Definitive hematopoietic stem cells arise in the aorta–gonad–mesonephros (AGM) region from hemangioblasts, common precursors for hematopoietic and endothelial cells. Previously, we showed that multipotential hematopoietic progenitors and endothelial cells were massively produced in primary culture of the AGM region in the presence of oncostatin M. Here we describe a role for macrophage–colony-stimulating factor (M-CSF) in the development of hematopoietic and endothelial cells in AGM culture. The number of hematopoietic progenitors including multipotential cells was significantly increased in the AGM culture of op/opembryos. The addition of M-CSF to op/op AGM culture decreased colony-forming unit (CFU)-GEMM, granulocyte macrophage–CFU, and erythroid–CFU, but it increased CFU-M. On the other hand, the number of cells expressing endothelial markers, vascular endothelial-cadherin, intercellular adhesion molecule 2, and Flk-1 was reduced in op/op AGM culture. The M-CSF receptor was expressed in PCLP1+CD45− cells, the precursors of endothelial cells, and M-CSF up-regulated the expression of more mature endothelial cell markers—VCAM-1, PECAM-1, and E-selectin—in PCLP1+CD45− cells. These results suggest that M-CSF modulates the development of hematopoiesis by stimulating the differentiation of PCLP-1+CD45− cells to endothelial cells in the AGM region.
Introduction
Hematopoiesis is initiated sequentially in different tissues during development. Primitive hematopoiesis occurs in the yolk sac at mouse embryonic day 7.5 (E7.5), and definitive hematopoiesis starts in the embryonic para-aortic splanchnopleural region (pSp) at E8.5.1 Primitive hematopoiesis is characterized by the production of fetal erythrocytes with the nucleus and the lack of lymphocytes and myeloid cells except for macrophages. Definitive hematopoiesis generates enucleated erythrocytes, various kinds of myeloid and lymphoid cells, and long-term reconstituting hematopoietic stem cells (LTR-HSCs). Although lymphocytes are found in the E8.5 pSp region,2 LTR-HSCs first appear at E10.5 in the aorta–gonad–mesonephros (AGM) region.3 4 Thereafter hematopoiesis takes place in the fetal liver until bone marrow is formed.
Extensive studies on the early development of chick embryos indicate that hematopoietic cells and endothelial cells arise from the common precursor termed hemangioblast.5-7 A series of the grafting experiments using chicks and quails demonstrated that the splanchnopleural mesoderm generates hematopoietic cells and endothelium.8 Cell marking studies using chick embryos indicated that intra-aortic hematopoietic clusters are derived from endothelium and migrate to the para-aortic region.9,10CD34+ cells in the dorsal aorta and vitelline artery of human embryos were capable of generating hematopoietic cells in vitro,11 and LTR-HSC activity was detected in the dorsal aorta at E11.12 Disruption of the Cbfa2 (also known as AML-1, Runx-1) gene in mice resulted in the lack of definitive hematopoiesis in fetal liver13 and in the loss of hematopoietic clusters in the dorsal aorta.14 These studies indicate that endothelial cells in the dorsal aorta generate hematopoietic cells.
Hematogenic activity is regulated by cytokines such as vascular endothelial growth factor (VEGF). In the splanchnopleural mesoderm, cells expressing VEGF receptor 2 (VEGF-R2) were shown to form hematopoietic and endothelial colonies.15 Similarly, mouse cells expressing Flk-1 (mouse counterpart of VEGF-R2) and vascular endothelial cadherin (VE-cadherin) in the yolk sac and the pSp region at E9.5 mouse embryos gave rise to lymphohematopoietic cells in vitro.16 Flk-1 knockout mice exhibited severe defects in hematopoiesis and vasculogenesis in the yolk sac,17,18 and a similar phenotype was found by gene disruption of the SCL/Tal-1 transcription factor.19-21 In addition, recent studies on the in vitro differentiation of embryonic stem cells also indicate that hematopoietic cells and endothelial cells are derived from the same origin in the presence of VEGF.22-24 Taken together, these studies establish that VEGF plays a key role in the development of hematopoietic and endothelial cells.
To investigate the mechanisms underlying the development of hematopoietic and endothelial cells in the AGM region, we previously established a primary culture system of the AGM region.25In this culture, hematopoietic cells and endothelial cells are massively produced in the presence of stem cell factor (SCF), basic fibroblast growth factor (bFGF), and oncostatin M (OSM), an interleukin (IL)-6 family cytokine. Numerous endothelial-like cell clusters are formed in AGM culture, and hematopoietic cells are generated from endothelial-like cell clusters, indicating that the endothelial-like cell clusters contain hemangioblasts. Using a hemangioblastlike cell line derived from AGM culture, we identified podocalyxinlike protein 1 (PCLP1) as a marker for endothelial precursors.26PCLP1+CD45− cells were shown to differentiate to endothelial cells in the presence of OSM, VEGF, and OP9 stromal cells and were also capable of generating LTR-HSCs, whereas hemangioblasts remain to be identified.
Among various cytokines that affect hematopoiesis, macrophage–colony-stimulating factor (M-CSF) was initially characterized as a cytokine that supports the survival, proliferation, and differentiation of macrophages. M-CSF was also shown to affect osteoclasts,27 neural cells,28 uterine and placental cells,29 and smooth muscle cells.30As osteoclasts are derived from monocytes and M-CSF induces their differentiation, M-CSF–deficient mice (op/op) exhibit osteopetrosis.27 We found that the number of immature hematopoietic progenitors in the AGM culture of op/op mice was significantly larger than that of wild-type AGM culture and that the addition of M-CSF to op/op AGM culture reduced the number of immature progenitors. We present evidence that M-CSF modulates the development of hematopoiesis in AGM by promoting the differentiation of PCLP-1+CD45− cells to mature endothelial cells.
Materials and methods
Primary culture of AGM
AGM tissues removed from embryos were washed once with 0.25% trypsin in HEPES-buffered saline (Gibco-BRL, Gaithersburg, MD) and incubated at 37°C for 9 minutes in fresh trypsin solution. Trypsin was then removed, and the AGM cells in Dulbecco modified Eagle medium containing 15% heat-inactivated fetal calf serum were passed through a micropipette to make a single-cell suspension. These cells were plated onto a gelatin-coated dish at a density of 1 embryo-derived cell per well of a 6-well plate (Costar, Cambridge, MA). The number of cells recovered from a single AGM tissue was approximately 2 × 105. Cytokines added in culture were SCF (100 ng/mL) (provided by Kirin Brewery, Tokyo, Japan), bFGF (Gibco-BRL, Gaithersburg, MD) (1 ng/mL), and OSM (10 ng/mL). Cells were incubated at 37°C in 5% CO2 for various periods of time and were used for further analysis. M-CSF (R&D Systems, Minneapolis, MN) at 10 ng/mL was added to op/op AGM culture in some experiments, as described below.
Expression of hematopoietic cytokines in AGM culture
On the 10th day of AGM culture, adherent cells were harvested and treated with the Trizol reagent (Gibco-BRL, Gaithersburg, MD) for the preparation of total RNA. A first-strand cDNA synthesis kit (Pharmacia Biotech) was used to prepare ssDNA from total RNA pretreated with RNase-free DNase I (Gibco-BRL). Primers sequences used were: OSM sense, TCCGCCTCCAAAACCTGAACA, antisense, ATGGTATCCCCAGAGAAAGC; LIF sense, GGAGTCCAGCCCATAATGAAGGTC, antisense, GGCCTGGACCACCACACTTATGAC; IL-6 sense, GATAGTCAATTCCAGAAACCGCTA, antisense, TACTCCAGGTAGCTATGGTACTCC; G-CSF sense, GACGGCTCGCCTTGCTCTGCACCA, antisense, ACCTGGCTGCCACTGTTTCTTTAGG; VEGF-B sense, TCCGTCGCCTGCTGCTTGTT, antisense, CAGCTTGTCACTTTCGCGGC; VEGF-C sense, AGAGGACCTCAGCCTCAC, antisense, AGTGCTAAGTGCTGTTCC; VEGF-D sense, CTAGTTTGGAGGAGTTGCTG, antisense, TCTGGGGTCTGAATGGATCT; M-CSF sense, GACAGGCCGTTGACAGAGGTGAACCC, antisense, ATAGAATCCTTTCTATACTGGCAGTTC; and c-fms sense, CACAGATAAAATTGGAGCCTA, antisense, TGGAAGTTCATGGTGGCCG. Polymerase chain reaction (PCR) was performed under the following conditions: denaturation at 94°C for 30 seconds, annealing at 55°C to 57°C for 45 seconds, and elongation at 72°C for 90 seconds by the GeneAmp PCR system 2400 (Perkin-Elmer, Norwalk, CT). Amplification was linear for 30 to 35 cycles of PCR reactions.
Northern blot analysis of Flt-1 in AGM culture
The expression of Flt-1 was detected by Northern blot analysis. Total RNA (10 μg) was electrophoresed on a 1.0% agarose gel, transferred to a positively charged nylon membrane, and hybridized with digoxin (DIG)-labeled Flt-1 probes. After overnight hybridization, the membrane was washed to remove nonspecific binding of the probe and was incubated with anti-DIG antibody. Hybridized fragments were detected according to the instructions supplied.
DiI-Ac-LDL incorporation assay
Cells prepared from the AGM region were cultured in 4-well chamber slides (Nalge Nunc International, Naperville, IL). After 8 days of culture, the adherent cells were incubated with 10 μg/mL 1,1′-dioctadecyl-3,3,3′,3′-tetoramethylindo-carbocyanine perchlorate acetylated low-density lipoprotein (DiI-Ac-LDL) in culture medium (Biomedical Technologies, Cambridge, MA) for 4 hours at 37°C. These cells were then fixed with 4% paraformaldehyde in phosphate-buffered saline for 15 minutes at room temperature. A confocal microscope with the appropriate filters was used to detect the emitted fluorescence.
Flow cytometry of adherent cells in AGM culture
Adherent cells in AGM culture were harvested with cell dissociation buffer (Gibco-BRL) and were filtered through a 70-μm nylon mesh (Cell Strainer; Falcon). Cells were incubated with mouse serum to prevent nonspecific binding of antibodies. Antibodies against Flk-1 and VE-cadherin were obtained from PharMingen (San Diego, CA). Anti–intercellular adhesion molecule 2 (ICAM-2) antibody was prepared in our laboratory (unpublished results, M. Tanaka and T. Hara, 1998), and an anti-PCLP1 antibody was described previously.26 Fluorescein isothiocyanate (FITC)–conjugated anti-rat immunoglobulin (Ig)G obtained from Pierce (Rockville, IL) was used as a secondary antibody, and FACSCalibur (Becton Dickinson) was used for flow cytometry. When AGM cells were cocultured with OP9 stromal cells, biotinylated antibodies against platelet endothelial cell adhesion molecule (PECAM)-1, vascular cell adhesion molecule (VCAM)-1, and E-selectin were used for flow cytometry after the blocking of nonspecific binding by anti-mouse CD16/CD32 antibody (PharMingen).
Colony-forming unit assays
Nonadherent cells in AGM culture were collected, and 1 × 104 cells were plated in a 35-mm plate with α-minimal essential medium containing 0.8% methylcellulose, 30% fetal calf serum, 1% deionized bovine serum albumin, 100 μM 2-mercaptoethanol, 10 ng/mL IL-3, 100 ng/mL IL-6, and 2 U/mL erythropoietin (EPO). IL-3 was prepared as previously described.31 EPO was kindly provided by Kirin Brewery, and IL-6 was purchased from R&D Systems. Colonies were counted at the seventh day of culture.
Retroviral infection of AGM cells
Murine-soluble Flt-1 cDNA was provided by Dr M. Shibuya (Institute of Medical Science, University of Tokyo, Japan),32 and the coding region of sFlt-1 was inserted into the retroviral vector, pMIG, which carries internal ribosomal entry site–enhanced green fluorescent protein (EGFP) after the multicloning sites. BOSC23 packaging cells were transfected with the plasmid constructs using the lipofectamine plus reagent (Gibco-BRL), and the supernatants were harvested at 48 hours after transfection. AGM cells were cultured with the supernatants for 5 days and used for CFU-C assays and fluorescence-activated cell sorter (FACS) analysis.
Matrigel assay
Cell suspension was passed through a 40-μm cell strainer (Falcon) to remove cell aggregates, and 2.0 × 105 cells were replated onto Biocoat Matrigel basement membrane (Becton Dickinson) in a 6-well plate with 1% fetal calf serum. After 12 hours of culture, cell morphology was observed through a microscope.
Results
Suppression of hematopoiesis by M-CSF in AGM culture
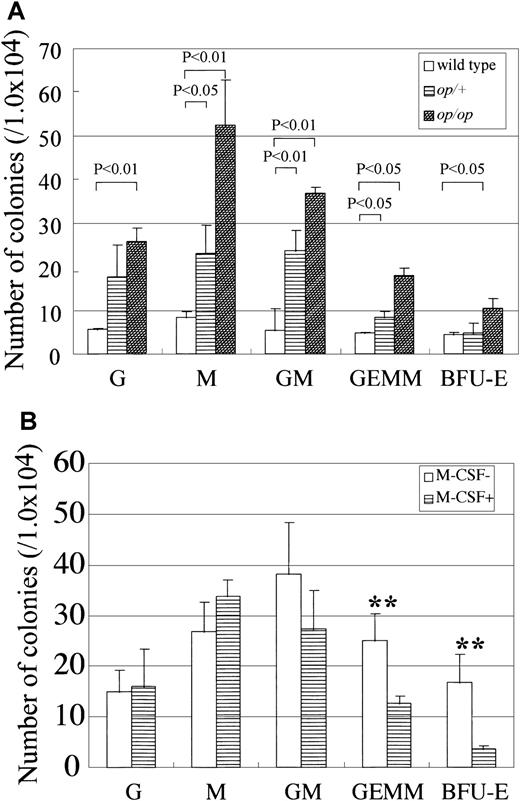
To investigate the hematopoietic activity in AGM cultures derived from wild-type and op/op mutant mice, nonadherent cells were harvested after 8 days of incubation, and colony-forming activity was evaluated (Figure 1A). Inspection of colonies indicated that the numbers of erythroid blast-forming units (BFU-E) and mixed CFU (CFU-Mix) were increased significantly inop/op AGM culture. As a result, more culture CFU (CFU-C) were found in op/op AGM culture than in wild-type AGM culture. Consistent with the increased number of granulocyte, erythrocyte, macrophage, and megakaryocyte (GEMM) in op/opAGM culture, we previously showed that more c-Kit+ cells were present in nonadherent cells in op/op AGM culture than in wild-type AGM culture.25 These results suggested that M-CSF deficiency altered the development of myeloid cells, leading to the accumulation of immature hematopoietic cells in AGM culture. We therefore added M-CSF to op/op AGM culture to test whether exogenous M-CSF could reduce colony formation. The number of hematopoietic cells grown in op/op AGM culture with M-CSF was slightly smaller than that in op/op AGM culture without M-CSF (data not shown). The numbers of BFU-E, CFU-GM, and CFU-GEMM were all reduced by the addition of M-CSF, whereas the number of CFU-M was increased by M-CSF (Figure 1B). These results suggest that M-CSF promotes hematopoietic differentiation at an early stage in AGM culture. Because M-CSF stimulates mainly the monocyte–macrophage lineage but not other hematopoietic lineages, it is likely that it affects hematopoiesis through monocyte–macrophages or nonhematopoietic cells.
Colony-forming activity of nonadherent cells inop/op AGM culture and the effect of exogenous M-CSF on the colony-forming activity in op/op AGM culture.
(A) The AGM region was removed from E11.5 embryos, and the cells were dissociated into a single-cell suspension by trypsin and incubated at 37°C for 10 days. Nonadherent cells in culture were harvested and plated in methylcellulose culture in the presence of IL-3, IL-6, and EPO. Colonies were counted on the seventh day of culture. Numbers in the figure indicate P values (by Student t test). (B) AGM cultures were prepared as above. Cells of op/op AGM culture were incubated in the presence of exogenous M-CSF on the first day of culture. Double asterisks indicate P < .05 (by Student t test). Each experiment was repeated 3 times, and typical results are presented in figures.
Colony-forming activity of nonadherent cells inop/op AGM culture and the effect of exogenous M-CSF on the colony-forming activity in op/op AGM culture.
(A) The AGM region was removed from E11.5 embryos, and the cells were dissociated into a single-cell suspension by trypsin and incubated at 37°C for 10 days. Nonadherent cells in culture were harvested and plated in methylcellulose culture in the presence of IL-3, IL-6, and EPO. Colonies were counted on the seventh day of culture. Numbers in the figure indicate P values (by Student t test). (B) AGM cultures were prepared as above. Cells of op/op AGM culture were incubated in the presence of exogenous M-CSF on the first day of culture. Double asterisks indicate P < .05 (by Student t test). Each experiment was repeated 3 times, and typical results are presented in figures.
Reduced production of cytokines in op/op AGM culture
We considered the possibility that the altered hematopoiesis inop/op AGM resulted from the production of hematopoietic cytokines in culture because we observed that various cytokines—including IL-6, OSM, LIF, G-CSF, and VEGF-B, -C, and -D—were expressed in AGM culture (see below). To test this possibility, adherent cells in the wild-type, op/+, andop/op AGM culture were harvested, and the expression of various cytokines was analyzed by reverse transcription (RT)-PCR. Although no significant differences were observed in the expression of VEGFs (Figure 2) or of IL-3, IL-7, and IL-11 (data not shown) between wild-type and op/op AGM cells, the expression of OSM, LIF, IL-6, and G-CSF was significantly reduced in op/op AGM cells (Figure 2). These results suggest that M-CSF and these cytokines may modulate the differentiation of hematopoietic cells. It is also possible that M-CSF regulates the expression of cytokines in other types of cells.
Expression of hematopoietic cytokines and VEGFs inop/op AGM culture.
Floating cells were washed out with phosphate-buffered saline (−), and adherent cells in AGM culture were harvested for synthesis of ssDNAs. PCR was performed to examine the expression of hematopoietic cytokine using a specific set of primers. Lane 1, wild-type; lane 2,op/+; lane 3, op/op; lane 4,op/op + M-CSF.
Expression of hematopoietic cytokines and VEGFs inop/op AGM culture.
Floating cells were washed out with phosphate-buffered saline (−), and adherent cells in AGM culture were harvested for synthesis of ssDNAs. PCR was performed to examine the expression of hematopoietic cytokine using a specific set of primers. Lane 1, wild-type; lane 2,op/+; lane 3, op/op; lane 4,op/op + M-CSF.
We then examined by RT-PCR whether M-CSF increased the expression of these cytokines in op/op AGM culture. We found that the expression of IL-6, OSM, LIF, and G-CSF was up-regulated inop/op AGM culture by M-CSF (Figure 2). These results suggest that the increased numbers of immature hematopoietic cells inop/op AGM culture can be partly explained by a modulation of cytokine production by M-CSF in AGM culture.
Alteration of endothelial cell differentiation in op/opAGM culture
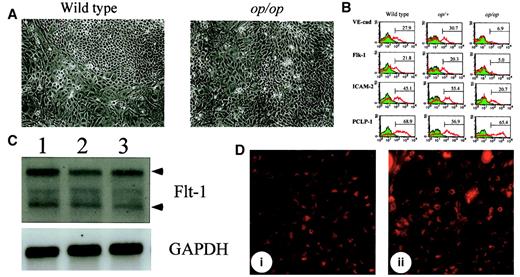
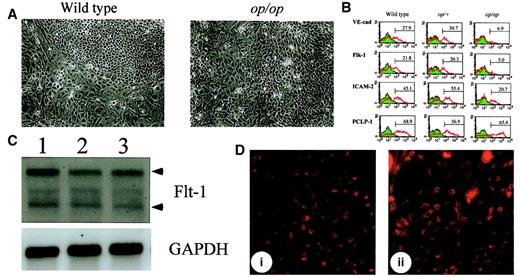
Although M-CSF may modulate the differentiation of committed hematopoietic progenitors by regulating cytokine production, it is also possible that it affects hematopoiesis at a much earlier stage. Given that hematopoietic progenitors are derived from hemangioblasts, the differentiation of endothelial cells might be altered by the lack of M-CSF in AGM culture. Because hematopoietic cells are generated from endothelial-like cell clusters in AGM culture, these cell clusters are thought to contain hemangioblasts. However, there was no significant difference in the number of such cell clusters between wild-type andop/op AGM cultures (data not shown). Therefore, we examined the expression of endothelial cell markers in adherent cells in AGM culture by flow cytometry, semiquantitative RT-PCR, and Northern blot analysis. There was no apparent difference in morphology and viability of cells between cultures (Figure 3A), indicating that the proliferation and viability of cells was not altered by the lack of M-CSF. The expression of PCLP-1, which was previously identified as a marker for immature endothelial cells,26 was not altered in op/op AGM cells (Figure 3B). Immunostaining of the AGM cells in plates showed that endothelial-like cell clusters in wild-type and op/op AGM culture were equally stained with anti-PCLP1 antibody (data not shown). PCLP1+CD45− cells from op/op AGM culture exhibited an endothelial cell morphology in gelatin-coated plates similar to that from wild-type AGM culture (data not shown). Moreover, the expression of Flt-1, a receptor for VEGF, was unchanged in the op/op AGM culture by Northern blot analysis (Figure3C), and the endothelial-like cells in op/op AGM culture incorporated DiI-Ac-LDL at a level similar to that in wild-type AGM culture (Figure 3D). These results suggest that the numbers of endothelial precursors were not different between op/op and wild-type AGM culture. In contrast, flow cytometric analysis revealed that the expressions of Flk-1, VE-cadherin, and ICAM-2 were decreased in nonadherent cells of op/op AGM culture compared with those in wild-type culture (Figure 3B). Furthermore, the expression of mature endothelial markers such as PECAM-1 and von Willebrand factor analyzed by semiquantitative RT-PCR was also decreased in theop/op AGM culture (data not shown). These results suggest that most of the endothelial-like cells in op/op AGM culture were already committed to the endothelial cell lineage but failed to differentiate to the stage that wild-type AGM cells differentiated.
Alterations of endothelial cell markers inop/op AGM culture.
(A) Morphology of adherent cells in wild-type and op/op AGM culture. AGM cultures were prepared from wild-type and op/op embryos, and photographs of the adherent cells were taken before FACS analysis was performed. Adherent cells proliferated in both cultures, and no significant difference in cell number was observed. Cells appeared healthy, and no dead cells were accumulated in either culture. (left) Wild-type. (right)op/op. (B) Expression of endothelial cell markers on adherent cells. Adherent cells were harvested from 3 independent cultures with the same genotype by using cell dissociation buffer. For FACS analysis, these cells were mixed and were incubated with mouse serum for blocking nonspecific binding of antibody. Antibodies against VE-cadherin, Flk-1, ICAM-2, and PCLP-1 were then added to the cells. FITC-conjugated anti-rat IgG was used to stain the cells. Numbers in figures indicate the percentage of positive cells. The experiment was performed twice, and generally the same results were obtained. Note that the expression of endothelial cell markers VE-cadherin, Flk-1, and ICAM-2 was reduced in op/op AGM culture, whereas the expression of a hemangioblast marker, PCLP-1, was unchanged. (C) Expression of Flt-1 in AGM culture. Adherent cells in AGM culture at the eighth day were used to prepare total RNA. RNA (10 μg) was electrophoresed and hybridized with DIG-labeled Flt-1 probe. Flt-1 expression was detected in each genotype. Arrowhead shows the specific band for the Flt-1 gene. Lane 1, wild type; lane 2,op/+; lane 3, op/op. (D) Incorporation of DiI-Ac-LDL. After 8 days of incubation, 10 μg/mL of DiI-Ac-LDL was added to the AGM culture and was incubated for 4 hours. Using a confocal microscope, the DiI-Ac-LDL–positive cells were detected in the clusters of endothelial cells of wild-type (i) and op/op(ii). i, wild-type; ii, op/op.
Alterations of endothelial cell markers inop/op AGM culture.
(A) Morphology of adherent cells in wild-type and op/op AGM culture. AGM cultures were prepared from wild-type and op/op embryos, and photographs of the adherent cells were taken before FACS analysis was performed. Adherent cells proliferated in both cultures, and no significant difference in cell number was observed. Cells appeared healthy, and no dead cells were accumulated in either culture. (left) Wild-type. (right)op/op. (B) Expression of endothelial cell markers on adherent cells. Adherent cells were harvested from 3 independent cultures with the same genotype by using cell dissociation buffer. For FACS analysis, these cells were mixed and were incubated with mouse serum for blocking nonspecific binding of antibody. Antibodies against VE-cadherin, Flk-1, ICAM-2, and PCLP-1 were then added to the cells. FITC-conjugated anti-rat IgG was used to stain the cells. Numbers in figures indicate the percentage of positive cells. The experiment was performed twice, and generally the same results were obtained. Note that the expression of endothelial cell markers VE-cadherin, Flk-1, and ICAM-2 was reduced in op/op AGM culture, whereas the expression of a hemangioblast marker, PCLP-1, was unchanged. (C) Expression of Flt-1 in AGM culture. Adherent cells in AGM culture at the eighth day were used to prepare total RNA. RNA (10 μg) was electrophoresed and hybridized with DIG-labeled Flt-1 probe. Flt-1 expression was detected in each genotype. Arrowhead shows the specific band for the Flt-1 gene. Lane 1, wild type; lane 2,op/+; lane 3, op/op. (D) Incorporation of DiI-Ac-LDL. After 8 days of incubation, 10 μg/mL of DiI-Ac-LDL was added to the AGM culture and was incubated for 4 hours. Using a confocal microscope, the DiI-Ac-LDL–positive cells were detected in the clusters of endothelial cells of wild-type (i) and op/op(ii). i, wild-type; ii, op/op.
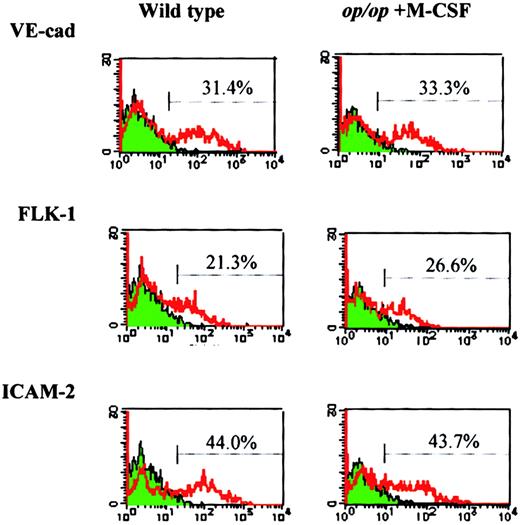
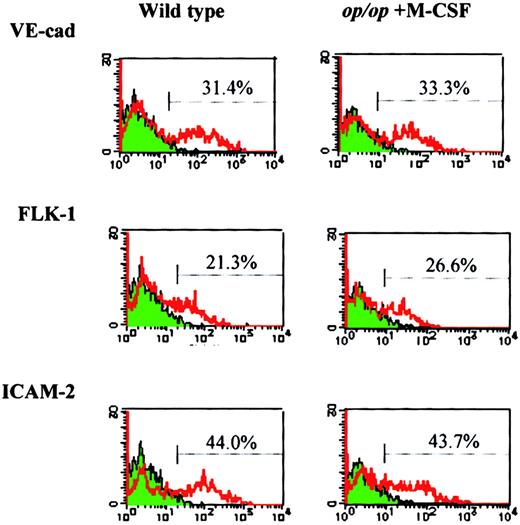
To investigate the role of M-CSF in endothelial differentiation, M-CSF was added to op/op AGM culture, and the expression of endothelial markers was examined after 8 days of incubation in the presence or absence of M-CSF. The addition of M-CSF to op/opAGM culture increased the expression of Flk-1, VE-cadherin, and ICAM-2 to the levels found in wild-type AGM culture (Figure4), indicating that M-CSF is involved in the expression of these endothelial cell markers.
M-CSF restores expression of endothelial cell markers inop/op adherent cells.
AGM culture of op/op mouse was supplemented with M-CSF on the first day of incubation. On the eighth day of culture, adherent cells were harvested and analyzed by flow cytometry using the antibodies against endothelial cell markers, VE-cadherin, Flk-1, ICAM-2, and FITC-labeled secondary antibodies. Wild-type AGM culture without M-CSF was used as a control. Note that the expression of the endothelial cell markers in op/op AGM culture was restored to the level in wild-type AGM culture by the addition of M-CSF.
M-CSF restores expression of endothelial cell markers inop/op adherent cells.
AGM culture of op/op mouse was supplemented with M-CSF on the first day of incubation. On the eighth day of culture, adherent cells were harvested and analyzed by flow cytometry using the antibodies against endothelial cell markers, VE-cadherin, Flk-1, ICAM-2, and FITC-labeled secondary antibodies. Wild-type AGM culture without M-CSF was used as a control. Note that the expression of the endothelial cell markers in op/op AGM culture was restored to the level in wild-type AGM culture by the addition of M-CSF.
Role of VEGF for hematopoiesis in AGM culture
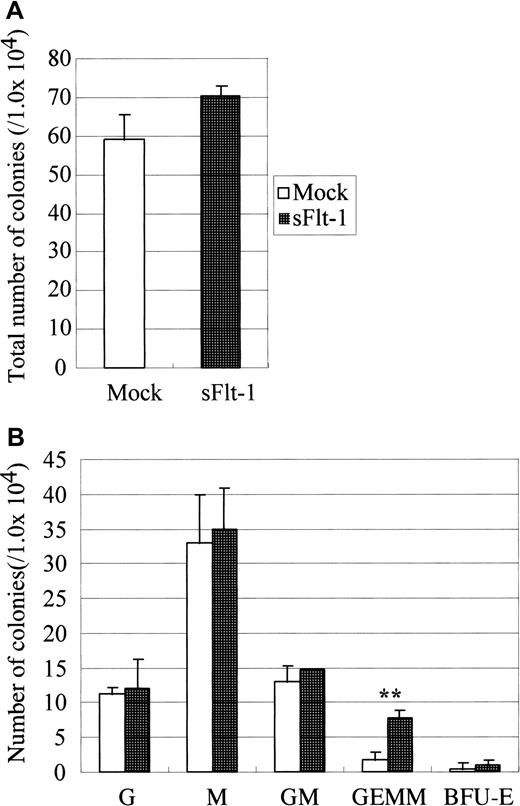
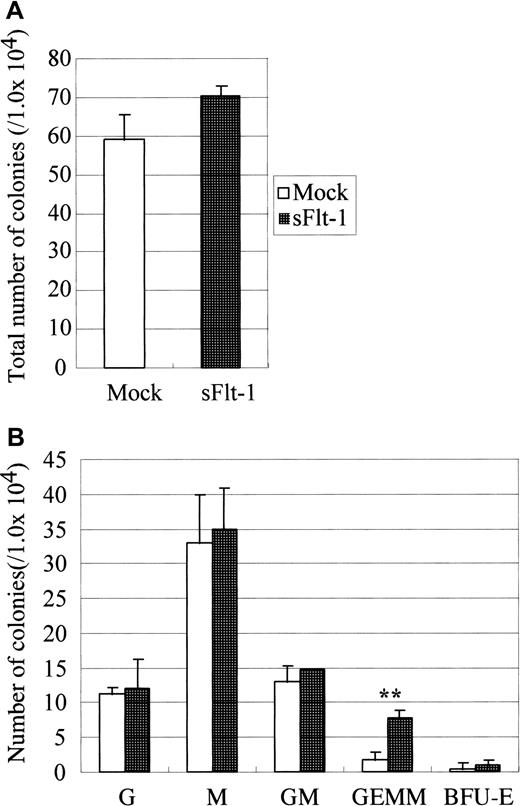
Because endothelial differentiation was halted and simultaneously the expression of Flk-1 (VEGF receptor 2) was reduced inop/op AGM culture, we examined the role of VEGF in the development of hematopoietic and endothelial cells. To inhibit endogenous VEGF in AGM culture, we used soluble Flt-1. Flt-1 is a tyrosine kinase receptor that binds VEGF-A and VEGF-C more strongly than Flk-1. However, because Flt-1 fails to induce proliferation signals, it functions as an antagonist of VEGF.33 It was demonstrated that a soluble form of Flt-1, sFlt-1, is produced in the human peripheral blood and inhibits VEGF signaling through Flk-1 by competing the VEGF binding.34 We used the retrovirus vector to overexpress sFlt-1 in AGM culture (Figure5). The number of immature hematopoietic progenitors produced in AGM culture infected with sFlt-1 virus was larger than that in culture with the empty vector, though the total number of colonies was unchanged.
Effect of soluble Flt-1 on hematopoiesis in AGM culture.
On the second day of culture, sFlt-1 virus was inoculated. On the next day, medium was changed to normal growth medium. Nonadherent cells were harvested after 10 days of incubation and were plated onto methylcellulose culture in the presence of IL-3, IL-6, and EPO. Note that sFlt-1 significantly increased the number of GEMM. Error bar shows standard deviation. Double asterisk indicates P < .01 (Student t test). This experiment was repeated 2 times, and generally the same results were obtained. Virus vectors used are Mock, pMIG vector, sFlt-1, pMIG-sFlt-1.
Effect of soluble Flt-1 on hematopoiesis in AGM culture.
On the second day of culture, sFlt-1 virus was inoculated. On the next day, medium was changed to normal growth medium. Nonadherent cells were harvested after 10 days of incubation and were plated onto methylcellulose culture in the presence of IL-3, IL-6, and EPO. Note that sFlt-1 significantly increased the number of GEMM. Error bar shows standard deviation. Double asterisk indicates P < .01 (Student t test). This experiment was repeated 2 times, and generally the same results were obtained. Virus vectors used are Mock, pMIG vector, sFlt-1, pMIG-sFlt-1.
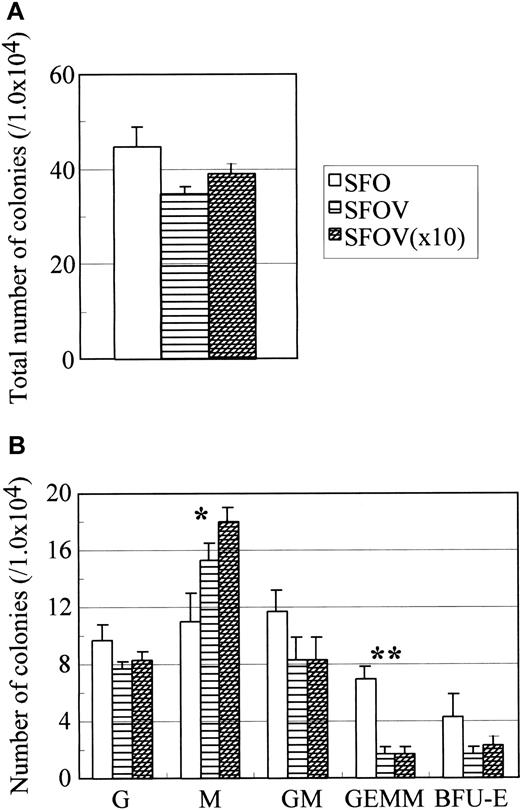
To further elucidate the role of VEGF in AGM hematopoiesis, we tested whether exogenous VEGF affects hematopoiesis. After 10 days of incubation of AGM cells in the presence or absence of VEGF-A, the colony-forming activity of nonadherent cells was examined (Figure6). The addition of VEGF-A reduced the number of colony-forming cells of immature progenitors but not CFU-M in wild-type AGM culture. Although the reason for the increase of CFU-M by VEGF-A is unknown, it was reported that VEGF induced the migration of monocytes and differentiation to macrophagelike cells from human cord blood CD34+ cells, suggesting that VEGF has the potential to stimulate the monocytic lineage.35 Previously we showed that adherent cells, which had incorporated DiI-Ac-LDL and formed clusters of endothelial-like cells, gave rise to DiI-Ac-LDL–positive hematopoietic cells in AGM culture.26 These results suggest that the production of hematopoietic progenitors from hemangioblasts or the differentiation of hematopoietic progenitors is negatively regulated by VEGF in AGM culture. Alternatively, VEGF may play a role in lineage commitment and differentiation of hematopoietic cells or colony-forming cells. In contrast, neither sFlt-1 nor VEGF had a significant effect on the expression of Flk-1 and VE-cadherin.
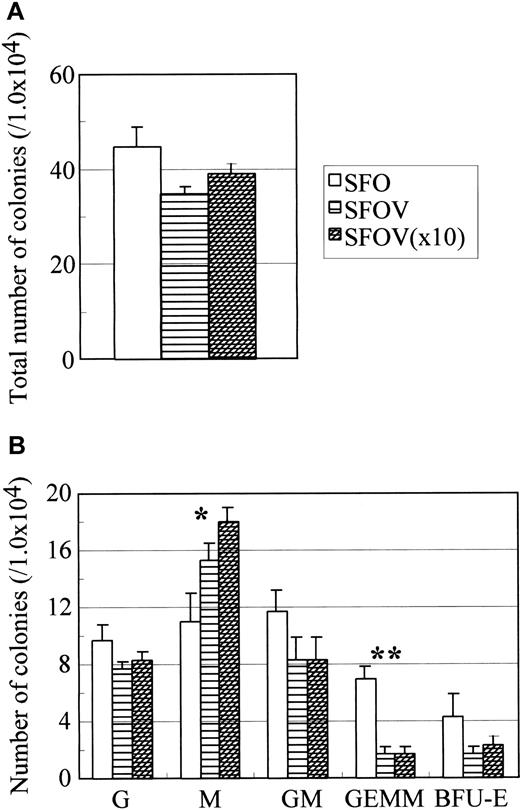
Effect of VEGF on hematopoiesis in AGM culture.
Cells in the AGM region of wild-type embryos were incubated in the presence or absence of various concentrations of VEGF for 10 days. Nonadherent cells were analyzed for colony-forming activity in the presence of IL-3, IL-6, and EPO. Colonies were enumerated on the seventh day of culture. Culture conditions: SFO—SCF, bFGF, and OSM; SFOV—SCF, bFGF, OSM, and 10 ng/mL VEGF; SFOV (×10)—SCF, bFGF, OSM, and 100 ng/mL VEGF. Note that M-CSF increases the number of CFU-M in a dose-dependent manner (indicated by asterisk) and reduces the number of CFU-GEMM (indicated by double asterisk). P < .01 (Student t test). Sizes of error bars, shown in GEMM, were 1.0, 0.6, and 0.6, respectively. P < .05 (Studentt test).
Effect of VEGF on hematopoiesis in AGM culture.
Cells in the AGM region of wild-type embryos were incubated in the presence or absence of various concentrations of VEGF for 10 days. Nonadherent cells were analyzed for colony-forming activity in the presence of IL-3, IL-6, and EPO. Colonies were enumerated on the seventh day of culture. Culture conditions: SFO—SCF, bFGF, and OSM; SFOV—SCF, bFGF, OSM, and 10 ng/mL VEGF; SFOV (×10)—SCF, bFGF, OSM, and 100 ng/mL VEGF. Note that M-CSF increases the number of CFU-M in a dose-dependent manner (indicated by asterisk) and reduces the number of CFU-GEMM (indicated by double asterisk). P < .01 (Student t test). Sizes of error bars, shown in GEMM, were 1.0, 0.6, and 0.6, respectively. P < .05 (Studentt test).
M-CSF promotes endothelial differentiation of PCLP-1+CD45− cells
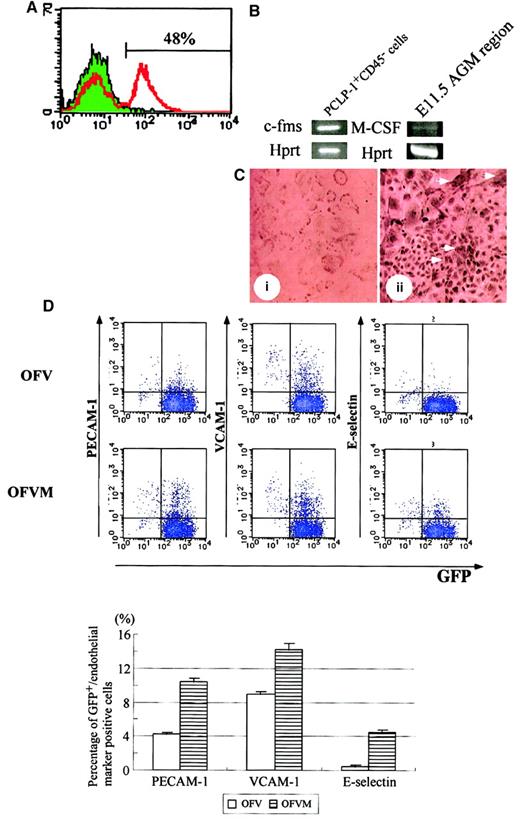
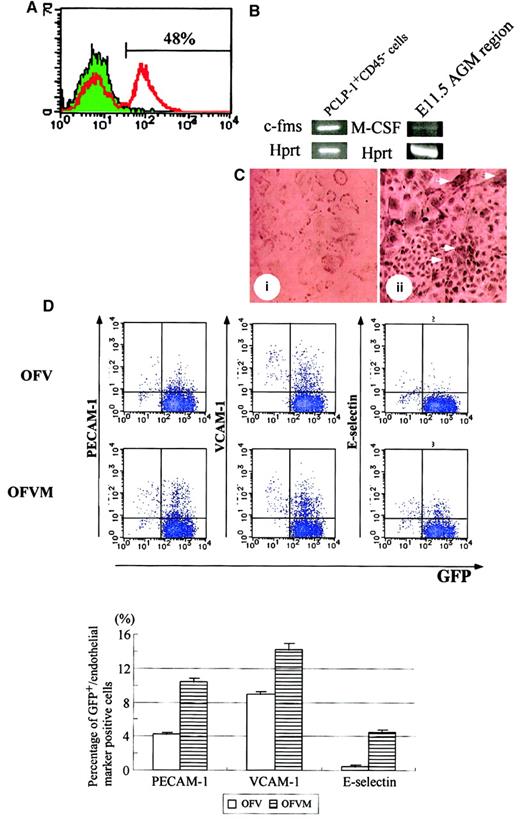
The results described above suggest that M-CSF affects the development of hematopoietic and endothelial cells. Given that we previously demonstrated that the PCLP-1+CD45−population in the AGM region gave rise to hematopoietic and endothelial cells in coculture with OP9 stromal cells, it is likely that M-CSF acts on PCLP1+CD45− cells. If this is the case, the M-CSF receptor encoded by the c-fms gene must be present on PCLP1+CD45− cells. To test the expression of the M-CSF receptor, adherent cells in AGM culture were analyzed by using anti–M-CSF receptor antibody, and 48% were found to express the M-CSF receptor (Figure 7A). We next isolated the PCLP-1+CD45− cells from AGM culture by flow cytometry and analyzed the expression ofc-fms by RT-PCR. The specific fragment of c-fmswas amplified by RT-PCR (Figure 7B), indicating that the M-CSF receptor was expressed on PCLP-1+CD45− cells in AGM culture. On the other hand, the expression of M-CSF was detected in the AGM region at 11.5 days postcoitum (Figure 7B) and AGM culture (data not shown). Finally, immunocytochemistry using the anti–M-CSF receptor antibody revealed positive signals within clusters of endothelial-like cells and within large and flattened cells surrounding the clusters, which looked like smooth muscle cells (Figure7C).
M-CSF induces the expression of mature endothelial cell markers in PCLP-1+CD45− cells.
(A) Expression of the M-CSF receptor in adherent cells in AGM culture. Adherent cells in AGM culture were harvested on the eighth day and were analyzed by FACS using anti–M-CSF receptor antibody with FITC-conjugated anti-rat IgG. Approximately 48% of cells in AGM culture expressed the M-CSF receptor. (B) After 10 days of incubation, adherent cells in AGM culture were harvested and sorted using PCLP-1 and CD45 antibodies. A set of specific primers for c-fms,M-CSF, and hprt were used for amplification of the DNA fragments. The expression of c-fms was detected in PCLP-1+CD45− cells, and the expression of M-CSF was detected in the AGM region at 11.5 dpc. (C) Immunostaining of the M-CSF receptor. Positive cells were detected within and around the clusters of endothelial cells (arrowhead). (D) PCLP-1+CD45− cells were isolated from the AGM region of the GFP transgenic mouse at E11.5 and were cocultured with OP9 stromal cells in the presence (OFVM) and the absence (OFV) of M-CSF. After 8 days of incubation, adherent cells were harvested and analyzed by FACS using antibodies against PECAM-1, VCAM-1, and E-selectin. Numbers in the figure indicate the percentage of cells positive (upper) and negative (lower) for GFP-positive cells. Results were summarized in the bottom panel. This experiment was performed 3 times, and generally the same results were obtained. P < .05 in PECAM-1, VCAM-1, and E-selectin. Culture conditions: OFV—OSM, bFGF, and VEGF; OFVM—OSM, bFGF, VEGF, and M-CSF.
M-CSF induces the expression of mature endothelial cell markers in PCLP-1+CD45− cells.
(A) Expression of the M-CSF receptor in adherent cells in AGM culture. Adherent cells in AGM culture were harvested on the eighth day and were analyzed by FACS using anti–M-CSF receptor antibody with FITC-conjugated anti-rat IgG. Approximately 48% of cells in AGM culture expressed the M-CSF receptor. (B) After 10 days of incubation, adherent cells in AGM culture were harvested and sorted using PCLP-1 and CD45 antibodies. A set of specific primers for c-fms,M-CSF, and hprt were used for amplification of the DNA fragments. The expression of c-fms was detected in PCLP-1+CD45− cells, and the expression of M-CSF was detected in the AGM region at 11.5 dpc. (C) Immunostaining of the M-CSF receptor. Positive cells were detected within and around the clusters of endothelial cells (arrowhead). (D) PCLP-1+CD45− cells were isolated from the AGM region of the GFP transgenic mouse at E11.5 and were cocultured with OP9 stromal cells in the presence (OFVM) and the absence (OFV) of M-CSF. After 8 days of incubation, adherent cells were harvested and analyzed by FACS using antibodies against PECAM-1, VCAM-1, and E-selectin. Numbers in the figure indicate the percentage of cells positive (upper) and negative (lower) for GFP-positive cells. Results were summarized in the bottom panel. This experiment was performed 3 times, and generally the same results were obtained. P < .05 in PECAM-1, VCAM-1, and E-selectin. Culture conditions: OFV—OSM, bFGF, and VEGF; OFVM—OSM, bFGF, VEGF, and M-CSF.
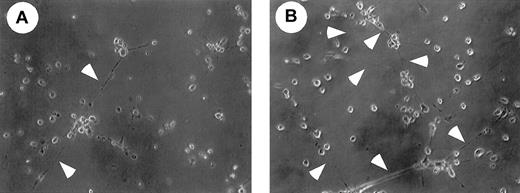
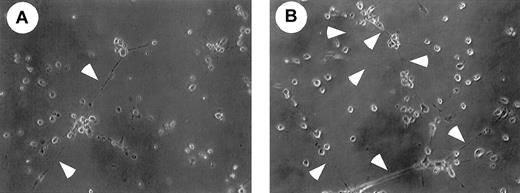
We then examined whether M-CSF had any effect on PCLP1+CD45− cells. PCLP-1+CD45− cells were isolated from E11.5 AGM of green fluorescence protein (GFP) transgenic mice and were cocultured with OP9 cells in the presence or absence of M-CSF. Transgenic mice expressed GFP ubiquitously by the β-actin promoter,36 and this mouse was used to distinguish the descendants of GFP+PCLP-1+CD45−from GFP− OP9 stromal cells. Because it has been shown that PECAM-1, VCAM-1, and E-selectin are expressed in mature endothelial cells, we examined the expression of these molecules in PCLP-1+CD45− cells. Expression levels of PECAM-1, VCAM, and E-selectin were higher in the PCLP-1+CD45− cells cultured with M-CSF than in those without M-CSF (Figure 7D). To investigate the differentiation of endothelial cells, PCLP1+CD45− cells cultured for 8 days with OP9 stromal cells in the presence or absence of M-CSF were replated in Matrigel culture. After 12 hours of culture, a tubelike morphology, a typical structure of mature endothelial cells in Matrigel, was more frequently observed in the cells cultured with M-CSF (Figure 8). These results suggest that M-CSF enhances, though modestly, the differentiation of PCLP-1+CD45− cells in the AGM region.
Matrigel analysis of PCLP-1+CD45− cells incubated with or without M-CSF.
After coculture of PCLP-1+CD45− cells with or without M-CSF in the presence of bFGF and OSM, adherent cells were replated onto Matrigel, and the photograph was taken after 12 hours of incubation. Arrowheads indicate the sprouting of cell cytoplasm. Note that cellular networks are more frequently found in the culture of PCLP-1+CD45− cells that were incubated with OP9 stromal cells in the presence of M-CSF.
Matrigel analysis of PCLP-1+CD45− cells incubated with or without M-CSF.
After coculture of PCLP-1+CD45− cells with or without M-CSF in the presence of bFGF and OSM, adherent cells were replated onto Matrigel, and the photograph was taken after 12 hours of incubation. Arrowheads indicate the sprouting of cell cytoplasm. Note that cellular networks are more frequently found in the culture of PCLP-1+CD45− cells that were incubated with OP9 stromal cells in the presence of M-CSF.
Discussion
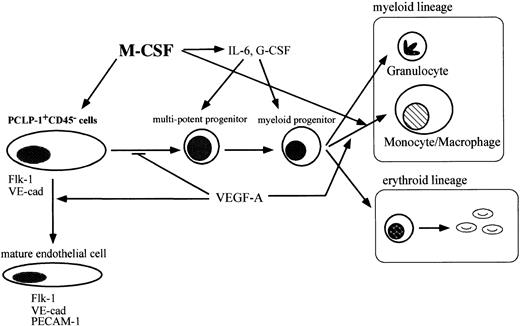
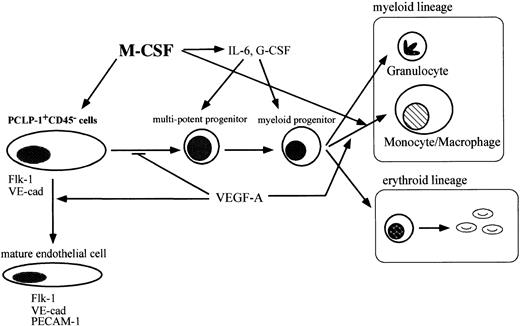
In this report, we present evidence that M-CSF has the potential to modulate the development of hematopoietic and endothelial cells in the AGM region. A lack of M-CSF results in the accumulation of immature hematopoietic progenitors in AGM culture, as evidenced by increased numbers of CFU-GEMM, CFU-GM, and BFU-E. Consistent with this, the addition of M-CSF to op/op AGM culture decreased the number of colony-forming cells. M-CSF also affects the differentiation of endothelial cells, as the expression of endothelial cell markers, such as Flk-1, VE-cadherin, and ICAM-2, was decreased in op/opendothelial-like cells in AGM culture. The results in this study can be summarized as shown in Figure9.
M-CSF affects the differentiation of hematopoietic cells and endothelial cells in AGM culture.
M-CSF stimulates the differentiation of macrophages. VEGF regulates hematopoiesis negatively by enhancing the differentiation of endothelial cells from hemangioblasts in AGM culture. M-CSF induces the expression of hematopoietic cytokines from macrophages and other types of cells and also induces the expression of Flk-1 in hemangioblasts. Inop/op AGM culture, a lack of M-CSF leads to the reduction of the response of hemangioblasts to VEGF by decreasing the expression of Flk-1. Immature hematopoietic cells accumulate in op/opAGM culture.
M-CSF affects the differentiation of hematopoietic cells and endothelial cells in AGM culture.
M-CSF stimulates the differentiation of macrophages. VEGF regulates hematopoiesis negatively by enhancing the differentiation of endothelial cells from hemangioblasts in AGM culture. M-CSF induces the expression of hematopoietic cytokines from macrophages and other types of cells and also induces the expression of Flk-1 in hemangioblasts. Inop/op AGM culture, a lack of M-CSF leads to the reduction of the response of hemangioblasts to VEGF by decreasing the expression of Flk-1. Immature hematopoietic cells accumulate in op/opAGM culture.
Because M-CSF does not act directly on most hematopoietic cells, it is likely that its effects on CFU-GEMM, CFU-GM, and BFU-E are indirect. In addition, because BFU-E and CFU-GEMM numbers were increased inop/op AGM culture, M-CSF affects hematopoiesis at a very early stage. We found that the production of cytokines such as IL-6, G-CSF, and LIF was decreased in op/op AGM culture and was increased by the addition of M-CSF in op/op AGM culture, suggesting that these cytokines regulate hematopoiesis at an early stage. Although the production of these cytokines may not solely account for the development of hematopoietic cells in AGM culture, it likely contributes to hematopoiesis in the AGM region. Monocytes–macrophages are a major target of M-CSF and have the potential to produce various cytokines. M-CSF may modulate hematopoiesis by enhancing cytokine production through the stimulation of monocytes–macrophages. On the other hand, given that smooth muscle cells in the bone marrow are known to contribute to hematopoiesis and smooth muscle cells were also found at the periphery of endothelial-like cell clusters in AGM culture as α-smooth muscle actin-positive cells, they might also contribute to hematopoiesis. However, α-smooth muscle actin-positive cells were distributed inop/op AGM culture in a manner similar to that of wild-type AGM culture, suggesting that smooth muscle cells develop normally in op/op AGM culture (data not shown).
We show that the addition of M-CSF recovered the expression of Flk1, ICAM-2, and VE-cadherin in op/op AGM culture, suggesting that their expression also requires signaling through the M-CSF receptor in AGM culture. Promoter analysis revealed that binding sites for SCL, GATA, and Ets are present in the promoter regions of theflk-1 gene.37 The mutational analysis of theflk-1 promoters indicated that SCL, GATA, and Ets are required for the endothelial cell-specific expression.38In addition, it was reported that Ets-binding sites are present in the promoter region of the VE-cadherin gene39 and that the overexpression of Ets1 induces the expression of VE-cadherin.40 M-CSF was shown to activate Ets family proteins by phosphorylation,41,42 and it may induce the expression of Flk-1 and VE-cadherin in endothelial cells by regulating those transcription factors. Consistent with this idea, the expression of transcription factors SCL, GATA, and Ets was detected in the PCLP1+CD45− cells (data not shown). Therefore, the expression of Flk-1 and VE-cadherin might be induced by M-CSF through the activation of these transcription factors in AGM culture. Several lines of evidence indicate that VEGF is involved in the development of hematopoietic cells. In this study, we found that expression of the VEGF receptor, Flk-1, was decreased inop/op AGM culture and that the addition of VEGF to wild-type AGM culture resulted in a reduction of CFU-GEMM, whereas the inhibition of VEGF by overexpression of sFlt-1, an Flk-1 antagonist, in AGM culture increased CFU-GEMM. Hamada et al43 showed that the production of hematopoietic cells was reduced by the addition of VEGF in pSp explant culture with OP9 stromal cells. Based on our results, VEGF-A reduced the number of CFU-GEMM colonies but did not affect the total number of hematopoietic cells in culture. Because the nature of hematopoietic cells grown in pSp culture was not described, our results cannot be directly compared with theirs. The difference between these results might be attributed to the difference in the culture systems—ie, organ culture of pSp in the presence of OP9 cells versus culture of dissociated AGM cells in the absence of OP9 cells. It is also possible that the concentration of VEGF is important in affecting the development of hematopoietic cells. Adherent cells in AGM culture expressed Flk-1, and hematopoietic cells arose from clusters of endothelial-like cells. However, because the addition of VEGF-A in AGM culture did not change the number of Flk-1–positive cells, it is likely that VEGF affects the development of hematopoietic cells from hemangioblasts rather than the proliferation of Flk-1–positive cells in AGM culture, consistent with the observation in the avian system.15
It is known that op/op mice exhibit excessive bone formation, and this abnormality can be corrected by either M-CSF or VEGF.44-46 Toothless rats exhibit the same phenotype asop/op mice and show an abnormal microvasculature of the distal femoral chondro-osseous junction.47 Administration of M-CSF in toothless rats ameliorated not only excessive bone formation but also the defect of the microvasculature.47We showed that the addition of VEGF-A increased the number of CFU-M in wild-type AGM culture, indicating that VEGF affects the development of the monocytic lineage. We also observed that the M-CSF receptor was expressed in PCLP1+CD45− cells and that the expression of endothelial markers was decreased in op/op AGM culture and was increased by the addition of M-CSF. Moreover, M-CSF promoted the differentiation of PCLP-1+CD45−cells to mature endothelial cells modestly in the presence of OP9 stromal cells in vitro, as evidenced by increased expression of PECAM-1, VCAM-1, and E-selectin and by the Matrigel assay. These observations, together with the results in toothless rats, suggest that M-CSF plays a role in the normal development of monocytic cell lineages and, in part, in the normal development of endothelial cells.
Expression of Flk-1, VE-cadherin, and ICAM-2 was decreased inop/op nonadherent AGM cells and the reduction in Flk-1 expression in op/op mice may lead to a reduced response to VEGF, resulting in unfavorable conditions for endothelial differentiation. Although M-CSF likely stimulates differentiation of endothelial cells through the modulation of Flk-1 expression, it remains possible that M-CSF directly stimulates the differentiation of endothelial cells. Because M-CSF itself is a growth and differentiation factor for macrophages, the lack of M-CSF may lead to the accumulation of immature hematopoietic cells. These results suggest that the accumulation of immature hematopoietic cells in op/op AGM culture is caused by the block or delay of endothelial differentiation from hemangioblasts and the reduced differentiation of monocytes.
We thank Drs M. Shibuya (The University of Tokyo) and S. Hayashi (Tottori University) for providing us with sFlt-1 cDNA andop/+ mice, respectively.
Supported in part by Grants-in-Aid for Scientific Research and Special Coordination Funds from the Ministry of Education, Culture, Sports, Science, and Technology of Japan, a research grant from Core Research for Evolutionary Science and Technology, and a grant from the Organization for Pharmaceutical Safety and Research.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Atsushi Miyajima, Institute of Molecular and Cellular Biosciences, The University of Tokyo, 1-1-1 Yayoi, Bunkyo-ku, Tokyo 113-0032, Japan; e-mail: miyajima@ims.u-tokyo.ac.jp.