Abstract
In contrast to severe aplastic anemia (sAA), the appropriate management of patients with moderate pancytopenia is unclear. In this study, we examined the efficacy of a humanized monoclonal antibody recognizing interleukin-2 receptor (daclizumab), which has proven to be a successful immunosuppressive agent in solid organ and bone marrow transplantation. We treated 17 patients with moderate aplastic anemia (mAA) with 1 mg/kg every 2 weeks for 3 months. mAA was defined as depression of 2 of the 3 blood counts: absolute neutrophil count 1200/mm3 or less, platelet count 70 000/mm3 or less, hemoglobin level 8.5 g/dL or lower, and absolute reticulocyte count 60 000/mm3 or less. The primary end point of our protocol was a hematologic response in at least one affected peripheral blood value. Daclizumab had little toxicity. Six of the 16 (38%) evaluable patients responded to treatment. Two patients with previously chronic disease showed complete return of normal counts, which were sustained for more than 2 years following treatment. Four patients had single-lineage responses. Two previously transfusion-dependent patients became transfusion independent; one patient with many neutropenia-related infections had a normal neutrophil count following treatment. Daclizumab appears safe; its efficacy in this pilot protocol suggests that expanded study of this monoclonal antibody in immune-mediated bone marrow failure syndrome is warranted. (Blood. 2003; 102:3584-3586)
Introduction
Immunosuppression with antithymocyte globulin (ATG) and cyclosporine (CsA) has been extraordinarily successful in producing clinical responses and prolonging survival of patients with aplastic anemia (AA).1 Intensive immunosuppressive treatment, however, is associated with significant toxicity and requires hospitalization and considerable supportive care. Although such treatment is appropriate in patients with severe aplastic anemia (sAA; defined as absolute neutrophil count [ANC] ≤ 500/mm3, platelet count ≤ 20 000/mm3, and absolute reticulocyte count ≤ 60 000/mm3), who require urgent management, it may be argued that patients with moderate or nonsevere aplastic anemia (mAA) may be observed for prolonged periods of time without definitive treatment.2-4 However, failure to treat might subject the patient's bone marrow to continued immune attack and lead to progressive loss of progenitor cells over time, ultimately resulting in the development of severe disease.5 Patients with mAA received less aggressive modalities such as androgens and growth factors,6-14 but these treatments do not clearly address the immune pathology and may only result in improvement in cytopenias without sparing progenitor cells. mAA has been treated with CsA alone12,15 and although good response rates have been achieved, patients are required to take the drug for substantial periods and frequently have a relapse following its discontinuation. In addition, irreversible renal pathology is a risk of therapy.
Daclizumab is a genetically engineered human IgG1 incorporating the antigen-binding regions of a parent murine monoclonal antibody (mAb) that specifically recognizes the 55-kDa α chain of the heterotrimeric interleukin 2 receptor (IL-2R).16 IL-2R is present only on activated T cells and is required for their clonal expansion and continued viability; blockade of IL-2R results in decreased activation and proliferation of T-cell clones. The mechanism of action of anti-IL-2R mAb is believed to be similar to that of ATG. Daclizumab has had few toxicities when used to treat patients rejecting solid organ transplants. Thus, daclizumab is an attractive, less toxic alternative to ATG.
In this study we treated a group of patients with mAA with daclizumab, assessing its toxicity, efficacy, and biologic effects.
Study design
Patients
Eligible patients with mAA were entered into the study after obtaining informed consent according to protocol approved by the Institutional Review Board of the National, Heart, Lung and Blood Institute (Bethesda, MD). We treated all consecutive patients over the age of 7 years with mAA, defined according to the Consensus Conference on Treatment of Aplastic Anemia,2 by a hypocellular bone marrow (cellularity < 30%) and depression of at least 2 of 3 blood counts below the normal values: ANC 1200/mm3 or less, platelet count ≤ 70 000/mm3 or less, anemia with hemoglobin level 8.5 g/dL or less, and absolute reticulocyte count 60 000/mm3 or less. Patients with severe pancytopenia were excluded from the protocol. The average of 3 measurements of blood counts obtained within a 2-week period prior to enrollment into the study was used to assess study eligibility. Transfusion independence was not an exclusion criterion. For the purpose of this study, transfusion dependence was defined as requirements of at least 2 U red blood cells (RBCs) or 5 U platelets per month for a period of 2 months prior to enrollment. We excluded patients with a current diagnosis or past history of myelodysplastic syndrome, abnormal cytogenetics, Fanconi anemia, or other congenital forms of AA.
Patients with a history of initial failure to respond to ATG, antilymphocyte globulin (ALG), or another immunosuppressive agent were also excluded. Patients received 1 mg/kg daclizumab infused intravenously every other week for a total of 5 doses. Response was defined at 3 months by the criteria delineated in Table 1; the primary end point of this trial was a hematologic response in at least one affected peripheral blood parameter as determined by 3 measurements in the first 12 weeks after completion of the study. Change in the transfusion requirements served as a secondary end point. Patients had a bone marrow evaluation with cytogenetics prior to and 3 months following treatment. During the course of the study weekly blood counts were obtained.
Criteria for response
Neutrophils | |
| If baseline ANC below 500/mm3 | Increase in ANC ≥ 300/mm3 |
| If baseline ANC above 500/mm3 | Increase in ANC ≥ 500/mm3 |
| Platelets | |
| If baseline platelet count 70 000/mm3 or below but above 50 000/mm3 | Increase in platelet count ≥ 30 000/mm3 |
| If baseline platelet count 50 000/mm3 or below | Increase in platelet count ≥ 20 000/mm3 blood |
| Transfusion dependence | |
| Transfusion independence | No transfusions for > 8 wk |
| Decreased requirement | Decrease in transfusion number over time >50% for either RBC, platelets, or both |
Neutrophils | |
| If baseline ANC below 500/mm3 | Increase in ANC ≥ 300/mm3 |
| If baseline ANC above 500/mm3 | Increase in ANC ≥ 500/mm3 |
| Platelets | |
| If baseline platelet count 70 000/mm3 or below but above 50 000/mm3 | Increase in platelet count ≥ 30 000/mm3 |
| If baseline platelet count 50 000/mm3 or below | Increase in platelet count ≥ 20 000/mm3 blood |
| Transfusion dependence | |
| Transfusion independence | No transfusions for > 8 wk |
| Decreased requirement | Decrease in transfusion number over time >50% for either RBC, platelets, or both |
Flow cytometry
Intracellular staining for interferon γ (IFN-γ) expression was performed using the PharMingen intracellular staining kit (San Diego, CA).17 Double-color surface staining was first performed with phycoerythrin (PE)-conjugated anti-CD4 and anti-CD8 mAbs and then cells were permeabilized using a saponin-based method (PharMingen) and stained with fluorescein isothiocyanate (FITC)-anti-IFN-γ or IL-4 mAbs (PharMingen; BioSource, Camarillo, CA). Specificity of the antibody was confirmed by showing elimination of staining with blocking antibody. To perform this test 10 μg of a purified unconjugated antibody was added to the fixed/permeabilized cells, which was then incubated for 20 minutes prior to addition of the conjugated antibody. Samples were analyzed using the Coulter EPICS V flow cytometer (Hialeah, FL). Lymphocytes were gated initially by forward scatter/side scatter; secondary gates were set based on staining with isotypic control mAbs such that less than 1% of cells stained positive. Staining was compared to that for 30 normal controls. Results of 30 normal controls as well as the reproducibility of the technique were previously published by our laboaratory.17
Results and discussion
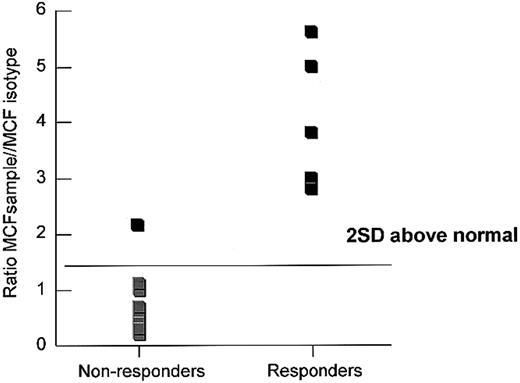
Six of 16 evaluable patients (38%) with mAA responded to treatment within 90 days of receiving the last dose of daclizumab (Table 2; Figure 1). One patient, lost to follow-up, was excluded from analysis and was not evaluated at the 3-month evaluation period. Of the 16 evaluable patients with mAA (one patient was lost to follow-up), 3 were newly diagnosed and 13 had chronic mAA. Patients no. 1 and 2 with previously chronic disease showed complete return of normal blood counts within the first 4 months of receiving daclizumab. Single-lineage responses were seen in 3 patients with chronic disease (patients no. 3, 9, and 16). Two patients (nos. 1 and 9) previously dependent on RBC transfusions no longer require transfusions after more than 2 years of follow-up. Another patient (no.3), who had experienced many neutropenia-related infections, now has a normal white blood cell count. Only one patient progressed despite treatment and was later treated with ATG/CsA and responded. Although follow-up time was limited, only one patient had a relapse, but she responded promptly to further daclizumab treatments. Daclizumab was associated with no toxicity except for a mild first infusion reaction in one patient (itching), which did not require treatment and did not recur. As in a previous study of sAA,17 CD8 cells from 11 patients were tested for the presence of IFN-γ; 5 of the 6 testing positive by the assay responded to daclizumab, whereas all 5 patients testing negative failed to clinically respond to daclizumab (Figure 2).
Patient characteristics
Patient no. . | . | . | . | ANC/mm3 . | . | Platelet count/mm3 . | . | Reticulocyte count/mm3 . | . | . | . | Follow-up, mo . | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| . | Age, y . | Ethnicity . | Sex . | Before . | After . | Before . | After . | Before . | After . | TI* . | Response . | . | |||
| 1 | 57 | White | F | 1852 | 3148 | 45 | 78 | 55 | 134 | T1 | CR at 5 mo | 31 | |||
| 2 | 43 | White | M | 670 | 1360 | 45 | 98 | 49 | 45 | CR at 4 mo | 32 | ||||
| 3 | 58 | White | F | 667 | 1490 | 78 | 80 | 57 | 55 | PR | 25 | ||||
| 4 | 14 | Asian | F | 2832 | ND | 5 | ND | 4 | ND | LTF | LTF | ||||
| 5 | 53 | White | F | 888 | 786 | 55 | 61 | 96 | 66 | NR | 25 | ||||
| 6 | 43 | Hispanic | F | 1200 | 1398 | 65 | 32 | 119 | 153 | NR | 24 | ||||
| 7 | 41 | White | M | 625 | 494 | 34 | 39 | 72 | 79 | NR | 23 | ||||
| 8 | 30 | White | M | 616 | 584 | 24 | 33 | 53 | 51 | NR | 22 | ||||
| 9 | 9 | White | F | 1088 | 2400 | 35 | 30 | 22 | 134 | T1 | PR | 21 | |||
| 10 | 37 | White | F | 3012 | 3229 | 33 | 26 | 21 | 28 | NR | 19 | ||||
| 11 | 40 | Hispanic | F | 760 | 1124 | 18 | 20 | 58 | 59 | NR | 17 | ||||
| 12 | 30 | Hispanic | M | 1068 | 1225 | 115 | 111 | 22 | 22 | NR | 20 | ||||
| 13 | 25 | White | M | 1293 | 1060 | 33 | 36 | 71 | 102 | NR | 4 | ||||
| 14 | 26 | White | M | 804 | 928 | 23 | 22 | 34 | 36 | NR | 10 | ||||
| 15 | 67 | Asian | M | 800 | 100 | 47 | 57 | 70 | 115 | PR | 13 | ||||
| 16 | 38 | White | F | 1134 | 1800 | 51 | 51 | 35 | 105 | PR | 11 | ||||
| 17 | 24 | Asian | F | 1378 | 500 | 25 | 24 | 72 | 65 | NR | 20 | ||||
Patient no. . | . | . | . | ANC/mm3 . | . | Platelet count/mm3 . | . | Reticulocyte count/mm3 . | . | . | . | Follow-up, mo . | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| . | Age, y . | Ethnicity . | Sex . | Before . | After . | Before . | After . | Before . | After . | TI* . | Response . | . | |||
| 1 | 57 | White | F | 1852 | 3148 | 45 | 78 | 55 | 134 | T1 | CR at 5 mo | 31 | |||
| 2 | 43 | White | M | 670 | 1360 | 45 | 98 | 49 | 45 | CR at 4 mo | 32 | ||||
| 3 | 58 | White | F | 667 | 1490 | 78 | 80 | 57 | 55 | PR | 25 | ||||
| 4 | 14 | Asian | F | 2832 | ND | 5 | ND | 4 | ND | LTF | LTF | ||||
| 5 | 53 | White | F | 888 | 786 | 55 | 61 | 96 | 66 | NR | 25 | ||||
| 6 | 43 | Hispanic | F | 1200 | 1398 | 65 | 32 | 119 | 153 | NR | 24 | ||||
| 7 | 41 | White | M | 625 | 494 | 34 | 39 | 72 | 79 | NR | 23 | ||||
| 8 | 30 | White | M | 616 | 584 | 24 | 33 | 53 | 51 | NR | 22 | ||||
| 9 | 9 | White | F | 1088 | 2400 | 35 | 30 | 22 | 134 | T1 | PR | 21 | |||
| 10 | 37 | White | F | 3012 | 3229 | 33 | 26 | 21 | 28 | NR | 19 | ||||
| 11 | 40 | Hispanic | F | 760 | 1124 | 18 | 20 | 58 | 59 | NR | 17 | ||||
| 12 | 30 | Hispanic | M | 1068 | 1225 | 115 | 111 | 22 | 22 | NR | 20 | ||||
| 13 | 25 | White | M | 1293 | 1060 | 33 | 36 | 71 | 102 | NR | 4 | ||||
| 14 | 26 | White | M | 804 | 928 | 23 | 22 | 34 | 36 | NR | 10 | ||||
| 15 | 67 | Asian | M | 800 | 100 | 47 | 57 | 70 | 115 | PR | 13 | ||||
| 16 | 38 | White | F | 1134 | 1800 | 51 | 51 | 35 | 105 | PR | 11 | ||||
| 17 | 24 | Asian | F | 1378 | 500 | 25 | 24 | 72 | 65 | NR | 20 | ||||
Seventeen patients with mAA were treated with daclizumab. Of these, 16 were evaluable; patient no. 5 was lost to follow-up before the 3-month evaluation period. The ANC, absolute reticulocyte count, and platelet counts on entry to the study are shown.
Blood counts are the average of 3 consecutive measurements performed prior to study entry and 3 months after the last dose of daclizumab (first course as in the case of patient 1).
TI indicates transfusion independence; CR, complete response; PR, partial response; ND, not done; LTF, lost to follow-up; NR, no response.
Transfusion independence in patients who were previously transfusion dependent.
Response of patients with mAA to daclizumab. Serial blood counts of patients treated with daclizumab. Each panel represents data from 1 patient. Platelets and red cells were given to Patient 1 (panel A) for bleeding.
Response of patients with mAA to daclizumab. Serial blood counts of patients treated with daclizumab. Each panel represents data from 1 patient. Platelets and red cells were given to Patient 1 (panel A) for bleeding.
IFN-γ staining of peripheral blood lymphocytes of responding and nonresponding patients with mAA. Lymphocytes from 11 previously untreated patients with mAA were surface stained with CD4-tricolor (phycoerythrin-cyanine 5) and CD8-PE and intracellularly stained with IFN-γ-FITC. Patients with mAA received daclizumab therapy and were designated as responders or nonresponders based on improvement of bone marrow function by 3 months following treatment. The figure shows the ratio of MCFsample/MCFisotype of staining of peripheral blood lymphocytes. Values more than 2 SD above the mean of normal were said to be positive for IFN-γ.17 MCF indicates mean channel fluorescence.
IFN-γ staining of peripheral blood lymphocytes of responding and nonresponding patients with mAA. Lymphocytes from 11 previously untreated patients with mAA were surface stained with CD4-tricolor (phycoerythrin-cyanine 5) and CD8-PE and intracellularly stained with IFN-γ-FITC. Patients with mAA received daclizumab therapy and were designated as responders or nonresponders based on improvement of bone marrow function by 3 months following treatment. The figure shows the ratio of MCFsample/MCFisotype of staining of peripheral blood lymphocytes. Values more than 2 SD above the mean of normal were said to be positive for IFN-γ.17 MCF indicates mean channel fluorescence.
The goal of immunosuppressive regimens in AA is elimination of T cells effecting the immune-mediated destruction of hematopoietic progenitor and stem cells. Although this study included only patients who were symptomatic from their bone marrow failure, immunosuppressive therapy, if sufficiently innocuous and also convenient, could potentially be used to prevent stem cell depletion in patients with evidence of immune-mediated bone marrow destruction in an early phase of hematologic disease. The relapse rate after treatment with daclizumab is not known at this point, but the one relapse reported here might be indicative of the need for further immunosuppression in some patients. Further long-term studies are needed both to determine systematically the effect of early treatment on the course of mAA, as well as to establish the optimal dosing regimen for daclizumab and the need for further immunosuppression. Other roles for daclizumab in AA can now be envisioned including treatment of relapsed sAA, in myelodysplastic syndrome, for pure RBC aplasia and other single-lineage failure diseases, and as a replacement for CsA in treatment-dependent patients with sAA where CsA is contraindicated or has shown toxicity.
Prepublished online as Blood First Edition Paper, July 24, 2003; DOI 10.1182/blood-2003-04-1032.
J.P.M. and E.M.S. contributed equally to this study.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal