Abstract
The effects of thrombopoietin (TPO; c-mpl ligand), FLT3/FLK-2 ligand (FL), and interleukin-6 (IL-6) on the survival of murine hematopoietic long-term reconstituting cells (LTRC) were studied by using lineage-negative, Sca-1–positive, c-kit–positive (Lin−Sca-1+c-kit+) marrow cells from 5-fluorouracil–treated mice. We tested the ability of these cytokines to maintain the viability of LTRC by transplanting the cultured cells to lethally irradiated Ly-5 congenic mice together with compromised marrow cells. As a single agent, only TPO could maintain the LTRC. Neither IL-6 nor FL was effective by itself, but they acted synergistically to maintain the LTRC. We examined whether the maintenance of LTRC by these cytokines was due to the survival of stem cells or was the result of active cell divisions and self-renewal. To monitor cell division, we used membrane dye PKH26. Enriched cells were stained with PKH26 on day 0 and incubated in suspension culture with TPO or with IL-6 and FL for 7 days. On day 7, PKH26low and PKH26high cells were prepared by sorting and their in vivo reconstituting abilities were tested by transplantation into lethally irradiated Ly-5 congenic mice together with compromised marrow cells. PKH26high populations cultured with both TPO alone and the combination of IL-6 and FL showed greater reconstitution activity than that of PKH26low populations. These data indicate that TPO alone and the combination of IL-6 and FL can support the survival of stem cells without stimulating their active cell proliferation.
PROLIFERATION OF primitive hematopoietic progenitors is regulated by a number of cytokines. Based primarily on serial observations of the development of murine and human blast cell colonies, a model of cytokine interactions regulating the proliferation kinetics of primitive progenitors has been proposed.1 In this model, the cytokines are classified into three functional groups.1 Interleukin-3 (IL-3),2IL-4,3 and granulocyte-macrophage colony-stimulating factor (GM-CSF)4,5 form the first group of cytokines that individually can support proliferation of multipotential progenitors after they exit from the cell-cycle dormant state (G0period). A group of IL-6,6 IL-11,7,8IL-12,9 granulocyte colony-stimulating factor (G-CSF),10 and leukemia-inhibitory factor (LIF)11,12 forms the second group. A group consisting of steel factor (SF; c-kit ligand)13,14 and FLT3/FLK-2 ligand (FL)15 forms the third group. Both SF and FL are ligands for receptors with tyrosine kinase activity and they interact with the cytokines in the first and second groups.13-15
Cytokines are also capable of supporting the survival of primitive progenitors. In the murine system, IL-1α,16IL-3,17-20 IL-4,18 G-CSF,18SF,19-21 and FL22 were shown to support the survival of progenitors including colony-forming units-spleen (CFU-S). IL-3,17-19 IL-4,18 and SF19,23-25were reported also to support the survival in culture of long-term reconstituting cells (LTRC). In the human system, IL-3,12,26 GM-CSF,12 SF,26 and FL27 support the survival of colony-forming cells. Leary et al12 in our laboratory observed that cell cycle dormant progenitors in the CD34+HLA-DR− human marrow fraction require either IL-3 or GM-CSF for survival in culture. Brandt et al26 reported that IL-3 or SF supports the survival of human colony-forming cells by testing CD34+HLA-DR−c-kit+ bone marrow cells. Using lineage-negative (Lin−) CD34++CD38− light-density fetal liver cells, Muench et al27 demonstrated that FL supports the survival of human colony-forming cells.
Despite their similarity, there appears to be some differences in the effects of SF and FL on primitive hematopoietic progenitors.15,28,29 Earlier, we noted that blast-like cells persist longer in FL-containing cultures than in SF-containing cultures.15 More recently, we reported that a combination of FL and IL-11 supports maintenance of the in vivo reconstituting ability of cultured cells longer than the combination of SF and IL-11.28 Petzer et al29 reported that FL alone stimulated a net increase in human long-term culture-initiating cells (LTC-IC), but SF did not.
Recently, thrombopoietin (TPO) was found to possess positive effects on primitive hematopoietic progenitors.30-32 TPO was originally identified as a lineage-specific regulator of megakaryocytopoiesis and thrombopoiesis.33-44 TPO acts through a specific receptor termed c-mpl, which has been detected in not only megakaryocytes and plateletes, but also in cell populations containing stem cells and progenitors.45-49 As mentioned above, Ku et al30 and Kobayashi et al31 in our laboratory and Sitnicka et al32observed that TPO can synergize with SF and/or IL-3 in support of the formation of murine and human multilineage colonies. TPO30 can trigger the cell division of dormant (G0) multipotential progenitors in a manner similar to that previously reported for the second group of cytokines such as IL-6,6 IL-11,7,8 IL-12,9G-CSF,10 and LIF.11 Injection of TPO accelerated erythroid recovery of myelosuppressed mice50,51and, in combination with G-CSF, neutrophil recovery.52Megakaryocyte, erythroid, and myeloid progenitors were reduced in the TPO- or c-mpl–deficient mice.53,54 Administration of TPO to the TPO-deficient mice significantly increased the number of myeloid, erythroid, and mixed progenitors.54 TPO increased production of human LTC-IC in suspension culture.29Together, these observations indicate that TPO may be a regulator of the proliferation of multipotential progenitors.
In this report, we present studies of the ability of TPO to maintain the survival of murine LTRC. Comparison was made with FL and IL-6. As a single agent, TPO appears to be more potent than FL or IL-6 in the maintenance of stem cell activities. In contrast, synergism between IL-6 and FL supported the survival of LTRC. These results are in agreement with the observations in the gene knock-out mice and indicate that TPO has a physiological role in the maintenance of stem cell activity. TPO may be an important factor for in vitro manipulation of hematopoietic stem cells.
MATERIALS AND METHODS
Cytokines.
Purified recombinant human TPO was prepared by the Cytokine Production Group of Kirin Brewery (Takasaki, Japan).38,39 Recombinant human FL was produced in yeast and purified as described previously.55 Purified recombinant human IL-6 was a gift from M. Naruto (Toray Industries, Kamakura, Japan). Purified recombinant murine SF was obtained from Immunex (Seattle, WA). Purified recombinant murine IL-3 was purchased from R&D Systems (Minneapolis, MN). Purified recombinant human erythropoietin (EPO) was a gift from F.-K. Lin (Amgen, Thousand Oaks, CA). Unless otherwise specified, the concentration of cytokines used were as follows: 100 ng/mL TPO, 100 ng/mL FL, 100 ng/mL IL-6, 100 ng/mL SF, 10 ng/mL IL-3, and 2 U/mL EPO.
Monoclonal antibodies (MoAbs).
Hybridoma D7 (anti-Ly-6A/E [anti-Sca-1] rat IgG2a) was a gift from P. Kincade (Oklahoma Medical Research Foundation, Oklahoma City, OK). MoAb ACK4 (anti-c-kit; rat IgG2a) was provided by S.I. Nishikawa (Kyoto University, Kyoto, Japan). Hybridoma RB6-8C5 (antimouse granulocytes; rat IgG2b) was provided by R.L. Coffman (DNAX, Palo Alto, CA). MoAb TER119 (anti-erythrocytes; rat IgG2b) was a gift from T. Kina (Kyoto University). Hybridomas 14.8 (anti-B220; rat IgG2b), M1/70.15.11.5 (anti-macrophages; rat IgG2b), GK1.5 (anti-CD4; rat IgG2b), and 53-6.72 (anti-CD8; rat IgG2a) were purchased from American Type Culture Collection (Rockville, MD). 53-2.1 (biotin-conjugated anti–Thy-1.2; rat IgG2a), RA3-6B2 (biotin-conjugated anti-CD45R/B220; rat IgG2a), RB6-8C5 (biotin-conjugated anti–Gr-1; rat IgG2b), and M1/70 (biotin-conjugated anti–Mac-1; rat IgG2b) were purchased from Pharmingen (San Diego, CA). A20-1.7 (fluorescein isothiocyanate [FITC]-conjugated anti–Ly-5.1; mouse IgG1) was provided by H. Fleming (Emory University, Atlanta, GA).
Cell preparations.
Cells from 10- to 16-week-old female C57Bl/6 mice (Charles River Laboratories, Raleigh, NC) were used in suspension and clonal cultures, and cells from 10- to 16-week-old C57Bl/6 mice (Jackson Laboratories, Bar Harbor, ME) that are congenic for Ly-5 allotypes were used in transplantation experiments. 5-Fluorouracil (5-FU; Adria Laboratories, Columbus, OH) was administered intravenously through the tail vein at 150 mg/kg body weight, and bone marrow cells were harvested 2 days later. Cells prepared from pooled femurs and tibiae were washed twice and then subjected to density gradient separation by using Nycodenz (Accurate Chemical and Scientific Corp, Westbury, NY). Cells with densities ranging from 1.063 to 1.077 g/mL were collected.56 Cells reacting to a cocktail of lineage-specific rat MoAbs (RB6-8C5, 14.8, M1/70.15.11.5, GK1.5, TER119, and 53-6.72) were removed twice by using immunomagnetic beads (Dynabeads M-450 coupled to sheep antirat IgG; DYNAL, Great Neck, NY). The resulting Lin− cells were treated with normal rat IgG (Jackson ImmunoResearch Laboratories, West Grove, PA) at 20 μg/106 cells to prevent nonspecific binding of MoAbs to Fc receptors and then stained with FITC-conjugated rat MoAb D7 (anti–Sca-157) and biotin-conjugated rat MoAb ACK4 (anti–c-kit58). The cells were washed twice before staining with streptavidin-conjugated R-phycoerythrin (PE) (Jackson ImmunoResearch Laboratories). In the experiments using PKH26, streptavidin-conjugated allophycocyanin (APC; Caltag Laboratories, San Francisco, CA) was used instead of PE, because the emission wave length of PKH26 is similar to that of PE. Both FITC-conjugated rat IgG2a and biotin-labeled rat IgG2a (Caltag Laboratories) were used as isotype controls. Sca-1+c-kit+ cells were collected by sorting on FACS Vantage (Becton Dickinson Immunocytometry Systems, San Jose, CA).
Clonal cell culture.
Methylcellulose cell culture was performed in 35-mm suspension culture dishes (Falcon, Lincoln Park, NJ). One milliliter of culture contained 100 Lin−Sca-1+c-kit+ cells, α-medium (ICN, Irvine, CA), 1.2% 1,500-cp methylcellulose (Shinetsu Chemical, Tokyo, Japan), 30% (vol/vol) fetal calf serum (Intergen, Purchase, NY), 1% deionized fraction V bovine serum albumin (Sigma Chemical, St Louis, MO), 1 × 10−4 mol/L 2-mercaptoethanol (Sigma), and cytokine(s). Dishes were incubated at 37°C in a humidified atmosphere flashed with 5% CO2. Colony types were determined on day 14 by in situ observation on an inverted microscope according to the criteria described previously.59 Megakaryocyte colonies were scored when the colony contained 4 or more megakaryocytes. Abbreviations for colony types are as follows: GM, granulocyte/macrophage colonies; GEM, granulocyte/erythrocyte/macrophage colonies; GMM, granulocyte/macrophage/megakaryocyte colonies; GEMM, granulocyte/erythrocyte/macrophage/megakaryocyte colonies59; and Meg, megakaryocyte colonies.
Survival of progenitors and reconstituting cells in suspension culture.
One thousand Lin−Sca-1+c-kit+cells were incubated in each well of 24-well plate (Falcon) in suspension culture. The culture medium consisted of α-medium, 20% (vol/vol) fetal calf serum, 1% deionized bovine serum albumin, 1 × 10−4 mol/L 2-mercaptoethanol, and cytokine(s). On day 7 of incubation, aliquots of cells were analyzed for progenitors in clonal cell culture and for the in vivo reconstituting cells. Clonal cell culture was performed as described above. Colonies were scored on day 8 of incubation by in situ observation of the plates on an inverted microscope.59
In vivo reconstitution experiments.
Ten- to 12-week-old female C57Bl/6-Ly-5.2 mice were administered with a single 850-cGy dose of total body irradiation via a 4 × 106 V linear accelerator. After irradiation, 200 freshly sorted Lin−Sca-1+c-kit+ cells of male C57Bl/6-Ly-5.1 mice were injected into the tail vein of the recipients together with 4 × 105 compromised marrow cells of female C57Bl/6-Ly-5.2 mice. Compromised marrow cells had been subjected to two previous rounds of transplantation and regeneration in female mice.60 Cells cultured in suspension were also tested for reconstituting capabilities after 7 days of incubation with a single cytokine or combinations of cytokines. Fractions equivalent to one fifth of a day-7 culture that had been initiated with 1,000 Lin−Sca-1+c-kit+ cells were injected into female C57Bl/6-Ly-5.2 mice together with compromised marrow cells. Peripheral blood was obtained from the retro-orbital venous plexus using heparin-coated micropipettes (Drummond Scientific Co, Broomall, PA) 2 and 5 months after transplantation. Red blood cells were lyzed by 0.15 mol/L NH4Cl. The samples were then used for flow cytometric analysis of donor-derived cells by staining with FITC-conjugated anti–Ly-5.1 (A20-1.7). Donor cells in T-cell, B-cell, granulocyte, and monocyte/macrophage lineages at 5 months posttransplantation were analyzed by staining with biotin-conjugated anti–Thy-1.2, biotin-conjugated anti-CD45R/B220, biotin-conjugated anti–Gr-1, and biotin-conjugated anti–Mac-1. For indirect staining of cells with biotin-conjugated antibodies, cells were first incubated with biotin-conjugated antibodies, followed by staining with streptavidin-conjugated PE.
PKH26 experiments.
Lin−Sca-1+c-kit+ cells of male C57Bl/6-Ly-5.1 mice were stained with PKH26 (Sigma ImmunoChemicals, St Louis, MO) according to the manufacturer's instruction. Briefly, 1 × 105 cells were suspended in 1 mL of diluent (Sigma ImmunoChemicals) and immediately transferred into a polypropylene tube containing 1 mL of 4 × 10−7 mol/L PKH26 in diluent. After incubation for 5 minutes at room temperature, the staining reaction was stopped by adding 2 mL of fetal calf serum. One minute later, the total volume was brought up to 8 mL with α-medium supplemented with 10% fetal calf serum, and the cells were washed three times by using the serum-containing medium. After the third wash, cells were incubated with TPO alone or the combination of IL-6 and FL in a 75-cm2 flask (Corning Coster Co, Cambridge, MA) in 100 mL for 7 days. After washing and staining with 1 μg/mL of propidium iodine (PI), the cells were then analyzed for PKH26 fluorescense on FACS Vantage. Dead (PI-positive) cells were excluded from the sort gate. The viable cells were separated into PKH26low and PKH26high populations by sorting and their reconstituting abilities were tested by transplanting the sorted cells into lethally irradiated Ly-5 congenic mice together with 4 × 105compromised marrow cells. Flow cytometric analyses of the donor-derived cells were performed as described above at 5 months after transplantation. For indirect staining of cells with biotin-conjugated antibodies, cells were first incubated with biotin-conjugated antibodies, followed by staining with streptavidin-conjugated APC instead of PE, because the emission wave length of PE is similar to that of PKH26.
RESULTS
Effects of cytokines on colony formation.
Colony formation from Lin−Sca-1+c-kit+ cells of 5-FU–treated mice is presented in Table 1. As a single agent, FL, IL-6, and TPO in either 100 or 1,000 ng/mL concentrations failed to support significant colony formation. However, IL-6 in synergy with FL supported formation of a number of colonies. Colony types could not be determined in situ, because most colonies supported by the combination of IL-6 and FL contained a number of immature cells.15 Cultures containing a combination of TPO and FL yielded only few Meg colonies, indicating that TPO does not synergize with FL. Lin−Sca-1+c-kit+ cells exhibited about 45% colony-forming efficiency when cultured in cytokine cocktails, SF, IL-3, IL-6, FL, TPO, and EPO.
Effects of cytokines on maintenance of cells and progenitors.
We next studied the effects of cytokines on the maintenance of cells and colony-forming cells in 7-day suspension culture. The results shown in Table 2 are representative of three experiments. No viable cells were present after 7 days of incubation in medium alone. TPO was the most effective single agent to maintain the survival of progenitors, including CFU-GEMM and CFU-Meg, whereas IL-6 showed minimal activity and FL failed to support the survival of progenitors. The maintenance of progenitors by TPO reached a plateau at concentrations of less than 100 ng/mL. There was no synergy between TPO and FL, because the addition of FL to TPO did not enhance the survival of total CFU. This observation is in agreement with the study of colony formation in methylcellulose culture (Table 1). In contrast, the combination of IL-6 and FL significantly expanded the total population of CFU.
Effects of cytokines on the maintenance of long-term repopulating cells.
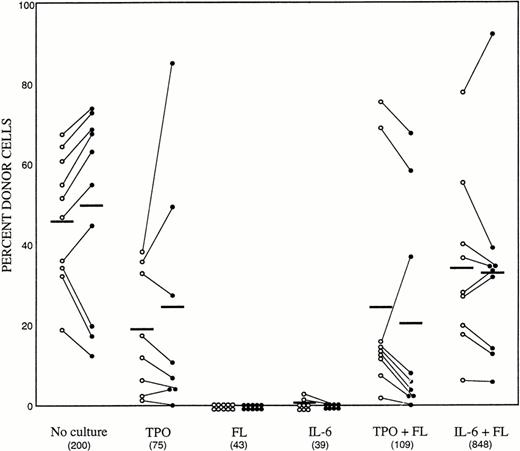
We then tested the in vivo reconstituting ability of the cultured cells. Based on the observations presented in Tables 1 and 2, the experiment was performed with 100 ng/mL TPO, 100 ng/mL FL, and 100 ng/mL IL-6. As also described in Table 2, the suspension cultures were initiated with 1,000 Lin−Sca-1+c-kit+ C57Bl/6-Ly-5.1 cells in a final volume of 1 mL. After 7 days of incubation, cells in each well were harvested and 1/5 aliquots were injected individually into 5 lethally irradiated Ly-5.2 recipients. As a control group, we transplanted 200 freshly prepared Lin−Sca-1+c-kit+ C57Bl/6-Ly-5.1 cells. The results of analyses of peripheral blood nucleated cells are presented in Fig 1. The numbers of cells transplanted per animal are also presented. Again, the cell population expanded only in the group containing IL-6 and FL. Transplantation of 200 fresh enriched cells resulted in engraftment in 10 of 10 mice, at the levels of 46.4% ± 16.2% and 49.1% ± 24.7% donor cells at 2 and 5 months posttransplantation, respectively. As single agents, both FL and IL-6 failed to support the survival of stem cells. In contrast, the recipients of the cells incubated with TPO alone showed donor cell engraftment in 8 of 8 mice at 2 months (19.7% ± 17.1%) and in 7 of 8 mice at 5 months (24.2% ± 29.8%) posttransplantation. Similar to the result of maintenance of colony-forming cells, the addition of FL to TPO had no benefit. Recipients of the cells cultured with IL-6 and FL showed engraftment levels of 34.3% ± 21.7% and 32.8% ± 25.3% in the peripheral blood at 2 and 5 months posttransplantation, respectively. These data indicated that TPO alone has an ability to maintain the stem cells in culture and that IL-6 acts synergistically with FL to promote the maintenance of LTRC.
Long-term repopulating ability of freshly sorted bone marrow cells and cultured cells. (○) Two months posttransplantation. (•) Five months posttransplantation. Results from individual animals are linked by lines. The numbers in parentheses indicate the actual number of cells transplanted per recipient.
Long-term repopulating ability of freshly sorted bone marrow cells and cultured cells. (○) Two months posttransplantation. (•) Five months posttransplantation. Results from individual animals are linked by lines. The numbers in parentheses indicate the actual number of cells transplanted per recipient.
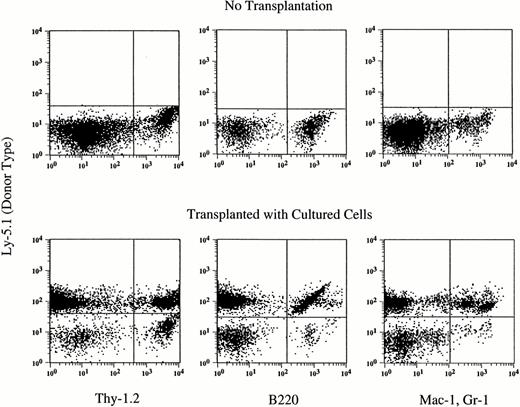
At 5 months posttransplantation, the proportions of donor blood nucleated cells in each of the T-cell, B-cell, and myeloid (granulocyte and monocyte/macrophage) compartments were determined. As shown in Table 3, T and B lymphocytes and myeloid cells were detected in the peripheral blood of all engrafted recipients and the myeloid/lymphoid ratios did not differ between groups. An example of the analysis of a mouse transplanted with the cells that were cultured with TPO alone is shown in Fig 2.
Hematopoietic reconstitution by cells cultured with TPO alone. Nucleated cells of peripheral blood were analyzed by flow cytometry 5 months after transplantation. Thy-1.2+ cells, B220+ cells, and Gr-1+Mac-1+cells of donor (Ly-5.1) origin are seen. The analyses of additional samples are presented in Table 4.
Hematopoietic reconstitution by cells cultured with TPO alone. Nucleated cells of peripheral blood were analyzed by flow cytometry 5 months after transplantation. Thy-1.2+ cells, B220+ cells, and Gr-1+Mac-1+cells of donor (Ly-5.1) origin are seen. The analyses of additional samples are presented in Table 4.
Mechanisms of LTRC maintenance.
Earlier, we reported that primitive murine progenitors in G0 are stimulated to proliferate in culture by a combination of IL-6 and FL.15 Therefore, the maintenance of LTRC in culture by the combination in of IL-6 and FL may be the result of active cell division and self-renewal of stem cells rather than the survival of LTRC in dormancy. To test this hypothesis, we performed a reconstitution experiment by using PKH26. This dye has been used previously to study division history of hematopoietic cells in culture,61-64 because its intensity is reduced by roughly one half with each cell division. Dot plots of the cells stained with PKH26 dye before (A) and after suspension culture (B and C) are presented in Fig 3. We made an arbitrary separation of PKH26high from PKH26lowpopulations as shown in Fig 3. Before culture, all cells were PKH26high. After 7 days of culture, the majority of the cells became PKH26low. The four cell populations defined in Fig 3 (BI, BII, CI, and CII) were prepared by sorting, and their in vivo reconstituting abilities were tested by transplantation into lethally irradiated Ly-5.2 mice together with 4 × 105compromised marrow cells of female C57Bl/6-Ly-5.2 mice. One fourth aliquots of the sorted PKH26low cells (2,720 cells) or 1/4 aliquots of the PKH26high cells (250 cells) cultured with TPO were injected individually into each of 4 recipients. One fifth aliquots of the sorted PKH26low cells (36,415 cells) or 1/5 aliquots of PKH26high cells (1,055 cells) cultured with IL-6 and FL were injected individually into each of 5 recipients. Of the cells cultured under either conditions, PKH26highpopulations showed greater reconstitution activity than that of PKH26low populations (Fig 4). In the IL-6 plus FL group, PKH26low cells exhibited some reconstituting capability. There may have been a limited amount of self-renewal. The proportions of donor blood nucleated cells in each of T-cell, B-cell, and myeloid (granulocyte and monocyte/macrophage) compartments were determined. T and B lymphocytes and myeloid cells were detected in the peripheral blood of all engrafted recipients (Table 4). These data indicated that TPO alone and combination of IL-6 and FL can support the survival of stem cells without stimulating their active cell proliferation.
Flow cytometric analysis and sorting windows of cells tracked with PKH26. (A) PKH26 fluorescence of freshly sorted Lin−Sca-1+c-kit+ cells stained with PKH26 on day 0. (B and C) PKH26 fluorescence of the cell populations after 7 days of suspension culture with TPO (B) or IL-6 and FL (C). Viable (PI-negative) cells were arbitarily divided into PKH26low populations (I) and PKH26highpopulations (II): BI, 89%; BII, 8%; CI, 94%; and CII, 2.7%.
Flow cytometric analysis and sorting windows of cells tracked with PKH26. (A) PKH26 fluorescence of freshly sorted Lin−Sca-1+c-kit+ cells stained with PKH26 on day 0. (B and C) PKH26 fluorescence of the cell populations after 7 days of suspension culture with TPO (B) or IL-6 and FL (C). Viable (PI-negative) cells were arbitarily divided into PKH26low populations (I) and PKH26highpopulations (II): BI, 89%; BII, 8%; CI, 94%; and CII, 2.7%.
Hematopoietic reconstitution by PKH26 stained cells. The four cell populations in Fig 3 (BI, BII, CI, and CII) were prepared by sorting and their in vivo reconstituting abilities were analyzed at 5 months posttransplantation.
Hematopoietic reconstitution by PKH26 stained cells. The four cell populations in Fig 3 (BI, BII, CI, and CII) were prepared by sorting and their in vivo reconstituting abilities were analyzed at 5 months posttransplantation.
DISCUSSION
In this study, we have examined the effects of TPO, FL, and IL-6 on the survival of murine LTRC. We used Lin−Sca-1+c-kit+ marrow cells that had been prepared from 5-FU–treated mice. This cell population represents 0.04% of the unfractionated marrow cells and has been shown to be capable of hematopoietic reconstitution in lethally irradiated mice.28,65 As a single agent, TPO was the best for supporting the maintenance of colony-forming cells and survival of LTRC in 7-day suspension culture. Earlier, Kaushansky et al32,66had reported that TPO alone can support the survival without proliferation of a fraction of mouse Hoescht 33342lowRhodamine123low cells that are highly enriched for LTRC. Together, those results are consistent with the recent observations that megakaryocyte, erythroid, and myeloid progenitors are reduced in TPO-deficient mice.53 54 TPO appears to be an important cytokine in the physiological regulation of hematopoietic stem cells.
FL as a single agent failed to maintain the viability of colony-forming cells or LTRC. Muench et al27 described that FL can support the survival of human fetal liver colony-forming cells in suspension culture for 7 days. Human fetal liver cell progenitors may respond to FL differently from adult murine marrow progenitors. Veiby et al22 reported that FL alone can support the survival of murine bone marrow colony-forming cells in suspension culture for 40 hours. This incubation period was much shorter than ours. As summarized in a recent review, a number of investigators have shown synergy between FL and early-acting cytokines, such as IL-6, IL-11, and G-CSF, on primitive hematopoietic progenitors.67 In this report, we demonstrated that FL and IL-6 synergize to support in vitro survival of stem cells. These observations are consistent with the report from Mackarehtschian et al68 that bone marrow cells of FLT3/FLK-2–deficient mice have impaired competitive long-term repopulating ability.
We reported previously that TPO and IL-6 belong to the same group of cytokines that trigger the dormant hematopoietic progenitors into cell cycle.6,30 Because of their functional similarity, we compared their effects on the survival of colony-forming cells and LTRC. In contrast to TPO, IL-6 alone could not support the survival of LTRC, which is in agreement with the report by Li and Johnson.24 Although IL-6 and FL showed little effects as single agents, they acted synergistically to promote the survival of LTRC. Earlier, McKinstry et al69 reported that the IL-6 receptor is expressed by the cell population highly enriched for LTRC. These observations are consistent with the report from Bernad et al70 that absolute numbers of CFU-Sd12, pre-CFU-S and LTRC are decreased in IL-6–deficient mice.
PKH26high populations cultured with either TPO alone or the combination of IL-6 and FL were more efficient in their bone marrow reconstitution activity than PKH26low populations. Traycoff et al64 documented similar results by using cells cultured with SF, IL-1α, IL-3, and IL-6. These results suggest that the ability of cultured cells to contribute to long-term reconstitution may be derived primarily from quiescent cells and not from de novo-generated progeny stem cells.
ACKNOWLEDGMENT
The authors thank Dr Haiqun Zeng for assistance in cell sorting, Dr Pamela N. Pharr and Anne G. Livingston in preparation of this manuscript, and the staff of the Radiation Oncology Department of the Medical University of South Carolina for irradiation of mice.
Supported by National Institutes of Health Grants No. RO1 DK32294 and RO1 DK48714 and by the Office of Research and Development, Medical Research Service, Department of Veterans Affairs.
Address reprint requests to Makio Ogawa, MD, PhD, Ralph H. Johnson Medical Center, 109 Bee St, Charleston, SC 29401-5799.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" is accordance with 18 U.S.C. section 1734 solely to indicate this fact.
© 1998 by the American Society of Hematology.