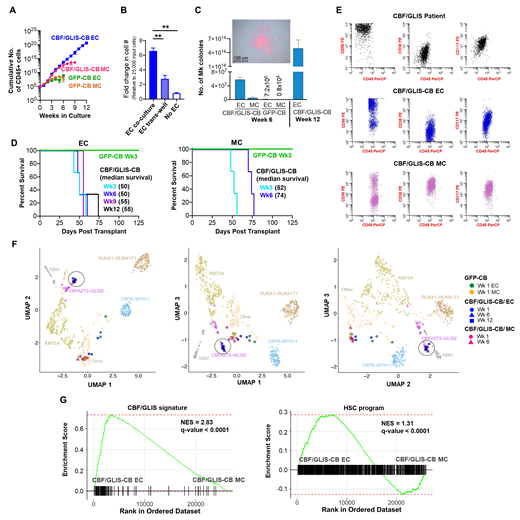
Lack of recurrent cooperating mutations suggests that CBF/GLIS fusion might be sufficient for malignant transformation. To test this, we expressed the CBF/GLIS fusion or GFP control in CB HSPCs (CBF/GLIS-CB or GFP-CB) by lentiviral transduction and placed transduced cells in either EC co-culture or myeloid-promoting culture (MC). CBF/GLIS-CB cells expanded faster with prolonged lifespan in EC co-culture compared to MC (Figure 1A). Proliferation of CBF/GLIS-CB cells declined after transfer to either an EC trans-well culture or in suspension culture (Figure 1B), suggesting that direct contact as well as secreted factors are required for optimal growth of transduced cells.
The CBF/GLIS fusion has been shown to confer enhanced megakaryocytic differentiation. At 6 weeks, CBF/GLIS-CB cells in EC co-culture formed significantly more megakaryocytic colonies than CBF/GLIS-CB cells grown in MC or CBF/GLIS-GFP cells grown in either condition (Figure 1C). At 12 weeks, CBF/GLIS-CB cells cultured in EC co-culture continued to produce numerous megakaryocytic colonies, demonstrating long lived self-renewal and enhance megakaryocytic differentiation of CBF/GLIS-CB cells co-cultured with ECs.
To determine whether the EC niche promotes generation and propagation of LICs, we evaluated the murine engraftment of CBF/GLIS-CB cells expanded on ECs or in MC following 3, 6, 9 and 12 weeks of culture. CBF/GLIS-CB cells cultured in EC co-culture at each time point exhibited robust engraftment that progressed to frank leukemia in vivo (Figure 1D), demonstrating that EC co-culture promotes long-term maintenance of functional LICs. CBF/GLIS-CB cells grown in MC also induced leukemia from 3- and 6-week cultures but then became senescent at 9 and 12 weeks, suggesting limited preservation of the LICs.
Flow cytometric analysis of CBF/GLIS-CB cells identified a malignant population that is of the RAM immunophenotype (CD56 hi, CD45 dim, and CD38 dim/-) previously reported in infants with CBF/GLIS AML in both culturing conditions. However, CBF/GLIS-CB cells in EC co-culture constituted an almost homogeneous population that expressed the RAM immunophenotype, whereas only a subset was detected in MC at week 6 (Figure 1E).
To determine the fidelity of transformation to primary leukemia, we performed RNA-sequencing of CBF/GLIS-CB cells cultured with ECs or in MC. Unsurpervised clustering analysis demonstrated that the CBF/GLIS-CB cells from weeks 6 and 12 in EC co-culture clustered with primary CBF/GLIS-positive patient samples, but not CBF/GLIS-CB cells cultured in MC nor GFP controls (Figure 1F). Further transcriptome analysis revealed CBF/GLIS and HSC signature genes, previously identified to be associated with CBF/GLIS AML, were both significantly enriched in CBF/GLIS-CB cells grown in EC culture relative to MC (Figure 1G). These results suggested that the signaling pathways that are aberrantly dysregulated in primary CBF/GLIS leukemia are faithfully recapitulated in CBF/GLIS-CB cells co-cultured with ECs.
Despite concerted efforts, previous attempts to model CBF/GLIS AML in murine hematopoietic cells have failed to generate overt leukemia. In this study, we demonstrate that in an EC co-culture system, the CBF/GLIS oncogenic fusion is sufficient to transform human CB HSPCs that faithfully recapitulates the morphology, transcriptome and immunophenotype of CBF/GLIS AML as well as highly aggressive leukemia in xenograft models. Furthermore, the EC co-culture system provides a tractable model system to further interrogate the mechanisms of leukemogenesis and identify biomarkers for disease diagnosis and targets for therapy in CBF/GLIS AML.
Hylkema: Quest Diagnostics Inc: Current equity holder in publicly-traded company; Moderna: Current equity holder in publicly-traded company. Pardo: Hematologics, Inc.: Current Employment. Eidenschink Brodersen: Hematologics, Inc.: Current Employment, Other: equity ownership. Loken: Hematologics, Inc.: Current Employment, Other: current equity holder in a privately owned company.