Abstract
The sequence of coagulant reactions in vivo following vascular injury is poorly characterized. Using quantitative immunoassays, the time courses were evaluated for activation of prothrombin, factor (F)V, FXIII, fibrinogen (Fbg) cleavage, and FVa inactivation in bleeding-time blood collected at 30-second intervals from 12 healthy subjects both before and after aspirin ingestion. Prothrombin decreased at a maximum rate of 14.2 ± 0.6 nM per second to 10% of initial values at the end of bleeding. Significant amounts of α-thrombin B chain appeared rapidly at 90 seconds of bleeding and increased at a maximum rate of 0.224 ± 0.03 nM per second to a peak value of 38 nM. Kinetics of prethrombin 2 generation was almost identical. Prothrombinase concentration reached a peak value of 22 pM at 150 seconds and then decreased to 9 pM at the end of bleeding. Prothrombin fragment 1.2 (F1.2) was produced explosively (0.673 ± 0.05 nM per second), whereas thrombin-antithrombin III (TAT) complexes were generated at a much slower rate (0.11 ± 0.008 nM per second;P = .002). FVa light chain was detectable 30 seconds later than the heavy chain (150 seconds) and was produced at a slightly slower rate (0.027 ± 0.001 nM per second) when compared with the heavy chain (0.032 ± 0.002 nM per second; P = .041). The 30 000 fragment (residues 307-506) of FVa heavy chain produced by activated protein C appeared as early as at 90 seconds and increased with time. Fbg was removed from the blood shed with a high rate of 0.047 ± 0.02 μM/s and became undetectable at approximately 180 seconds of bleeding. The velocity of FXIII activation correlated with thrombin B-chain formation. A 7-day aspirin administration (75 mg/d) resulted in significant reductions in maximum rates of (1) prothrombin removal (by 29%; P = .008); generation of α-thrombin B-chain (by 27.2%; P = .022), and prethrombin 2 (by 26%; P = .014); formation of F1.2 (by 31.4%;P = .009) and TAT (by 30.3%; P = 0.013); (2) release of FVa heavy chain (by 25%; P = .003) and FVa light chain (by 29.6%; P = .007); (3) Fbg depletion from solution (by 30.5%; P = .002); and (4) FXIII activation (by 28.6%; P = .003). Total amounts of the proteins studied, collected at every interval, also significantly decreased following aspirin ingestion. These results indicate that low-dose aspirin impairs thrombin generation and reactions catalyzed by this enzyme at the site of the injury.
Introduction
Hemostasis is triggered in vivo when vascular damage exposes membrane-bound tissue factor (TF) constitutively present in the subendothelium.1 The proteolytically active complex of TF and circulating 2-chain factor (F)VIIa, the extrinsic tenase, activates FX and FIX. FIXa and FXa, complexed with FVIIIa and FVa, respectively, form 2 enzyme-cofactor complexes—the intrinsic tenase and prothrombinase, which converts prothrombin to thrombin. The former complex activates FX at a 50-fold higher rate than the FVIIa-TF complex, whereas prothrombinase is 105 times more efficient in generating thrombin than FXa acting alone.2 The latter complexes assemble on negatively charged phospholipids, exposed predominantly on activated platelets.3,4 These enzymes are regulated by a number of stoichiometric protein inhibitors, including tissue factor protein inhibitor (TFPI), antithrombin III (ATIII), and heparin cofactor II (HCII), and by the dynamic inhibitor activated protein C (APC), which is produced by the catalyst thrombin-thrombomodulin.1 2
Generation of α-thrombin plays a central role in the blood coagulation cascade. Once formed, thrombin converts fibrinogen (Fbg) to insoluble fibrin via cleavages at Arg16 in the Aα-chain and at Arg14 in the Bβ-chain, releasing fibrinopeptides A (FPA) and B (FPB), respectively.5 Resistance of the polymerized fibrin monomers to plasmin is the consequence of the action of FXIII, which is also activated by α-thrombin. Following release of an NH2-terminal peptide from the A subunit of FXIII, its active form catalyzes the rapid formation of covalent cross-links between γ-carboxamido and ε-amino groups of glutamyl and lysyl residues in the γ chains.6 All these processes are critical to the formation of stable blood clots. Thrombin up-regulates its own generation through activation of FV, FVIII, and FXI.1 Thrombin bound to thrombomodulin in the endothelium catalyzes protein C activation, leading to the formation of APC. APC bound to thrombomodulin inactivates 2 crucial cofactors, FVa and FVIIIa.7
Blood coagulation has been studied extensively in purified systems,8-10 plasma,11 and anticoagulated and nonanticoagulated whole blood12,13 in vitro. A model of microvascular injury, introduced by Thorngren and coworkers,14 has also been applied to study coagulant events triggered by standardized bleeding-time skin incisions.15 This flowing blood system provides insight into the intricately interwoven hemostatic process involving tissue damage, plasma coagulation reactions, the activation of platelets, the endothelium, and subendothelial tissue.16,17 Weiss and Lages17 found in patients with deficiencies of FV or FX that the production of FPA in the wounds depends on activation of FX by the complex TF-FVIIa and is closely coupled with platelet activation, reflected by a rapid release of platelet factor 4 (PF4). Previous studies undertaken to quantitate thrombin generation in bleeding-time blood focused on the measurement of FPA, thrombin-antithrombin III complexes (TAT), and prothrombin fragment 1.2 (F1.2) concentrations.17-20 We wondered whether the model of microvascular injury could also be used to monitor other coagulant reactions that regulate thrombin formation, or are catalyzed by this enzyme (ie, activation of FV and FXIII or inactivation of FVa).
Aspirin has been proven effective in secondary prevention of coronary artery disease (CAD). Its efficacy has been ascribed to its antiplatelet action through inhibiting thromboxane A2-mediated platelet aggregation.21,22 In various experimental settings aspirin has been shown to reduce thrombin generation.12,23,24 Using the model of microvascular injury, it has been demonstrated that aspirin at a dose from 30 to 500 mg down-regulates thrombin formation in healthy subjects15,25 and patients with CAD.19 26
The purpose of the present study is to describe major coagulant events during blood clotting at the site of microvascular injury and to evaluate the effect of low-dose aspirin on the activation of prothrombin, FV, FXIII, and Fbg, and inactivation of FVa by APC.
Materials and methods
Materials
Tris-HCl and HEPES were purchased from Sigma (St Louis, MO) and Tween-20 from J. T. Baker (Phillipsburg, NJ). Murine monoclonal α-Fbg 3A was raised against the Aα chain of Fbg.14Murine monoclonal α-FVaHC no.17 and α-FVaLCno. 9 recognize the human FVa heavy chain (residues 307-506) and FVa light chain, respectively.27,28 Burro polyclonal α-prethrombin-1 antibody, which recognizes prothrombin, prethrombin 1, prethrombin 2, fragment 2, prothrombin, and α-thrombin B chain, was prepared in the Division of Hematology Research, Mayo Clinic (Rochester, MN).29 Rabbit polyclonal α-FXIII (D4679), raised against the subunit A, was a gift from Dr Gerry Lasser (Zymogenetics, Seattle, WA).30 Horseradish peroxidase–labeled goat anti–rabbit IgG, antimouse IgG, and antihorse IgG antibodies were purchased from Southern Biotech (Birmingham, AL). Molecular standards were purchased from Gibco-BRL (Gaithersburg, MD). Chemiluminescent substrate was purchased from NEN Life Science Products (Boston, MA).
Subjects
Twelve healthy male volunteers, aged 21 to 25 years, were studied. Routine laboratory blood tests, including prothrombin time, activated partial thromboplastin time, platelet count (2.3 ± 0.1 × 108 platelets/mL), and plasma levels of Fbg (2.61 ± 0.12 g/L) and ATIII (0.22 ± 0.08 g/L) determined nephelometrically (Dade Behring, Marburg, Germany) were within the normal range. None of the subjects had a personal or familial history of thrombotic or bleeding disorders. The volunteers denied taking any medication during at least 4 weeks preceding the study. Platelet aggregation in platelet-rich plasma was observed in response to arachidonic acid (300 μM). To evaluate effects of low-dose aspirin on blood coagulation, aspirin (75 mg/d) was given over the course of 7 days. The protocol was approved by the Jagellonian University School of Medicine Ethical Committee, Cracow, Poland and all subjects gave informed consent.
Model of microvascular injury
The TF-initiated coagulation was evaluated in samples of bleeding-time blood, as described previously.25 Briefly, after compressing the upper arm with a sphygmomanometer cuff to 40 mm Hg, 2 incisions were made on the lateral aspect of a forearm parallel to the antecubital crease using a Simplate II device (Organon Teknika, Durham, NC). The blood shed was collected at 30-second intervals (on average, 10 samples) using heparinized capillary tubes (Kabe Labortechnik, Nümbrecht-Elsenroth, Germany) and then expressed into Eppendorf tubes. An anticoagulant cocktail, provided by an FPA assay (Diagnostica Stago, Asnieres, France), contained sodium citrate, aprotinin, chloromethyl ketone, and heparin (vol/vol, 1:10). Volumes of all the samples and bleeding time were recorded. The procedure was performed by the same investigator. Soluble and insoluble material was immediately centrifuged at 4°C at 2000g for 20 minutes; then the supernatants were removed, aliquoted, and stored at −80°C for further analysis.
Gel electrophoresis and Western blotting
The supernatant aliquots were separated on 5% to 15% linear gradient sodium dodecyl sulfate–polyacrylamide electrophoresis (SDS-PAGE) gels under reducing (1% 2-mercaptoethanol) or nonreducing conditions according to modified Laemmli procedures.13,31Transfer from the gels to nitrocellulose membranes (Bio-Rad, Hercules, CA) was performed as described by Towbin and coworkers.32The membranes were blocked with 5% nonfat dry milk in HEPES buffered saline, pH 7.4 (HBS) at room temperature for 1 hour. After washing, the blots were incubated for an additional hour with the primary antibodies (in Tris buffered saline + 0.01% Tween 20, pH 7.4 [TBST]): α-prethrombin-1 (7.5 μg/mL), α-FVaHC no.17 (7.5 μg/mL), α-FVaLC no.9 (7.5 μg/mL), α-FXIII (5 μg/mL), and α-Fbg 3A (1.5 μg/mL). This was followed by a 60-minute incubation with the appropriate horseradish peroxidase–conjugated secondary antibody (1:5000-10 000 dilution): for FVa and Fbg—goat α-mouse; for prothrombin and its activation products—goat α-horse; for FXIII—goat α-rabbit. The proteins were visualized by the addition of a chemiluminescence reagent to the membranes. The blots were developed in a Kodak XOMAT as described by Rand and colleagues.13 FVa light- and heavy-chain generation, prothrombin activation, Fbg cleavage, and FXIII activation as a function of time were determined and quantified by densitometry of immunoblots on a Hewlett-Packard Scanjet 4C/T. Concentrations were estimated from serial dilutions of purified internal standard proteins by horizontal comparison of sample band density. Relative concentrations were determined by normalizing the data with regard to the maximum. Changes in the reaction rates, as reflected by the concentrations of immunoreactive fragments from the immunoblots, were analyzed using IGOR Pro Version 3.1 software (Wavemetrics, Lake Oswego, OR). A mean value was calculated from the compilation of 30-second interval densitometric analysis. On the basis of concentrations and volumes of blood samples, the absolute amounts of the proteins studied were calculated and analyzed.
In addition to immunoblotting, F1.2 and TAT concentrations in bleeding-time blood were measured by enzyme-linked immunosorbant assay (ELISA) using commercially available kits (Enzygnost F1.2 and Enzygnost TAT, Dade Behring). Concentrations of both markers were also measured in citrated plasma of peripheral venous blood (vol/vol, 1:9). Prothrombinase concentration was also calculated at each 30-second interval using an equation introduced by Rand and colleagues13: [prothrombinase] = v (Km + [S])/kcat [S], where [S] denotes free prothrombin concentration, determined by densitometry of immunoblots. A value of v was estimated by densitometric analysis of α-thrombin B-chain concentration. Profiles for all the parameters studied were constructed from the averaged data, determined for the 12 healthy subjects studied.
Statistical analysis
Data are presented as mean ± SEM. Measurements before and after aspirin administration were compared with the Wilcoxon signed-rank test. To evaluate correlations between variables, Spearman coefficient was calculated. P less than .05 was considered statistically significant.
Results
The initial bleeding time was 344.6 ± 18.4 seconds and aspirin at 75 mg/d caused a significant increase to 110.2 ± 14.1 seconds (P = .001). Blood volume increased to a maximum value at 150 seconds then declined until bleeding ceased. The total volume of blood collected from bleeding-time wounds was 124.2 ± 8.7 μL and 169.0 ± 13.5 μL (P = .007) before and after aspirin administration, respectively. At the first 8 time points, the volumes of samples were significantly larger (by about 20%) after aspirin than before treatment.
Although no correlation was found between the volume of the samples and concentrations of the parameters measured, we decided to express levels of the products studied in bleeding-time blood both as a concentration and as a total amount in the 30-second interval, because the extent to which vessels are injured by incisions is standardized and consistent from sample to sample. When posttreatment levels of the products are compared with those calculated before aspirin ingestion using both concentration and total methods, significant differences were observed using both analyses.
Markers of thrombin generation
Prior to aspirin, the mean plasma TAT concentration was 0.026 ± 0.006 nM and F1.2 was 0.72 ± 0.05 nM. There was no change in either parameter following low-dose aspirin (0.021 ± 0.004 and 0.67 ± 0.08 nM, P > .05, respectively). These values were within the normal range, according to the manufacturer of both ELISA kits.
The time courses for TAT and F1.2 formation in the 30-second bleeding-time blood samples are depicted in Figure1, with open symbols representing values before aspirin treatment. The data are plotted as concentration (triangles; nM, left y-axis) or total amount (squares; fmol, right y-axis) versus time (seconds). The generation of both thrombin markers (Figure 1A,B) follows a similar pattern of 2 phases: a 60- to 90-second “initiation” phase and a subsequent “propagation” phase, as seen in other models of blood coagulation.9,11,12 25Prothrombin F1.2 (Figure 1A, squares) is generated at a rate of 6.13 ± 0.5 fmol/s with total yields reaching 1250 fmol by 240 seconds (P = .03). In contrast TAT (Figure 1B, circles) complexes are formed at a much lower rate (1.43 ± 0.04 fmol/s) with total amounts reaching more than 210 fmol by 180 seconds. Overall, F1.2 was produced much more rapidly than TAT (P = .004). The velocity of the increase of F1.2 (triangles) concentrations was also significantly higher than that of TAT (diamonds) concentrations (at maximum, 0.673 ± 0.05 versus 0.11 ± 0.008 nM per second;P = .002). Levels of F1.2 and TAT were positively correlated (r = 0.79; P = .004).
Fl.2 and TAT complexes determined by ELISA, in the 30-second bleeding-time blood samples.
(A) Total amounts of F1.2 before (open squares) and after aspirin treatment (closed squares). Concentrations of F1.2 in each sample before (open triangles) and after aspirin treatment (closed triangles). (B) TAT before (open circles) and after aspirin treatment (closed circles). Concentrations of TAT in each sample before (open diamonds) and after aspirin treatment (closed diamonds). Values are plotted as means ± SEM.
Fl.2 and TAT complexes determined by ELISA, in the 30-second bleeding-time blood samples.
(A) Total amounts of F1.2 before (open squares) and after aspirin treatment (closed squares). Concentrations of F1.2 in each sample before (open triangles) and after aspirin treatment (closed triangles). (B) TAT before (open circles) and after aspirin treatment (closed circles). Concentrations of TAT in each sample before (open diamonds) and after aspirin treatment (closed diamonds). Values are plotted as means ± SEM.
Comparison of the time courses for TAT and F1.2 generation before (open symbols) and after aspirin (closed symbols) ingestion showed that low-dose aspirin greatly impairs the formation of both thrombin markers at all the 30-second intervals except for the first time point (Figure1). Maximum rates for F1.2 and TAT (expressed as the total amounts of both parameters) were reduced to 1.11 ± 0.08 fmol/s and 0.53 ± 0.03 fmol/s, respectively, which corresponds to 70.1% (P = .009) and 67.1% (P = .013) of the baseline values, respectively. Likewise, the velocity of F1.2 (triangles) and TAT (diamonds) formation, when expressed as a concentration in the bleeding-time blood samples, was significantly decreased by 31.4% (P = .007) and 30.3% (P = .019) of the initial values, respectively.
Prothrombin activation products
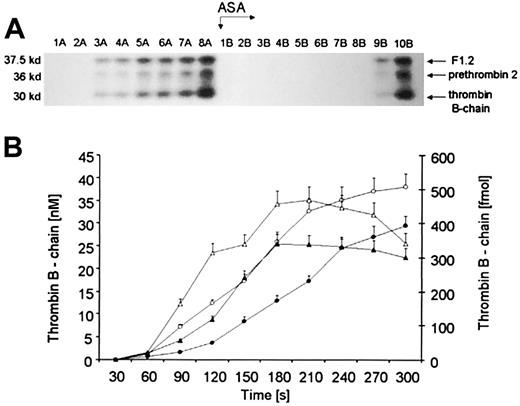
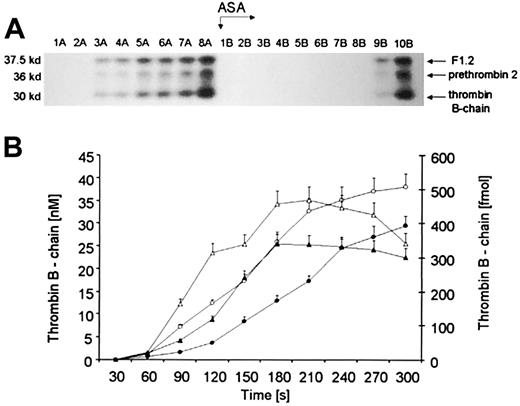
The Western immunoblot depicted in Figure2A displays fragments reactive to a polyclonal serum against human α-prethrombin-1 under reducing conditions. Thirty-second time points before (1A-8A) and after aspirin treatment (1B-10B) are illustrated above the blot. The bottom bands (Mr = 30 000; residues 321-579) correspond to α-thrombin B chain, derived from α-thrombin and meizothrombin, which are not distinguishable using this polyclonal antibody.13 The middle Mr = 36 000 bands have similar mobility to those reported for prethrombin 2 (residues 272-579 in prothrombin). The upper bands most likely represent the product of prothrombin activation, F1.2 (residues 1-271). The same triplet patterns of prothrombin activation products are seen on immunoblots following aspirin administration.
Quantitation of prothrombin activation products.
(A) A representative immunoblot displaying α-thrombin B chain (bottom band), prethrombin 2 (middle band), and F1.2 (upper band). Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under reducing conditions. Three activation products were probed with a polyclonal antibody, which recognizes prothrombin, prethrombin 1, prethrombin 2, fragment 2, prothrombin, and α-thrombin B chain. (B) Quantitative analysis of the generation of α-thrombin B chain (circles) in bleeding-time blood. This product, estimated by densitometry and comparison to standards, is presented as a function of time (seconds). Concentration of α-thrombin B chain before (open circles) and after aspirin (ASA) treatment (closed circles). Total amounts of thrombin B chain in each sample before (open triangles) and after aspirin treatment (closed triangles). Values are plotted as means ± SEM.
Quantitation of prothrombin activation products.
(A) A representative immunoblot displaying α-thrombin B chain (bottom band), prethrombin 2 (middle band), and F1.2 (upper band). Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under reducing conditions. Three activation products were probed with a polyclonal antibody, which recognizes prothrombin, prethrombin 1, prethrombin 2, fragment 2, prothrombin, and α-thrombin B chain. (B) Quantitative analysis of the generation of α-thrombin B chain (circles) in bleeding-time blood. This product, estimated by densitometry and comparison to standards, is presented as a function of time (seconds). Concentration of α-thrombin B chain before (open circles) and after aspirin (ASA) treatment (closed circles). Total amounts of thrombin B chain in each sample before (open triangles) and after aspirin treatment (closed triangles). Values are plotted as means ± SEM.
Before aspirin treatment, all 3 products of prothrombin activation appeared rapidly and simultaneously after an initial 60- to 120-second lag phase, defined as a time when its concentration, assessed by densitometry of immunoblots, is below 10 nM (Figure 2A, lanes 1-8A). Following aspirin ingestion (arrow ASA) the bands were detectable approximately 150 seconds (P = .04) later than before treatment (lanes 1-8B). The kinetics of prethrombin 2 and thrombin B chain, estimated by densitometry of Western blots, showed a striking resemblance (data not shown). Prior to the ingestion of aspirin, a maximum rate of α-thrombin B-chain (Figure 2B, open circles) formation was about 0.224 ± 0.03 nM per second. Total amounts of α-thrombin B chain (Figure 2B, triangles) in each time interval showed an increase with time (4.92 fmol/s at maximum), being almost identical to that observed for prethrombin 2 (4.57 fmol/s at maximum; data not shown). There was a strong correlation between both α-thrombin B-chain and prethrombin 2 levels (r = 0.89;P = .002).
Comparing relative amounts of prothrombin activation products in bleeding-time blood, we found that the sum of thrombin B-chain and prethrombin 2 concentrations constituted about 50% to 70% of the F1.2 levels, determined by ELISA. Because TAT concentration accounts for 80% of the surplus, we conclude that other products of thrombin with other inhibitors including thrombomodulin, fibrin, and HCII probably represent the remaining 20%.
Aspirin ingestion resulted in a significant reduction (27.2%,P = .022) in the maximum rate (to 0.163 ± 0.02 nM per second) of α-thrombin B-chain formation (Figure 2B, closed circles). There was an almost identical decrease in the velocity of prethrombin 2 generation (by 26%; P = .014; data not shown). Total amounts of α-thrombin B chain (Figure 2B, closed triangles), estimated in each 30-second interval, also decreased by 24.8% (P = .028) as a result of aspirin ingestion. A similar fall (by 19.6%) was observed in total amounts of prethrombin 2 (P = .035; data not shown).
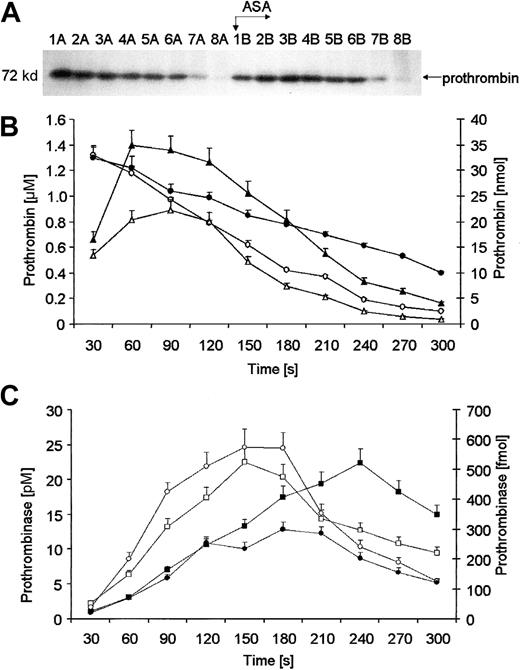
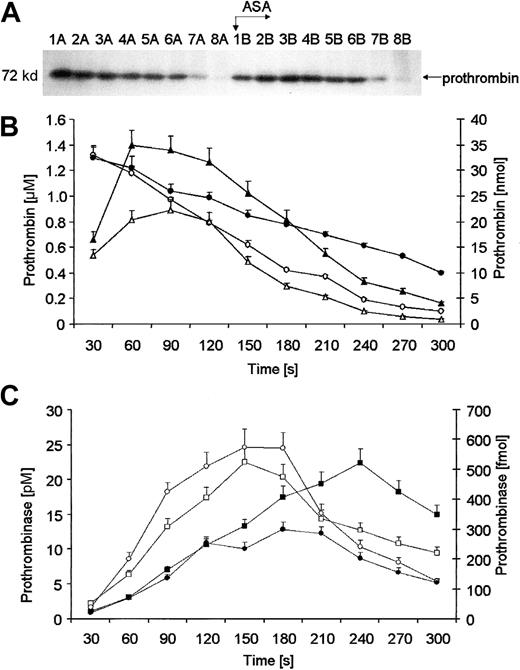
Prothrombin migrating at Mr = 72 000 was no longer detectable by approximately 210 seconds (Figure3A, lane 8A). Before aspirin administration, an initial concentration of 1.32 μM for prothrombin was detected and it disappeared from bleeding-time blood at a maximum rate of 14.5 ± 0.6 nM per second (Figure 3B, open circles). Following aspirin administration the rate of prothrombin removal was slowed by 25.4% (10.82 ± 0.4 nM per second at maximum;P = .03) and the protein could not be detected to the end of bleeding in 3 of 12 subjects (Figure 3A, lanes 1-8B and Figure 3B, closed circles). Absolute amounts of prothrombin were calculated for each 30-second interval (Figure 3B, diamonds). Analysis of such maximum rates of prothrombin removal showed that aspirin induced a significant decrease in the velocity of this process (closed diamonds) by 29.3% (0.266 ± 0.014 versus 0.188 ± 0.011 nmol/s;P = .034).
Quantitation of prothrombin.
(A) A representative immunoblot of prothrombin. Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under reducing conditions. Prothrombin was probed with a polyclonal antibody (Figure 2). Wells A—blood samples taken before aspirin (ASA) ingestion (75 mg/d for 7 days); wells B—samples after aspirin therapy. (B) Quantitative analysis of removal of prothrombin from supernatants of bleeding-time blood. Levels of prothrombin, determined by densitometry and comparison with standards, are presented as a function of time (seconds). Total amounts of prothrombin before (open triangles) and after aspirin treatment (closed triangles). Concentrations of prothrombin before (open circles) and after aspirin treatment (closed circles). (C) Prothrombinase formation calculated in each 30-second bleeding-time blood sample, as described in “Materials and methods.” Total amounts of prothrombinase before (open circles) and after aspirin treatment (closed circles). Concentrations of prothrombinase before (open squares) and after aspirin treatment (closed squares). Values are plotted as means ± SEM.
Quantitation of prothrombin.
(A) A representative immunoblot of prothrombin. Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under reducing conditions. Prothrombin was probed with a polyclonal antibody (Figure 2). Wells A—blood samples taken before aspirin (ASA) ingestion (75 mg/d for 7 days); wells B—samples after aspirin therapy. (B) Quantitative analysis of removal of prothrombin from supernatants of bleeding-time blood. Levels of prothrombin, determined by densitometry and comparison with standards, are presented as a function of time (seconds). Total amounts of prothrombin before (open triangles) and after aspirin treatment (closed triangles). Concentrations of prothrombin before (open circles) and after aspirin treatment (closed circles). (C) Prothrombinase formation calculated in each 30-second bleeding-time blood sample, as described in “Materials and methods.” Total amounts of prothrombinase before (open circles) and after aspirin treatment (closed circles). Concentrations of prothrombinase before (open squares) and after aspirin treatment (closed squares). Values are plotted as means ± SEM.
Using the Michaelis-Menten equation and results of densitometry of immunoblots for prothrombin and α-thrombin B chain, we calculated molar concentrations of prothrombinase (Figure 3C, squares) at 30-second time intervals and the functional amounts of this complex (Figure 3C, circles). Closed symbols represent values after aspirin treatment. Pretreatment concentrations of prothrombinase (open squares) increased rapidly within the first 150 seconds, reaching a maximum of 22 ± 2 pM, and then decreased slowly to about 10 ± 1.2 pM by the last sample. The highest rate of formation of the prothrombinase was estimated at 0.183 ± 0.09 pM per second. Aspirin ingestion resulted in a reduced rate of prothrombinase formation by 29% (P = .008). Posttreatment maximum of prothrombinase concentration (20 pM; closed squares) was equivalent and delayed to 240 seconds (Figure 3C). Analysis of total amounts of prothrombinase (Figure 3C, circles) confirmed that aspirin reduced its formation rate by 27.8% at maximum (P = .003), with only a delay in time to achieve equivalent concentration.
Factor V/Va
Factor V, derived from circulating blood and activated platelets (up to 20% of the total amount), could be seen on nonreduced gels, migrating at Mr = 330 000 (data not shown). This band, which is immunoreactive toward a monoclonal antibody raised against FVa heavy chain, disappeared slowly over the bleeding time. From these immunoblots, an initial concentration of FV was estimated to be 18.2 ± 2.1 nM and decreased to only 14.0 ± 1.1 nM by the last interval (P = .03). The maximal rate of disappearance of FV was calculated to be 0.022 ± 0.005 nM per second and was reduced (by 26%; P = .04) following aspirin administration (data not shown).
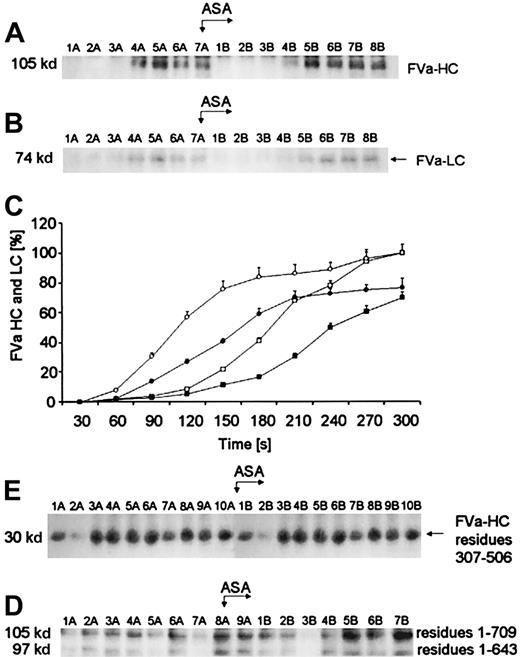
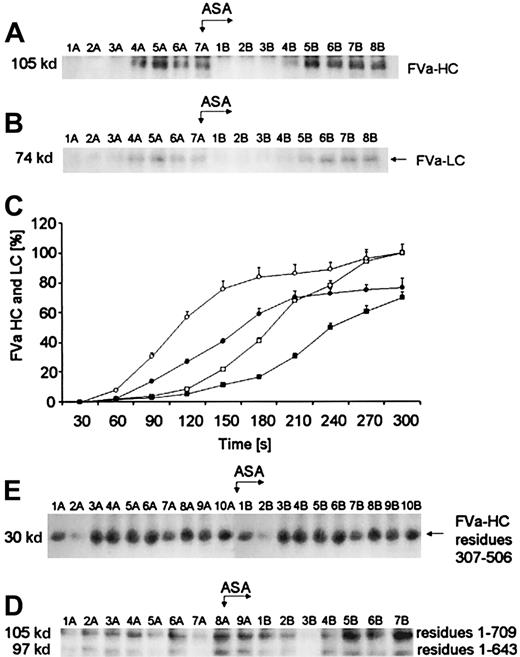
Active FV (FVa), derived from activation by α-thrombin or FXa, is composed of a noncovalently associated heavy chain (residues 1-709) and a light chain (residues 1546-2196). The Western immunoblots, seen in Figure 4, demonstrated that the maximum FVa heavy chain (Mr = 105 000) levels observed are generated within 120 seconds prior to aspirin administration (Figure4A, lane 4A). The light chain (Mr = 74 000) band is detectable almost at the same time (Figure 4B, lane 4A) but is significantly less dense. Quantitation of the FVa heavy and light chains, based on densitometry of the Western immunoblots, is presented in Figure 4C. The concentrations of the FV heavy and light chains inferred from internal standards are represented by circles and squares, respectively. Prior to aspirin therapy, the light chain (open squares) was produced at a similar rate (0.027 ± 0.001 nM per second) to that for the heavy chain (open circles, 0.032 ± 0.002 nM per second; P = .041). At 5 minutes, a decrease in FVa heavy chain concentration was observed in 3 of 12 subjects, whereas the bands representing FVa light chain reached a plateau of density after about 4 minutes of bleeding (data not shown). After aspirin administration, generation of FVa heavy and light chains was delayed by about 30 seconds (Figure 4A, lane 5B and Figure 4B, lane 5B,P = .023). By densitometry of the immunoblots, it was estimated that at the end of bleeding the maximal concentration of FVa heavy chain was 3.8 ± 0.2 nM and FVa light chain was 3.2 ± 0.15 nM (P = .01) prior to aspirin. Following aspirin ingestion peak values of both FVa chains were reduced approximately to 2.9 ± 0.11 nM (P = .021) and 2.1 ± 0.09 nM (P = .012), respectively. The posttreatment rate of FVa heavy-chain formation was reduced by 25% at maximum (P = .003), whereas that of the light chain fell by 29.6% (P = .007). The rates of FVa heavy- and light-chain generation were positively correlated (r = 0.73;P = .003). A similar correlation was found between the velocity of FVa light-chain and α-thrombin B-chain generations (r = 0.46; P = .025).
Generation of FVa heavy and light chains.
(A) An immunoblot showing generation of FVa heavy chain (FVa-HC) and (B) FVa light chain (FVa-LC) with disulfide bonds intact. In both panels, consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under nonreducing conditions. FVa-HC was probed with a monoclonal antibody reactive toward residues 307 to 506 of FVa, whereas FVa-LC was probed with a monoclonal antibody raised against the light chain of FVa. Wells A—blood samples taken before aspirin (ASA) ingestion (75 mg/d for 7 days); wells B—samples after aspirin administration. (C) Time courses for generation of both FVa chains in bleeding-time blood. Their relative levels are plotted versus time (seconds), taking a maximum concentration as 100. Concentration of FVa-HC before (open circles) and after aspirin treatment (closed circles). Concentration of FVa-LC before (open squares) and after aspirin treatment (closed squares). Values are plotted as means ± SEM. (D) An immunoblot of the product of FVa heavy-chain cleavage by APC (30 kd; residues 307-506). Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under nonreducing conditions. Immunoreactive fragments of FVa inactivation were probed with a monoclonal antibody against residues 307 to 506 of the heavy chain. Wells A—blood samples taken before aspirin ingestion (75 mg/d for 7 days); wells B—samples after aspirin treatment. (E) An immunoblot of FVa heavy chain (top band) and the 97-kd fragment of this chain (bottom band) in bleeding-time blood with disulfide bonds reduced. Both peptides are detected by a monoclonal antibody reactive toward residues 307 to 506 in FVa heavy chain. Wells A—blood samples taken before aspirin (ASA) treatment; wells B—samples after aspirin ingestion.
Generation of FVa heavy and light chains.
(A) An immunoblot showing generation of FVa heavy chain (FVa-HC) and (B) FVa light chain (FVa-LC) with disulfide bonds intact. In both panels, consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under nonreducing conditions. FVa-HC was probed with a monoclonal antibody reactive toward residues 307 to 506 of FVa, whereas FVa-LC was probed with a monoclonal antibody raised against the light chain of FVa. Wells A—blood samples taken before aspirin (ASA) ingestion (75 mg/d for 7 days); wells B—samples after aspirin administration. (C) Time courses for generation of both FVa chains in bleeding-time blood. Their relative levels are plotted versus time (seconds), taking a maximum concentration as 100. Concentration of FVa-HC before (open circles) and after aspirin treatment (closed circles). Concentration of FVa-LC before (open squares) and after aspirin treatment (closed squares). Values are plotted as means ± SEM. (D) An immunoblot of the product of FVa heavy-chain cleavage by APC (30 kd; residues 307-506). Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under nonreducing conditions. Immunoreactive fragments of FVa inactivation were probed with a monoclonal antibody against residues 307 to 506 of the heavy chain. Wells A—blood samples taken before aspirin ingestion (75 mg/d for 7 days); wells B—samples after aspirin treatment. (E) An immunoblot of FVa heavy chain (top band) and the 97-kd fragment of this chain (bottom band) in bleeding-time blood with disulfide bonds reduced. Both peptides are detected by a monoclonal antibody reactive toward residues 307 to 506 in FVa heavy chain. Wells A—blood samples taken before aspirin (ASA) treatment; wells B—samples after aspirin ingestion.
The loss of cofactor activity by FVa results from APC-mediated peptide bond cleavages in the heavy chain at Arg306, Arg506, and Arg679.28 On nonreduced gels, the complete inactivation product of FVa heavy chain, a fragment of Mr = 30 000 (residues 307-506; Figure 4D) was first seen as early as 90 seconds after incision (lane 3A). These bands migrated identically with the previously described inactivation products of FVa heavy chain.13 The density of the Mr = 30 000 bands tended to increase with some fluctuations over bleeding time. Factor Va inactivation was not influenced by a 7-day aspirin treatment (Figure 4D, lanes B).
A Western immunoblot in Figure 4E shows that the Mr = 97 000 fragment of FVa heavy chain (residues 1-643/644), which can be visualized under reducing conditions, is seen in 3 of 12 subjects who also displayed high concentrations of thrombin B-chain (> 38 nM). In all 3 subjects significant amounts of thrombin B chain as well as FVa heavy chain were detected as early as 60 seconds. By comparison with standards (not shown), the mobility of this band is identical to that observed with cultured human umbilical vein endothelial cells (HUVECs) in the presence of thrombin.33The formation of this FVa inactivation product is independent of APC activity. No relationship was identified between the velocity of APC inactivation of FVa and the Mr = 97 000 fragment release. Density of the Mr = 97 000 band increased slightly over bleeding time (Figure 4E). Aspirin had no effect on the kinetics of the Mr = 97 000 fragment formation in bleeding-time blood (Figure 4E).
Fbg
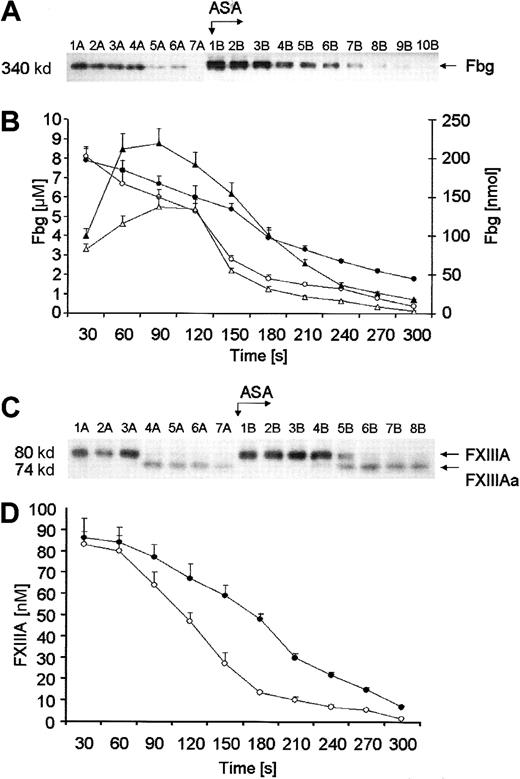
Fibrinogen in the fluid phase of bleeding-time blood was detectable by immunoblotting as a Mr = 340 000 band over the first 3 minutes (Figure 5A, lanes 1A-6A). Fbg was depleted rapidly from solution at a peak rate of 0.047 ± 0.02 μM/s (open circles, Figure 5B). The initial concentration of Fbg was estimated by densitometry to be 7.31 μM (2.48 g/L); close to the mean Fbg level determined nephelometrically (7.68 μM). Prior to aspirin ingestion Fbg was completely depleted at the end of bleeding in 9 of the 12 subjects studied, in contrast to only 3 of 12 subjects after aspirin treatment. A significant delay (60 seconds) in Fbg depletion from solution/fibrin formation was associated with aspirin ingestion (closed circles, P = .01). The rate of Fbg removal from bleeding-time blood after aspirin ingestion (closed circles) did not exceed 0.036 ± 0.006 μM/s, which corresponds to 30.5% ± 3.2% of the initial value (P = .002). The maximum rate of decrease in total amounts of Fbg (closed triangles, Figure 5B), estimated in each 30-second interval, was significantly lower by 24.4% following aspirin ingestion (P = .008). There was a significant correlation between the rate of Fbg depletion and that of α-thrombin B-chain formation (r = 0.52;P = .042). Surprisingly, the detection times at which α-thrombin B-chain is first observed and Fbg disappears were not significantly correlated (P = .17).
Quantitation of Fbg and FXIII.
(A) A representative immunoblot of Fbg. Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under nonreducing conditions. Wells A—blood samples taken before aspirin ingestion (75 mg/d for 7 days); wells B—samples after aspirin (ASA) administration. (B) Time courses for Fbg consumption. Levels of Fbg, determined by densitometry and comparison with standards, are presented as a function of time (seconds). Concentration of Fbg before (open circles) and after aspirin treatment (closed circles). Total amounts of Fbg in each sample before (open triangles) and after aspirin treatment (closed triangles). (C) A representative immunoblot of the subunit A of FXIII. Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under nonreducing conditions. FXIII and its active form were probed with a polyclonal antibody specific for both the unactivated (FXIIIA) and activated (FXIIIAa) A subunit. Wells A—blood samples taken before aspirin ingestion (75 mg/d for 7 days); wells B—samples after aspirin (ASA) administration. (D) Kinetics of FXIII activation in bleeding-time blood. FXIIIA concentration, determined by densitometry and comparison with standards, is presented as a function of time (seconds). Concentration of FXIIIA before (open circles) and after aspirin treatment (closed circles). Values are plotted as means ± SEM.
Quantitation of Fbg and FXIII.
(A) A representative immunoblot of Fbg. Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under nonreducing conditions. Wells A—blood samples taken before aspirin ingestion (75 mg/d for 7 days); wells B—samples after aspirin (ASA) administration. (B) Time courses for Fbg consumption. Levels of Fbg, determined by densitometry and comparison with standards, are presented as a function of time (seconds). Concentration of Fbg before (open circles) and after aspirin treatment (closed circles). Total amounts of Fbg in each sample before (open triangles) and after aspirin treatment (closed triangles). (C) A representative immunoblot of the subunit A of FXIII. Consecutive 30-second bleeding-time blood samples were separated on a 5% to 15% linear gradient SDS-PAGE gel under nonreducing conditions. FXIII and its active form were probed with a polyclonal antibody specific for both the unactivated (FXIIIA) and activated (FXIIIAa) A subunit. Wells A—blood samples taken before aspirin ingestion (75 mg/d for 7 days); wells B—samples after aspirin (ASA) administration. (D) Kinetics of FXIII activation in bleeding-time blood. FXIIIA concentration, determined by densitometry and comparison with standards, is presented as a function of time (seconds). Concentration of FXIIIA before (open circles) and after aspirin treatment (closed circles). Values are plotted as means ± SEM.
Factor XIII/XIIIa
The Western immunoblot seen in Figure 5C displays the disappearance over time of a Mr = 80 000 band that migrated identically with the purified subunit A of FXIII (FXIIIA). This fragment was not detected, on average, after 150 seconds of bleeding (range, 90-210 seconds). The activated form of subunit A (FXIIIAa) has an Mr = 74 000 and appears at 120 seconds (lanes 4A-7A) with its concentration increasing gradually. Comparison with standard amounts of FXIIIA gives approximately 86 ± 5 nM prior to aspirin ingestion. Densitometric analysis showed a rapid decrease of FXIIIA concentration in bleeding-time blood (0.784 ± 0.04 nM per second at maximum) to below 10% of the initial value (Figure 5D). After treatment, the concentration of FXIIIA was estimated approximately at 83 ± 3 nM and did not differ from the pretreatment value. Aspirin administration delayed FXIII activation by approximately 60 seconds (lane 5B, P = .031) and decreased the maximum rate of disappearance of FXIIIA, determined by densitometry of immunoblots, to 0.56 ± 0.05 nM per second (P = .033). The rate of increase in total amounts of FXIII also decreased significantly after aspirin ingestion (data not shown). A correlation between the detection time of α-thrombin B chain and FXIIIAa was significant (r = 0.72; P = .015). This corresponds to a rapid generation of α-thrombin B chain, the major activator of FXIII in vivo.30 The rates of FXIII activation and α-thrombin formation were also significantly correlated (r = 0.49;P = .024).
Discussion
The model of microvascular injury presented here provides insights into the complex coagulant reactions following tissue damage with attendant disruption of blood vessels, which ultimately lead to formation of the hemostatic plug and cessation of bleeding. Morphologic studies show that the hemostatic plugs, formed in wounds at the end of small arterioles and venules, consist of aggregated platelets surrounded by fibrin, present both in central areas and abundantly at the periphery close to collagen fibers.34 Thrombin formation via the extrinsic pathway of the coagulation system, as evidenced by a rapid fibrin formation,34 occurs as early as 30 seconds after injury. Therefore, this flowing blood system is a valuable tool to evaluate a series of events resulting in thrombin formation at the site of injury. Previous studies on blood coagulation at sites of hemostatic plug formation, however, have been limited to measurements of FPA, TAT, or F1.2 by commercially available ELISAs.18-20 In the present study, we have extended these observations to the determination of the kinetics of prothrombin, FV, and FXIII activation as well as Fbg consumption and inactivation of FVa using quantitative immunochemical techniques.
The principal conclusions from our analyses, performed in bleeding-time blood, are the following: (1) Prothrombin is rapidly and almost completely removed at 3 to 4 minutes of bleeding. (2) Thrombin B chain and prethrombin 2 appear at 60 to 120 seconds of bleeding and are produced in similar amounts. Their maximum concentrations reach approximately 35 to 40 nM at the end of bleeding. (3) Activation of FV is incomplete and at the end of bleeding FV level is about 75% of its initial concentration. FVa heavy chain is detectable at 120 seconds after vascular injury and appears somewhat faster than that of the light chain. (4) FVa inactivation products associated with the APC-mediated proteolysis of FVa heavy chain are observed simultaneously with the generation of FVa heavy chain. The products associated with the inactivation of FVa through thrombin and endothelial cells are also observed. (5) The maximum prothrombinase concentration (about 22 pM) is achieved by 150 seconds and is limited by the factor Xa concentration. (6) Fbg is removed very rapidly from the bleeding-time blood and is below detection limits by 3 minutes of bleeding. (7) Significant activation of FXIII is observed by 120 seconds. The extent of the activation process is correlated with α-thrombin B-chain generation.
Because the same antibodies have been used in the current bleeding-time blood model and the whole-blood system, described by Rand and colleagues,13 comparative evaluation of both models appears to be helpful and validated. In the present model, the amount of any protein in a volume unit downstream is determined by 3 factors—the activity of the complex generating this component, the velocity of the flow, and initial concentrations of the proteins studied. Given the fact that the blood flow is not constant, the increase in the blood flow could falsely lower levels of all the parameters measured. Therefore, we calculated the absolute amounts of any product formed within a 30-second time interval to make the analysis independent of the volume of blood samples. The total yields of a product divided by a volume unit (microliter of the blood shed) showed a similar behavior to those observed in the analyses based on concentrations (data not shown). It should be stressed that in the in vitro whole-blood system TF at a concentration of 12.5 to 25 pM is present in phospholipid vesicles, dispersed homogeneously in the reaction medium,13 which is in stark contrast to injured tissues as an abundant source of TF, though actual amounts of the TF available at the site of microvascular injury has not been quantitated.
There are some interesting similarities and differences between our model and that of Rand and colleages,13 which deserve special comments. At the site of microvascular injury, we detect prethrombin 2 (Figure 2A), a prothrombin activation product, which has been observed in the purified systems35 and in experiments in blood or plasma in vitro.13,36,37 The concentrations of thrombin B chain and prethrombin 2, observed at the end of bleeding (on average, 5 minutes), are almost identical to those found during clotting of whole blood at 10 minutes on addition of relipidated TF.13 Enhanced thrombin generation could be attributed to the potentiating effect of fibrin formed quickly at sites of microvascular injury.38
The amounts of α-thrombin B chain exceed the TAT formed by 50% to 80% at all time intervals in bleeding-time blood. In contrast, during the clotting of whole-blood in vitro both variables were similar.13 Another striking observation is that prothrombinase reaches its maximum (22 pM) at 150 seconds and fell to 9 pM before cessation of bleeding. In the whole-blood model,13 prothrombinase is increasing constantly within the first 5 minutes of clotting to approximately 150 pM. In both models, fibrinogen depletion occurred at prothrombinase concentrations in the range of 10 to 20 pM and a thrombin B-chain concentration of approximately 10 nM.
Activation of FV is faster but incomplete at sites of hemostatic plug formation when compared to the whole-blood model.13 As evidenced by a positive correlation between concentrations of FVa and α-thrombin B chain, a rapid FV activation at sites of the injury reflects a rapid generation of thrombin. More interestingly, the rapid appearance of FVa light chain is almost simultaneous to that of the heavy chain (Figure 4A,B), whereas in the whole blood model a delay in the appearance of FVa light chain is markedly longer.13This observation is in part caused by the more rapid consumption of the heavy chain by APC. However, formation of FVa light chain appears to be the limiting component of FV activation in both models.
Comparing FVa inactivation by APC in the current system and the whole-blood model,13 we have found that release of APC-mediated FVa inactivation products is much faster at the site of microvascular injury (Figure 4D). Lower concentrations of thrombomodulin, derived mostly from platelets,39 most likely account for a slow inactivation of FVa by APC in the whole-blood model.13 In the microvasculature, the protein C pathway plays a key role in anticoagulant mechanisms, due to high concentrations of thrombomodulin on the endothelial cells relative to the small volumes of blood.7 Our study convincingly supports this conclusion. It is of note that despite concomitant FVa proteolysis, amounts of FVa increase significantly in the consecutive blood samples, resulting in a rapid thrombin and finally fibrin formation. Furthermore, molar concentrations of FVa heavy chain are underestimated because APC-mediated cleavage of this chain lowers amounts of FVa detected by immunoblotting.
We identified the Mr = 97 000 fragment of FVa heavy chain at sites of microvascular injury (Figure 4E). Its mobility was identical to the product observed by Hockin and colleagues33 in the thrombin inactivation of FVa on HUVECs. Because significant amounts of this fragment were observed only at α-thrombin B-chain concentrations exceeding 35 nM, our data confirmed the hypothesis that the Mr = 97 000 peptide is released only in the presence of significant amounts of thrombin.33 The relevance of the 97 000 fragment formation in vivo awaits elucidation.
The bleeding-time blood model is also characterized by a rapid consumption of Fbg and prothrombin from blood collected from skin wounds (Figures 3A and 5A) when compared to the situation in the whole-blood model.13 Apart from Fbg cleavage by thrombin and activation of prothrombin, other mechanisms might contribute to the relatively high rates of both processes. It has been reported that Fbg can bind to hyaluronic acid,40 platelet thrombospondin,41 and endothelial cells via a process, which is enhanced by thrombin produced in loco,42 while prothrombin can attach to the subendothelium and platelets via β3 integrins.43
Collectively, the differences reported here provide evidence for the concept that kinetics of coagulation reactions under flow conditions cannot be extrapolated from static, closed systems. Moreover, cells of the vessel walls at the site of microvascular injury, as well as activated platelets and blood cells, provide membranes necessary for optimal function of most components of coagulation reactions known to be surface dependent4 and consequently, rates of most reactions are relatively high. The model, used in the current study, has some limitations: (1) a possible contact activation in test tubes is not eliminated, unlike in the whole-blood model,13 in which this pathway is suppressed by corn trypsin inhibitor, a selective inhibitor of FXIIa44; (2) our study focuses on early coagulant events (up to 5-7 minutes) in vessels less than 25 μm in diameter and therefore, later stages of blood coagulation, along with a process triggered by an injury of diseased large arteries, escape evaluation; (3) the bleeding-time technique used here requires expertise to achieve an appropriate reproducibility of results; and (4) volumes of the blood samples collected from skin wounds show a distinct interindividual variability. Nevertheless the present flowing blood model is a valuable tool in the in vivo coagulation studies.
In the current study, we demonstrated that thrombin generation is impaired by low-dose aspirin given for 7 days in healthy subjects. This finding is in keeping with most,15,23,25,26 though not all previous studies.24 In contact pathway-inhibited whole-blood coagulation studied in vitro, high-dose aspirin administered in vivo had no effect on exclusively TF-induced coagulation.45 This suggests that the aspirin effect in the present model may be tissue- or shear-dependent. In addition, our results provide the first evidence that at the site of vascular injury FV and FXIII activation, along with the removal of Fbg from the blood shed, are delayed, most likely as a consequence of impaired thrombin generation following aspirin ingestion.
The mode of action of aspirin on hemostasis has not been fully elucidated. It is well known that aspirin inhibits platelet function by irreversible acetylation of a serine at position 529 in the platelet cyclooxygenase.21 This, however, does not explain antithrombin properties of this drug,12,15,23-25 which appear to be related to platelet reactivity,46 through possible alterations in the exposure of negatively charged phospholipids on platelet surfaces47 or in membrane fluidity.48 It has also been shown that aspirin is able to acetylate some proteins, which are involved in the coagulation reactions, such as ATIII49 and Fbg.50 The existence of such phenomena has not been convincingly demonstrated in vivo. Another intriguing possibility results from observations showing that aspirin impairs activation of platelet glycoprotein GPIIb-IIIa.51 Studies on various GPIIb-IIIa receptor antagonists indicate that inhibition of this Fbg receptor leads to a significant suppression of thrombin generation.52 Recently it has been reported that these agents reduce platelet surface FV/Va binding and phosphatydylserine expression in whole blood taken from healthy volunteers.53
Because aspirin prolonged the lag phase of thrombin generation and the FVIIa-TF complex determines the duration of the lag phase in reconstituted systems,54 one might speculate that aspirin down-regulates TF expression. In fact, aspirin has been reported to inhibit synthesis of TF in human monocytes55 and to reduce TF expression in human atherosclerotic plaques.56 In addition, we found that aspirin decreases the maximum rate of thrombin generation in the propagation phase. This suggests that aspirin may affect other coagulant reactions, possibly via altered functions of the protein C system, regulating the propagation phase. Further studies are warranted to evaluate aspirin's impact on different stages of the TF-initiated blood coagulation.
Taken together, our results provide convincing evidence for the utility of the model of microvascular injury in the qualitative and quantitative analysis of the TF-initiated coagulation process in vivo. The present study extends the work of others showing a very rapid activation of prothrombin, FV, FXIII, and Fbg conversion to fibrin, along with FVa inactivation, which can be mediated by APC and other processes. The current model also enables monitoring of drug-induced alterations in the coagulation cascade. We have demonstrated a significant decrease in the rates of the activation of prothrombin, FV, FXIII, and Fbg cleavage following administration of low-dose aspirin. However, it remains to be clarified whether aspirin exerts similar effects on coagulation at sites of the injury in patients with CAD and if higher doses of aspirin produce more pronounced anticoagulant alterations. This issue is presently under investigation.
We thank Jan Brozek, Jed Pauls, and Grzegorz Guzik for their expert technical assistance, and Marek Grzywacz for performing ELISAs.
Supported by National Institutes of Health (NIH) grant HL-46703 (to K.G.M.) and NIH training grant T32 HL-07594 (to K.B. and K.G.M.). A.U. is a recipient of a Fulbright Fellowship.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Kenneth G. Mann, University of Vermont, Department of Biochemistry, Given Bldg, Rm E407, Burlington, VT 05405; e-mail:kmann@zoo.uvm.edu.