Abstract
Recently, significant progress has been made in identifying novel therapies, beyond conventional immunochemotherapy strategies, with efficacy in B-cell lymphomas. One such approach involves targeting the CD19 antigen on B cells with autologous-derived chimeric antigen receptor (CAR) cells. This strategy is highly effective in patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), as evidenced by recent regulatory approvals. Recent reports suggest that this is an effective strategy for high-grade B-cell lymphoma. The biological underpinnings of these entities and how they overlap with each other and DLBCL continue to be areas of intense investigation. Therefore, as more experience with CAR T-cell approaches is examined, it is interesting to consider how both tumor cell–specific and microenvironmental factors that define these highly aggressive subsets influence susceptibility to this approach.
Introduction
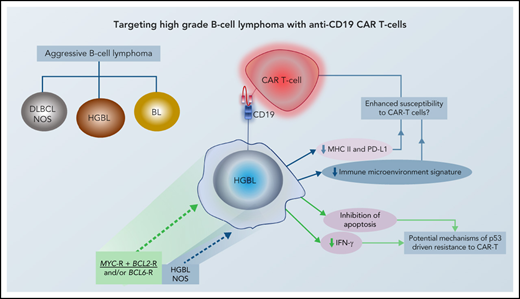
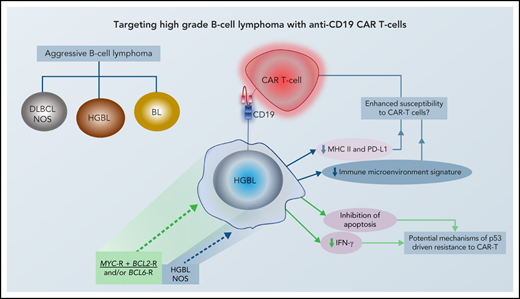
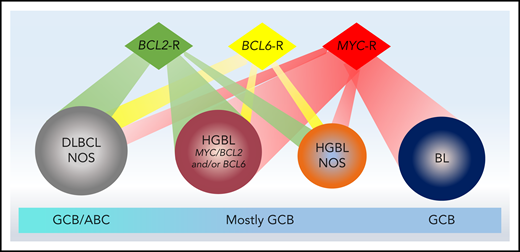
Aggressive B-cell lymphoma is a clinically and biologically heterogenous entity. While most cases are categorized as diffuse large B-cell lymphoma (DLBCL) not otherwise specified (NOS), a subset with high-grade morphology harbors distinct oncogenic mechanisms and displays inferior survival following R-CHOP. In the current World Health Organization (WHO) classification, these are categorized as high-grade B-cell lymphoma (HGBL) with MYC and BCL2 and/or BCL6 rearrangements (HGBL: double hit [DH] or triple hit [TH]) or HGBL-NOS. The separate categorization of HGBL from both DLBCL and Burkitt lymphoma (BL), based on distinct biology and clinical behavior, is helpful in setting the stage for investigation of novel approaches for the former category (Figure 1). Attempts to improve outcome of HGBL have included strategies such as high-dose chemotherapy (HDT) followed by autologous stem cell transplantation (ASCT) as well as allogeneic transplant in younger patients. However, survival for these patients remains suboptimal. Induction failure, early systemic relapse, and central nervous system (CNS) progression events occur with relatively high frequency in HGBL following standard approaches, and developing novel effective strategies for these lymphomas is an unmet need.
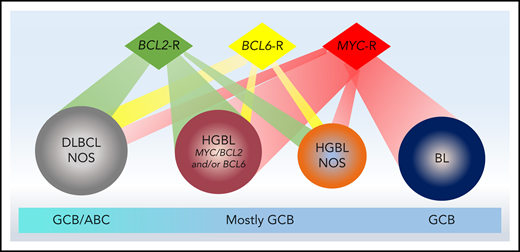
Biological features of aggressive B-cell lymphomas and relationship to cell of origin derivation. Aggressive B-cell lymphomas comprise a spectrum of diseases ranging from DLBCL-NOS to HGBL to BL. While all cases of BL harbor a MYC rearrangement and all cases of HGBL-DH/THL harbor MYC and BCL2 and/or BCL6 rearrangements, other cases with high-grade morphology harbor varying combinations of rearrangements. BL is derived from a germinal center B-cell (GCB) origin and most HGBL cases are also of GCB derivation; DLBCL-NOS may be of GCB or ABC origin.
Biological features of aggressive B-cell lymphomas and relationship to cell of origin derivation. Aggressive B-cell lymphomas comprise a spectrum of diseases ranging from DLBCL-NOS to HGBL to BL. While all cases of BL harbor a MYC rearrangement and all cases of HGBL-DH/THL harbor MYC and BCL2 and/or BCL6 rearrangements, other cases with high-grade morphology harbor varying combinations of rearrangements. BL is derived from a germinal center B-cell (GCB) origin and most HGBL cases are also of GCB derivation; DLBCL-NOS may be of GCB or ABC origin.
CD19-directed chimeric antigen receptor (CAR) T-cell therapy represents a promising modality for lymphoid malignancies, including B-cell lymphomas with aggressive biology. Herein, we describe how researchers have sought to characterize “high-grade” lymphoma and summarize the data that are available on the efficacy of CAR T-cell therapy in this subgroup of lymphomas. We also explore how certain molecular underpinnings of HGBL may be relevant to the application of CAR T-cell therapy in these patients.
Impact of biology on the prognosis of aggressive B-cell lymphoma
While clinical characteristics such as those incorporated in the International Prognostic Index (IPI) reliably predict outcome for patients with DLBCL, the prognostic role of biological factors is less well established and somewhat controversial. More than 20 years ago, Alizadeh and colleagues from the Lymphoma and Leukemia Molecular Profiling Project applied gene expression profiling to newly diagnosed cases of DLBCL and identified at least 2 molecularly distinct subtypes based on their cell of origin and gene expression patterns.1 Similar findings have been published by other groups, and recently, several studies have defined new and more refined molecular classifications of DLBCL that categorize groups based on mutational profiles within cell of origin defined subgroups.2-4
Historically, aggressive lymphoma cases harboring a MYC rearrangement (MYC-R) with BCL2 or BCL6 rearrangements have had adverse outcomes following standard therapy.5-7 Most studies demonstrating this have been retrospective and have frequently evaluated different chemotherapy platforms, confounding interpretation of some studies’ findings. One challenge has been reliably identifying which MYC-associated aberrations contribute most to adverse prognosis. Some studies have shown that cases harboring a single MYC-R do not fare worse; others suggest that the partner to which MYC has translocated has significant prognostic impact (immunoglobulin vs a non-immunoglobulin gene).8,9 The clinical and survival impact of numerical aberrations of MYC (such as extra MYC copies) on outcome is not well understood, with conflicting studies.10-14 Many other molecular markers have been studied but with contradictory reports, underscoring the complexity of the genetic pathogenesis of DLBCL and the importance of developing more robust predictive molecular tools.15-22
Molecular studies in aggressive B-cell lymphoma have also shed light on the overlapping biology between DLBCL and BL and provide insights into the characteristics of lymphomas on the interface of both parent entities. Early studies that set out to explore this question identified that some patients with a histological diagnosis of DLBCL had the typical gene expression profile (GEP) of BL, including higher level of expression of c-myc target genes with low level of expression of major histocompatibility complex (MHC) class I genes and nuclear factor-kB target genes.23,24 These cases benefited from more intensive chemotherapy regimens compared with relatively low-dose regimens such as R-CHOP. More recently, GEP has been used to identify patients with high-risk DLBCL applying a BL-like signature and a signature that is differentially expressed between DH/TH lymphoma and non-DH germinal center B-cell DLBCLs.25,26 Interestingly, these signatures were able to identify not only DH but also many non-DH lymphomas, indicating that other genetic and even epigenetic modifications can produce similar gene expression changes and possibly alter the outcomes of these cases. Moreover, common themes can be observed between the lymphoma groups identified in these studies, such as the low expression of MHC class I/II in the tumor, and the low expression of immune and inflammation signatures in the stroma, such as tumor-infiltrating CD4 T cells and tumor-associated macrophage. The cases that have been identified by these signatures were also enriched in mutations such as MYC, BCL2, EZH2, KMT2D, and TP53.
Mutational analysis has also been implemented to identify high-risk DLBCLs. Recent studies have uncovered genetic subtypes of DLBCL with distinct genotypic, epigenetic, and clinical features as well as disparate responses to chemotherapy.2,4,3 In 2 of these studies, lymphoma cases with cooccurrence of MYD88 L265P and CD79B mutations had less favorable outcomes.
In addition to tumor cell intrinsic alterations, tumor–host interactions have also been shown to be prognostic in aggressive B-cell lymphomas.27 Stromal signatures that reflect the nonmalignant cells and tumor microenvironment, such as markers of endothelial cells and regulators of angiogenesis, are prognostically unfavorable.28 Genetic deletion of MHC class I and II genes is also predictive of poor survival, likely due to the consequent loss of immunosurveillance,20,29 underscoring the significance of interaction between cell intrinsic alterations and host immune response. Similarly, PD-L1 and PD-1 are overexpressed in the tumor microenvironment (but not on tumor cells) with low tumor-associated macrophage content in MYC-R lymphomas.30,31 Furthermore, depleted lymphoma microenvironment with low levels of regulatory T cells (Tregs) and myeloid-derived suppressor cells has less favorable outcomes.32
These studies have provided significant and important insights into the biology of aggressive B-cell lymphoma; however, the translation of these molecular advances to identify high-risk patient subsets that may benefit from alternative treatment strategies has yet to be determined.
High-grade lymphoma in the WHO classification
In an attempt to reflect the impact of molecular features on the prognosis and outcomes of DLBCL, the WHO classification introduced a new category in 2016: high-grade B-cell lymphoma (HGBL) to describe a subset of very aggressive tumors in which the distinction between DLBCL and BL may be challenging.33 The new entity replaced another category that had been described in the 2008 WHO classification as B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and BL unclassifiable, which emphasized morphology rather than molecular features. The new entity (HGBL) included cases with MYC and BCL2 and/or BCL6 rearrangements (DH/TH lymphomas) as well as cases that appear blastoid or intermediate between DLBCL and BL but lack an MYC and BCL2 and/or BCL6 rearrangement. The latter was named HGBL-NOS. Various molecular abnormalities, other than MYC and BCL2 and/or BCL6 rearrangements, have been identified in HGBL-NOS, including single-hit MYC-R, MYC extra copies, MYC-R with BCL2 amplification, MYC amplification with BCL2-IGH rearrangement, and KMT2D/TP53 mutations.34-37
Recent clinical trials testing new therapies in lymphoma, including CAR T-cell trials, have applied the WHO classification when enrolling patients and analyzing outcome data. Although HGBL with MYC and BCL2 rearrangement can be reliably identified based on harboring the aforementioned mutations, HGBL-NOS has “difficult to reproduce” morphologic criteria, and one finds varying approaches to the diagnosis of these lymphomas in different centers. Indeed, many of these cases, when centrally reviewed, are reclassified as DLBCL or BL. The histologic and biologic overlap of these high-grade entities, including BL, presents interpretative challenges when evaluating the mechanisms and efficacy of various novel therapies, including CAR T cells.38 It is therefore critical to include these entities in trials of novel strategies, keeping in mind that their diagnostic definitions continue to evolve as we gain deeper insights into distinct oncogenic mechanisms that distinguish them from one other.39
Approach to HGBL: a therapeutic challenge
Considering that HGBL is a recently described entity with a paucity of outcome data, controversies abound as to its optimal management.39,40 Should patients receive standard DLBCL regimens such as R-CHOP or do their higher proliferation and adverse biological features warrant dose-intensification strategies? Considering their poor prognosis compared with DLBCL or BL, are these patients a subset who can benefit from consolidation with HDT and ASCT? Although this approach is appealing, the benefit has not been demonstrated, albeit due to limited experiences because of their rarity.41,42 Should HGBL-NOS cases be approached differently than HGBL-DH/TH? What about patients with single-hit MYC-R or MYC amplifications? Can relapsed/refractory HGBL be salvaged with HDT/ASCT, as has been a standard for DLBCL, or are CAR T-cell approaches more likely to be curative? Finally, what is the role of CNS-directed chemotherapy prophylaxis in HGBL, and should its implementation be uniform across these HGBL entities or individualized based on CNS-IPI and tumor mutational profile (eg, DH/TH vs SH-MYC)?
The heterogeneity of HGBL cases, the poorly reproducible morphologic criteria for HGB-NOS, the lack of prospective data, and the uncertainty of data extrapolation represent barriers to reaching consensus-based standards in HGBL. Additionally, HGBL cases frequently have distinct patterns of presentation (compared with DLBCL cases) such as high propensity for extranodal and CNS involvement and very high tumor proliferation rates. These unique features in themselves may help explain reduced chemosensitivity and inferior outcomes. Therapeutic approaches to DLBCL and BL are currently distinct, and it remains controversial if HGBL cases should be treated with a BL or DLBCL approach.43,44 As these questions are posed and answered in trials, the role of alternative tools such as CAR T-cell therapy in HGBL and BL should be prospectively investigated in parallel.
CAR T-cell therapy in relapsed and refractory HGBL
Since the initial reports that demonstrated potency of CD19-directed CAR T cells in chronic lymphocytic leukemia and acute lymphoblastic leukemia a decade ago,45,46 many trials have been conducted to test the efficacy and safety of CAR T cells (and natural killer cells) in a variety of hematological malignancies and even solid cancers.47,48 This treatment modality has particularly changed the therapeutic paradigm of multiply relapsed/refractory large B-cell lymphoma with encouraging rates of durable remission in up to 40% of these patients.49-51 The US Food and Drug Administration has approved 5 CAR T-cell products so far in a variety of hematological malignancies.
The pivotal trials that have been conducted in B-cell lymphoma enrolled a percentage of patients with HGBL, ranging between 7% and 20% of the total cohort in each study (Table 1). The WHO classification was used in these trials to define HGBL and involved mostly HGBL-DH/TH and HGBL-NOS. GEP, mutational analysis, or other molecular tools that are described above and have been shown by different research groups to detect aggressive biology and poorer outcomes were not used in CAR T-cell studies. The primary endpoints of these studies were objective response rate (ORR) and event-free survival (EFS), and subset analyses have been performed in most of these studies to evaluate the endpoints across subgroups. Cross-trial comparisons are confounded by different variables such as domains within the CAR (costimulatory domains, hinge and transmembrane region, T-cell activation domain), viral vectors used for gene transfer, culture methods, lymphodepleting regimens, and enrolled patient populations.52 Nevertheless, one can draw some inferences and conclusions. Taking the HGBL data in these trials together, subset analyses in these studies have not demonstrated less favorable ORRs in relapsed/refractory HGBL, with homogeneous and consistent ORRs across demographic and prognostic subgroups, including HGBL (Table 1). Nevertheless, the question remains whether these outcomes will be maintained long-term or when CAR T-cells are used outside clinical trials in the real world.
Recently, the long-term clinical outcomes (median follow-up of 40.3 months) of patients who were treated with tisagenlecleucel for relapsed/refractory B-cell lymphomas in the JULIET trial have been reported.53 The previously reported complete remission (CR) rates of 40% in the total cohort and 25% in HGBL were maintained. The report also supported the durability of responses, including in patients with HGBL. Interestingly, most of the patients who achieved CR at 3 and 6 months remained in remission (78.8% and 86.5%, respectively), suggesting that a short follow-up period is required to ensure long-term outcomes following CAR T-cell therapy. Similarly, long-term outcomes of patients with B-cell lymphoma, including HGBL, treated with axicabtagene ciloleucel in the ZUMA-1 trial were also maintained at 27 months and 3 years.54,55
In parallel and with the concern that the study population in clinical trials may not be representative of patients treated in clinical practice, emerging real-world data have shown similar response rates and durability in both relapsed/refractory B-cell lymphoma and HGBL treated with axicabtagene ciloleucel or tisagenlecleucel56-58 (Table 2).
Three recent randomized trials addressed the question of whether patients with relapsed/refractory large B-cell lymphoma would benefit from CAR T-cell therapy as second-line treatment compared with standard of care (SOC), which is the use of salvage therapy followed by HDT/ASCT59-61 (Table 1). The trials reached seemingly contradictory results, with ZUMA-7 and TRANSFORM showing higher frequency of CR rates and improved EFS in patients treated with axicabtagene ciloleucel and lisocabtagene maraleucel compared with SOC, whereas BELINDA failed to show any significant difference in these outcomes in patients treated with tisagenlecleucel compared with SOC. Although in the BELINDA trial, the percentage of patients with high-grade lymphomas was higher in the tisagenlecleucel group than in the standard care group, the contradictory results of the trials are likely due to other factors, particularly enrollment bias against patients with rapidly progressing or bulky disease in the ZUMA-7 trial and the allowance of bridging chemotherapy in the BELINDA trial.62 Data concerning the outcomes of HGBL cases in both arms were provided in the ZUMA-7 trial with ORR of 81% in the CAR T-cell therapy arm and 42% in the SOC arm (not different from ORR rates in total cohort: 83% and 50%, respectively). An improvement of EFS was also observed in patients with HGBL treated with CAR T-cell therapy compared with SOC, similar to the EFS improvement seen in the total cohort. Response rates and EFS outcomes in the HGBL subgroup of BELINDA and TRANSFORM were not provided. Collectively, these data suggest that HGBL cases that meet ZUMA-7 criteria (not bulky or rapidly progressing and not requiring bridging therapy more than steroids) may benefit from treatment with axicabtagene ciloleucel over ASCT as a second-line therapy, although establishing CAR T-cell therapy as the SOC in second-line setting requires further investigation.
Another area of investigation is the use of CD-19–directed CAR T-cell therapy earlier in the treatment of aggressive lymphomas such as HGBL. ZUMA-12 is a single-arm, phase 2, multicenter study that treated patients with high-risk large B-cell lymphoma, such as DH/TH lymphoma, with axicabtagene ciloleucel if positron emission tomography scan was positive (Deauville score of 4 or 5) after 2 cycles of anti-CD20 monoclonal antibody and anthracycline-containing regimen. The most recent update from the trial reported a high rate of rapid and complete responses in these population (ORR: 90%; CR: 80%)63 (Table 1).
Can the biology of HGBL enable sensitivity or resistance to CAR T-cell approaches?
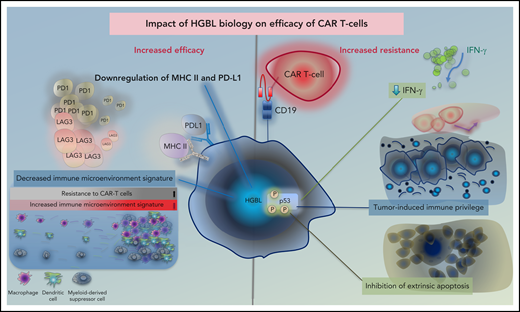
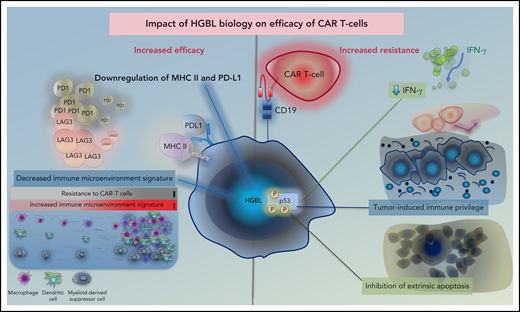
Tumor-intrinsic factors, disease histology, and tumor microenvironment are major factors affecting outcome and durability of tumor response following CAR T-cell therapy.64 In HGBL, many biological alterations that contribute to the aggressiveness of the disease may also have implications for the efficacy of CAR T-cell therapy in these cases (Figure 2). CAR binding to target antigens expressed on the cell surface is independent from the MHC receptor.65 Nevertheless, MHC class II and PD-L1, which are downregulated in HGBL, have been shown to suppress T cells via exhaustion markers such as LAG-3 and PD-1.66 In fact, tumor expression of PD-L1 and MHC II is associated with the lack of durable response after CAR T-cell therapy.67 Furthermore, the signatures of tumor-associated macrophages, which are present at lower numbers in HGBL, are enriched in patients who relapse early after CAR T-cell therapy due to lower peak of CAR T-cell expansion.67,68 Similarly, the above-described depleted microenvironment with low levels of regulatory T cells and myeloid-derived suppressor cells in lymphomas with less favorable outcomes may provide the immune-privileged site for CAR T cells to exert their effects.67,69 These data suggest that the baseline host innate immune dysregulation that contributes to the unfavorable outcomes of HGBL may be advantageous to adaptive immune approaches, such as CAR T cells, due to higher expansion and less exhaustion of these cells.
Impact of HGBL biology on efficacy of CAR T cells. Proposed impact of HGBL biology on efficacy of anti CD19 CAR T cells. HGBL may downregulate MHC-class II and PD-L1 antigens, which suppress CAR T cells via LAG-3 and PD-1, potentially leading to less T-cell exhaustion. Tumors that are resistant to CAR T cells have more activation of the immune microenvironment signature, which may be decreased in HGBL. Aberrations of p53 can increase tumor cell survival through downregulation of interferon signaling, induction of tumor-site immune privilege, and inhibition of extrinsic apoptosis.
Impact of HGBL biology on efficacy of CAR T cells. Proposed impact of HGBL biology on efficacy of anti CD19 CAR T cells. HGBL may downregulate MHC-class II and PD-L1 antigens, which suppress CAR T cells via LAG-3 and PD-1, potentially leading to less T-cell exhaustion. Tumors that are resistant to CAR T cells have more activation of the immune microenvironment signature, which may be decreased in HGBL. Aberrations of p53 can increase tumor cell survival through downregulation of interferon signaling, induction of tumor-site immune privilege, and inhibition of extrinsic apoptosis.
On the contrary, TP53 alterations (mutations and/or copy number alterations) have been shown recently to affect cellular processes that are relevant to CAR T-cell cytotoxicity, including downregulation of interferon and induction of an immune-privileged tumor microenvironment with reduced CD8+ T-cell infiltration, resulting in inferior CR and OS rates in relapsed/refractory DLBCL treated with CAR T-cell therapy.70 The reduced efficacy of CAR T cells could also be due to impairment of the extrinsic apoptosis pathway in TP53-altered lymphomas.70 Although CAR T cells exert their cytotoxicity via granzyme and perforin secretion, death receptors are also used. In vitro and in vivo, impaired cell death receptor pathways in tumor cells render the tumor cells more resistant to CAR T-cell cytotoxicity and the CAR T-cells are more prone to exhaustion due to prolonged CAR stimulation.71,72
Other mechanisms of resistance to CAR T-cell therapy have been described in the literature, such as genetic loss of antigen due to selective pressure,73 antigen masking,74 or trogocytosis.75 CAR T-cell exhaustion and impaired fitness of T cells before therapy (eg, in chronic lymphocytic leukemia) have also been reported as determinants of response or resistance to CD19 CAR T cells.76 Other predictors of response or resistance, such as the collection of sufficient T cells, cellular components in the product, construct design, and the degree of CAR T-cell expansion, have also been described.77 Whether these mechanisms or factors are relevant to the biology or efficacy of CAR T cells in HGBL is unknown.
It is likely that these molecular findings will be integrated into future prognostic and predictive therapeutic models. HGBL is sensitive to many novel therapeutic modalities, and in the context of modulating resistance to CAR T-cell approaches, an important question centers on what are the most promising rational CAR T combinations to focus on in HGBL. Combinatorial approaches with immune checkpoint inhibitors, for example, are intriguing biologically in that they have the potential to impact T-cell function as well as affect tumor cells and the microenvironment.78 Pathways that overexpress MYC and BCL2 are likely important targets in HGBL, suggesting that small molecule inhibitors of these proteins may be synergistic in CAR T-cell combinations. The role of alternative CAR T-cell constructs, including multiantigen-specific CAR T cells in HGBL, is unknown, but as more data with these novel constructs become available, it will be interesting to identify if antigen targeting beyond CD19 can overcome B-cell tumor antigen escape. The insightful understanding of the impact of biology on the efficacy of CAR T cells will allow us to not only select the patients who are more likely to respond to this modality but also shed light on optimal approaches to overcome therapeutic resistance.
Future role of CAR-T cell approaches in HGBL: where do we go from here?
Given the rarity of HGBL and the limited experience with anti-CD19 CAR T cells in aggressive lymphoma thus far, the role and indications for this approach in the management of patients with HGBL have yet to be better defined. Are there subsets of patients with HGBL who may preferentially benefit from this strategy? As we accumulate more clinical data, it should be possible to better evaluate the association of clinical and biological characteristics with outcome and design future studies using this knowledge. The experience from anti-CD19 CAR T-cell studies in acute lymphoblastic leukemia as well as BL (diseases that share many biological characteristics with HGBL) may help inform future investigative strategies. In ALL studies, the emergence of CD19− relapses has been particularly problematic, and efforts are underway to understand and mitigate such immune escape phenomena. These lessons are likely relevant to therapeutic development of cellular therapies in HGBL. The early ZUMA-12 data are quite provocative in suggesting that subsets of newly diagnosed patients with aggressive lymphoma (based on interim response criteria) may preferentially benefit from an early switch to CAR T cells vs continuation of immunochemotherapy. As these and other data mature, randomized studies should focus on the role of CAR T cells in up-front therapeutic platforms. Other key areas in which to explore CAR T-cell indications are BL and HIV-associated high-grade B-cell lymphomas. Relapsed/refractory BL is rare and therefore difficult to study but presents an unmet need in which CAR T-cell approaches represent a rational therapeutic strategy. Thus far, clinical data using CAR T cells in BL are limited, but in pediatric settings, good efficacy has been documented.79-81 The biology of HGBL in the setting of HIV is poorly understood and is an area that requires deeper investigation of cellular immunotherapy strategies looking at response to and impact on host T-cell function and dynamics. There are many challenges and limitations of current CAR T-cell constructs, and next-generation CAR T-cell approaches aim to reduce CAR T-cell toxicities and improve therapeutic efficacy via enhanced bioengineering strategies.82 It is beyond the scope of this review to discuss the many other novel classes of therapy under investigation in aggressive B-cell lymphomas, but it is key in CAR T-cell development to investigate optimal combinatorial strategies incorporating agents that synergize with CAR T cells in select biological subtypes of lymphoma.
Conclusion
HGBLs that do not fit into the categories of DLBCL or BL are associated with poor outcomes following standard therapy, and augmenting curability for patients represents an area of unmet need. Some biological underpinnings of these lymphomas that likely drive treatment resistance have been identified, but many have yet to be elucidated. Early experience with anti-CD19 CAR T-cell approaches demonstrates particularly high activity in HGBL with outcomes indistinct from other large B-cell lymphomas. Given the favorable efficacy thus far, efforts should focus on investigating CAR T-cell and other cellular therapies in prospective studies that are selective for inclusion of HGBL and BL histologies. The unique biology of these intriguing lymphomas should be studied in the context of response to CAR T cells to identify subsets of patients within HGBL that may preferentially benefit from these approaches. Additionally, the impact of CAR T-cell persistence on outcome and role of combinatorial strategies that include CAR T cells should be looked at specifically in HGBL subsets to establish predictors of CAR T-cell failure or success.
Acknowledgment
The authors thank Dr. John DiPersio (Washington University in St. Louis) for his review of the manuscript and insightful suggestions and comments.
Authorship
Contribution: A.A., A.G., and K.D. wrote and approved the final manuscript.
Conflict-of-interest disclosure: A.G.: consulting or advisory role: AstraZeneca, Bristol Myers Squibb, Celgene, Hoffmann-La Roche, Janssen, Kite, Morphosys, Allopex, Gilead, Novartis, Vincerx, and Resilience; research funding (institutional): Acerta, AstraZeneca, Bristol Myers Squibb, Celgene, Genentech, Hoffmann-La Roche, Infinity, Janssen, Karyopharm, Kite, Morphosys, Pharmacyclics, Seattle Genetics, and Verastem. K.D.: consulting or advisory role: AstraZeneca, Beigene, AbbVie, Daiichi Sankyo, ADC Therapeutics, Incyte, Morphosys, and Genmab; research funding: Genentech (institutional). The remaining author declares no competing financial interests.
Correspondence: Alaa Ali, Division of Oncology, Lombardi Comprehensive Cancer Center, Georgetown University, 3800 Reservoir Rd NW, Washington, DC 20007; e-mail: alaa.ali@gunet.georgetown.edu.